Abstract
Introduction
Bacterial infection and aseptic loosening caused by bone resorption at the implant interface are major clinical complications during bone defect implantation surgery, and surface modification of the implant to address the aforementioned problems has long been a research focus.
Materials and methods
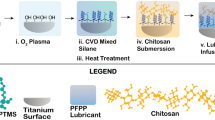
In this paper, a chitosan (CTS)–tannic acid (TA) colloid coating with a negative charge and excellent hydrophilicity was prepared on a Ti6Al4V (TC4) surface using a layer-by-layer assembly method. The physical properties, anti-osteoclast activity, and antimicrobial activity of the coatings were investigated.
Results
The findings showed that when the pH value was 5 and the ratio of CTS:TA was 0.8, the carrying rate of TA was the best. Furthermore, the CTS–TA coating had no cytotoxicity on the morphology and proliferation of BMSCs cells and effectively inhibited the differentiation of RAW264.7 cells into osteoclasts and the proliferation of Staphylococcus aureus and Escherichia coli. With the increase in the immersion time of TC4 in CTS–TA colloid solution, the inhibitory effects will also enhance.
Conclusion
Therefore, the preparation of the CTS–TA coating provides a revolutionary technique for implant surface modification to avoid postoperative bacterial infection and aseptic loosening.














Similar content being viewed by others
References
Kobbe P, Laubach M, Hutmacher DW, Alabdulrahman H, Sellei RM, Hildebrand F (2020) Convergence of scaffold-guided bone regeneration and RIA bone grafting for the treatment of a critical-sized bone defect of the femoral shaft. Eur J Med Res 25:70
Ibrahim M, Sarhan A, Yusof F, Hamdi M (2017) Biomedical materials and techniques to improve the tribological, mechanical and biomedical properties of orthopedic implants—a review article. J Alloy Compd 714:636–667
Boden S (2000) Biology of lumbar spine fusion and use of bone graft substitutes: present, future, and next generation. Tissue Eng 6:383–399
Agarwal S (2004) Osteolysis-basic science, incidence and diagnosis. Curr Orthop 18:220–231
Florencio-Silva R, Sasso G, Cerri E, Simoes M, Cerri P (2015) Biology of bone tissue: structure, function, and factors that influence bone cells. BioMed Res Int 2015:421746
Chung M, Song J, Lee J, Shin EJ, Park JH, Lee SH, Hwang JT, Choi HK (2019) Tannic acid, a novel histone acetyltransferase inhibitor, prevents non-alcoholic fatty liver disease both in vivo and in vitro model. Mol Metab 19:112–116
Trisi P, Rao W, Rebaudi A (1999) A histometric comparison of smooth and rough titanium implants in human low-density jawbone. Int J Oral Maxillofac Implants 14:689–698
Heinrich A, Dengler K, Koerner T, Haczek C, Deppe H, Stritzker B (2008) Laser-modified titanium implants for improved cell adhesion. Lasers Med Sci 23:55–58
Uezono M, Takakuda K, Kikuchi M, Suzuki S, Moriyama K (2013) Hydroxyapatite/collagen nanocomposite-coated titanium rod for achieving rapid osseointegration onto bone surface. J Biomed Mater Res B Appl Biomater 101:1031–1038
Schliephake H, Scharnweber D, Dard M, Rößler S, Sewing A (2002) Effect of RGD peptide coating of titanium implants on periimplant bone formation in the alveolar crest : an experimental pilot study in dogs. Clin Oral Implants Res 42:312–319
Wu Y, Zhong L, Yu Z, Qi J (2019) Anti-neuroinflammatory effects of tannic acid against lipopolysaccharide-induced BV2 microglial cells via inhibition of NF-KB activation. Drug Dev Res 80:715–718
Reddy D, Lee SM (2013) Application of magnetic chitosan composites for the removal of toxic metal and dyes from aqueous solutions. Adv Coll Interface Sci 201:68–93
Steffi C, Shi Z, Kong CH, Wang W (2019) Bioinspired polydopamine and polyphenol tannic acid functionalized titanium suppress osteoclast differentiation: a facile and efficient strategy to regulate osteoclast activity at bone-implant interface. J R Soc Interf 16:2018–2019
Yang X, Hu M, Chen S, Fang C, Cheng W, Yong T, Du C, Wang X, Hao Z, Shen M (2018) Tannic acid attenuated irradiation-induced apoptosis in megakaryocytes. Exp Cell Res 370:409–416
Steffi C, Shi ZL, Kong CH, Chong SW, Wang D, Wang W (2019) Use of polyphenol tannic acid to functionalize titanium with strontium for enhancement of osteoblast differentiation and reduction of osteoclast activity. Polymers (Basel) 11:1256–1259
Lee D, Lee SJ, Moon J-H, Kim J, Heo DN, Bang J, Lim H-N, Kwon IK (2018) Preparation of antibacterial chitosan membranes containing silver nanoparticles for dental barrier membrane applications. J Ind Eng Chem 66:196–202
Adeli H, Khorasani M, Parvazinia M (2019) Wound dressing based on electrospun PVA/chitosan/starch nanofibrous mats: fabrication, antibacterial and cytocompatibility evaluation and in vitro healing assay. Int J Biol Macromol 122:238–254
Gopalakrishnan L, Ramana LN, Sethuraman S (2014) Ellagic acid encapsulated chitosan nanoparticles as anti-hemorrhagic agent. Carbohydr Polym 111:215–221
Ferris C, Casas M, Lucero MJ, Paz MD, Jiménez-Castellanos MR (2014) Synthesis and characterization of a novel chitosan-N-acetyl-homocysteine thiolactone polymer using MES buffer. Carbohyd Polym 111:125–132
Fu JH, Ji J, Fan DZ, Shen JC (2010) Construction of antibacterial multilayer films containing nanosilver via layer-by-layer assembly of heparin and chitosan-silver ions complex. J Biomed Mater Res Part A 79:665–674
Costa E, Coelho M, Ilharco LM, Aguiar-Ricardo A, Hammond PT (2011) Tannic acid mediated suppression of PNIPAAm microgels thermoresponsive behavior. Macromolecules 44:612–621
Aelenei N, Popa I, Novac O, Lisa G, Balaita L (2009) Tannic acid incorporation in chitosan-based microparticles and in vitro controlled release. J Mater Sci Mater Med 20:1095–1102
Donlan R (2001) Biofilms and device-associated infections. Emerg Infect Dis 7:277–281
Piyapakorn P, Akagi T, Hachisuka M, Onishi T, Matsuoka H, Akashi M (2013) Structural analysis of unimer nanoparticles composed of hydrophobized polys (amino acid). Macromolecules 46:6187–6194
Jung BO, Kim CH, Choi KS, Lee YM, Kim JJ (1999) Preparation of amphiphilic chitosan and their antimicrobial activities. J Appl Polym Sci 72:1713–1719
M. Kamachi, A. Nakamura, New macromolecular architecture and functions: proceedings of the OUMS’95 Toyonaka, Osaka, Japan, 2–5 June, 1995[M]. 1996.
Ming K, Xi GC, Cheng SL, Chen GL, Xiang HM, Le JY (2008) Antibacterial mechanism of chitosan microspheres in a solid dispersing system against E. coli. Coll Surf B 65:197–202
Sudarshan NR, Hoover DG (1992) Antibacterial action of chitosan. Food Biotechnol 6:257–272
Guo ZY, Xing RG, Liu S, Zhong ZM, Wang XJL, Li PC (2007) The influence of the cationic of quaternized chitosan on antifungal activity. Int J Food Microbiol 118:214–217
Mellegård H, Strand S, Christensen B, Granum P, Hardy S (2011) Antibacterial activity of chemically defined chitosans: influence of molecular weight, degree of acetylation and test organism. Int J Food Microbiol 148:48–54
Guo ZY, Xing RG, Liu S, Zhong ZM, Wang XJL, Li PC (2008) The influence of molecular weight of quaternized chitosan on antifungal activity. Carbohydr Polym 71:694–697
Li MQ, Chen XG, Liu JM, Tang WFXX (2010) Molecular weight-dependent antifungal activity and action mode of chitosan against Fulvia fulva (cooke) ciffrri. J Appl Polym Sci 119:3127–3135
Acknowledgements
This work was supported by the Key R&D Project of Sichuan Province (2018JY0552) and the National Natural Science Foundation of China (No. 51675447).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Ethical approval
In our study, the cells used were derived from ATCC, no live animal experiments were performed, so there was no relevant ethical approval.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Liao, B., Xu, C., Wang, Z. et al. Preparation of chitosan–tannic acid coating and its anti-osteoclast and antibacterial activities in titanium implants. J Bone Miner Metab 40, 402–414 (2022). https://doi.org/10.1007/s00774-022-01309-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00774-022-01309-7