Abstract
Introduction
Alveolar bone, dentin, and cementum provide a striking example of structurally different collagen-based mineralized tissues separated only by periodontal ligament. While alveolar bone is strongly remodeled, this does not hold for dentin and cementum. However, additional dentin can be deposited on the inner surface of the pulp chamber also in older age. By investigating alveolar bone and molar of mice, the aim of our study is to detect changes in the mineral nanostructure with aging.
Materials and methods
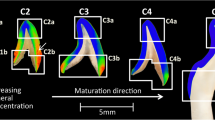
Buccal-lingual sections of the mandible and first molar from C57BL/6 mice of three different age groups (young 5 weeks, adult 22 weeks and old 23 months) were characterized using synchrotron small and wide-angle X-ray scattering. Local average thickness and length of the apatite particles were mapped with several line scans covering the alveolar bone and the tooth.
Results
In alveolar bone, a spatial gradient was seen to develop with age with the thickest and longest particles in the distal part of the bone. The mineral particles in dentin were found to be become thicker, but then decrease of average length from adult to old animals. The mineral particle characteristics of dentin close to the pulp chamber were not only different to the rest of the tooth, but also when comparing the different age groups and even between individual animals in the same age group.
Conclusions
These results indicated that mineral particle characteristics were found to evolve differently between molar and alveolar bone as a function of age.







Similar content being viewed by others
References
Naveh GRS, Chattah NLT, Zaslansky P, Shahar R, Weiner S (2012) Tooth-PDL-bone complex: response to compressive loads encountered during mastication—a review. Arch Oral Biol 57:1575–1584
Dunlop JW, Weinkamer R, Fratzl P (2011) Artful interfaces within biological materials. Mater Today 14:70–78
Ho SP, Marshall SJ, Ryder MI, Marshall GW (2007) The tooth attachment mechanism defined by structure, chemical composition and mechanical properties of collagen fibers in the periodontium. Biomaterials 28:5238–5245
Ho SP, Kurylo MP, Fong TK, Lee SS, Wagner HD, Ryder MI, Marshall GW (2010) The biomechanical characteristics of the bone-periodontal ligament–cementum complex. Biomaterials 31:6635–6646
Naveh GR, Foster JE, Santisteban TMS, Yang X, Olsen BR (2018) Nonuniformity in ligaments is a structural strategy for optimizing functionality. Proc Natl Acad Sci 115:9008–9013
Nikolaus A, Currey JD, Lindtner T, Fleck C, Zaslansky P (2017) Importance of the variable periodontal ligament geometry for whole tooth mechanical function: a validated numerical study. J Mech Behav Biomed 67:61–73
Ho SP, Kurylo MP, Grandfield K, Hurng J, Herber R-P, Ryder MI, Altoe V, Aloni S, Feng JQ, Webb S (2013) The plastic nature of the human bone–periodontal ligament–tooth fibrous joint. Bone 57:455–467
Weinkamer R, Fratzl P (2011) Mechanical adaptation of biological materials—the examples of bone and wood. Mater Sci Eng C 31:1164–1173
Meikle MC (2006) The tissue, cellular, and molecular regulation of orthodontic tooth movement: 100 years after Carl Sandstedt. Eur J Orthod 28:221–240
Goldberg M, Kulkarni AB, Young M, Boskey A (2011) Dentin: structure, composition and mineralization: the role of dentin ECM in dentin formation and mineralization. Front Biosci (Elite Ed) 3:711
Tjäderhane L, Carrilho MR, Breschi L, Tay FR, Pashley DH (2009) Dentin basic structure and composition—an overview. Endod Top 20:3–29
Tjäderhane L, Haapasalo M (2012) The dentin–pulp border: a dynamic interface between hard and soft tissues. Endod Top 20:52–84
Tziafas D (2007) Dentinogenic potential of the dental pulp: facts and hypotheses. Endod Top 17:42–64
Montoya C, Arango-Santander S, Peláez-Vargas A, Arola D, Ossa E (2015) Effect of aging on the microstructure, hardness and chemical composition of dentin. Arch Oral Biol 60:1811–1820
Nalla RK, Kruzic JJ, Kinney JH, Ritchie RO (2004) Effect of aging on the toughness of human cortical bone: evaluation by R-curves. Bone 35:1240–1246
Boskey AL, Coleman R (2010) Aging and bone. J Dent Res 89:1333–1348
Zioupos P, Currey J (1998) Changes in the stiffness, strength, and toughness of human cortical bone with age. Bone 22:57–66
Ager JW III, Nalla RK, Balooch G, Kim G, Pugach M, Habelitz S, Marshall GW, Kinney JH, Ritchie RO (2006) On the increasing fragility of human teeth with age: a deep-UV resonance Raman study. J Bone Miner Res 21:1879–1887
Nazari A, Bajaj D, Zhang D, Romberg E, Arola D (2009) Aging and the reduction in fracture toughness of human dentin. J Mech Behav Biomed 2:550–559
Montoya C, Arola D, Ossa EA (2018) Deformation behaviour of aged coronal dentin. Gerodontology 35:95–100
Saito M, Marumo K (2010) Collagen cross-links as a determinant of bone quality: a possible explanation for bone fragility in aging, osteoporosis, and diabetes mellitus. Osteoporosis Int 21:195–214
Akkus O, Adar F, Schaffler MB (2004) Age-related changes in physicochemical properties of mineral crystals are related to impaired mechanical function of cortical bone. Bone 34:443–453
Jäger I, Fratzl P (2000) Mineralized collagen fibrils: a mechanical model with a staggered arrangement of mineral particles. Biophys J 79:1737–1746
Zizak I, Roschger P, Paris O, Misof BM, Berzlanovich A, Bernstorff S, Amenitsch H, Klaushofer K, Fratzl P (2003) Characteristics of mineral particles in the human bone/cartilage interface. J Struct Biol 141:208–217
Tang T, Wagermaier W, Schuetz R, Wang Q, Eltit F, Fratzl P, Wang R (2019) Hypermineralization in the femoral neck of the elderly. Acta Biomater 89:330–342
Chatterji S, Wall JC, Jeffery JW (1981) Age-related changes in the orientation and particle size of the mineral phase in human femoral cortical bone. Calcif Tissue Int 33:567–574
Fratzl P, Gupta H, Paschalis E, Roschger P (2004) Structure and mechanical quality of the collagen–mineral nano-composite in bone. J Mater Chem 14:2115–2123
Razi H, Birkhold AI, Weinkamer R, Duda GN, Willie BM, Checa S (2015) Aging leads to a dysregulation in mechanically driven bone formation and resorption. J Bone Miner Res 30:1864–1873
Pabisch S, Wagermaier W, Zander T, Li CH, Fratzl P (2013) Imaging the nanostructure of bone and dentin through small- and wide-angle X-ray scattering. Method Enzymol 532:391–413
Pabisch S, Akabane C, Wagermaier W, Roschger A, Ogura T, Hyodo R, Kataoka S, Tobori N, Okano T, Murakami S, Fratzl P, Weinkamer R (2016) The nanostructure of murine alveolar bone and its changes due to type 2 diabetes. J Struct Biol 196:223–231
Märten A, Fratzl P, Paris O, Zaslansky P (2010) On the mineral in collagen of human crown dentine. Biomaterials 31:5479–5490
Sui T, Sandholzer MA, Le Bourhis E, Baimpas N, Landini G, Korsunsky AM (2014) Structure-mechanical function relations at nano-scale in heat-affected human dental tissue. J Mech Behav Biomed Mater 32:113–124
Tesch W, Eidelman N, Roschger P, Goldenberg F, Klaushofer K, Fratzl P (2001) Graded microstructure and mechanical properties of human crown dentin. Calcified Tissue Int 69:147–157
Sandholzer MA, Sui T, Korsunsky AM, Walmsley AD, Lumley PJ, Landini G (2014) X-ray scattering evaluation of ultrastructural changes in human dental tissues with thermal treatment. J Forensic Sci 59:769–774
Kinney JH, Nalla RK, Pople JA, Breunig TM, Ritchie RO (2005) Age-related transparent root dentin: mineral concentration, crystallite size, and mechanical properties. Biomaterials 26:3363–3376
Roschger P, Fratzl P, Eschberger J, Klaushofer K (1998) Validation of quantitative backscattered electron imaging for the measurement of mineral density distribution in human bone biopsies. Bone 23:319–326
Bloebaum RD, Skedros JG, Vajda EG, Bachus KN, Constantz BR (1997) Determining mineral content variations in bone using backscattered electron imaging. Bone 20:485–490
Paris O, Li CH, Siegel S, Weseloh G, Emmerling F, Riesemeier H, Erko A, Fratzl P (2007) A new experimental station for simultaneous X-ray microbeam scanning for small- and wide-angle scattering and fluorescence at BESSY II. J Appl Crystallogr 40:S466–S470
Fratzl P, Schreiber S, Klaushofer K (1996) Bone mineralization as studied by small-angle X-ray scattering. Connect Tissue Res 35:9–16
Fratzl P, Fratzl-Zelman N, Klaushofer K, Vogl G, Koller K (1991) Nucleation and growth of mineral crystals in bone studied by small-angle X-ray-scattering. Calcified Tissue Int 48:407–413
Benecke G, Wagermaier W, Li C, Schwartzkopf M, Flucke G, Hoerth R, Zizak I, Burghammer M, Metwalli E, Müller-Buschbaum P (2014) A customizable software for fast reduction and analysis of large X-ray scattering data sets: applications of the new DPDAK package to small-angle X-ray scattering and grazing-incidence small-angle X-ray scattering. J Appl Crystallogr 47:1797–1803
Maria R, Ben-Zvi Y, Rechav K, Klein E, Shahar R, Weiner S (2019) An unusual disordered alveolar bone material in the upper furcation region of minipig mandibles: a 3D hierarchical structural study. J Struct Biol 206:128–137
Johannessen L (1961) Dentine apposition in the mandibular first molars of albino rats. Arch Oral Biol 5:81–91
Porter AE, Nalla RK, Minor A, Jinschek JR, Kisielowski C, Radmilovic V, Kinney JH, Tomsia AP, Ritchie RO (2005) A transmission electron microscopy study of mineralization in age-induced transparent dentin. Biomaterials 26:7650–7660
Cölfen H, Antonietti M (2005) Mesocrystals: inorganic superstructures made by highly parallel crystallization and controlled alignment. Angew Chem Int Ed 44:5576–5591
Rao A, Cölfen H (2017) Mineralization schemes in the living world: mesocrystals. New perspectives on mineral nucleation and growth. Springer, pp 155–183
Bar-On B, Wagner DH (2013) Structural motifs and elastic properties of hierarchical biological tissues—a review. J Struct Biol 183:149–164
Acknowledgements
The authors want to thank Birgit Schonert and Gabriele Wienskol for technical support and Stefan Siegel and Chenghao Li for their help at the µ-Spot Beamline, BESSY II (Helmholtz-Zentrum Berlin, Germany). The support of Dr. Michiaki Murakoshi, Mr. Shinsuke Kataoka, Mr. Mitsuo Kimura, Ms. Tsuguno Yamaguchi, Mr. Yasushi Koike, Ms. Yuki Ogawa and Mr. Kenji Egashira is acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
CA, NT, and TO are employees of Lion Corporation. All other authors declare that they have no conflict of interest.
Ethical approval
All experimental procedures were approved by the Animal Care and Use Committee of LION Corporation (Protocol Number: 2014-246, 2015-045).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
About this article
Cite this article
Akabane, C., Pabisch, S., Wagermaier, W. et al. The effect of aging on the nanostructure of murine alveolar bone and dentin. J Bone Miner Metab 39, 757–768 (2021). https://doi.org/10.1007/s00774-021-01227-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00774-021-01227-0