Abstract
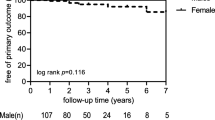
The RDT population, initially at 215 patients, exceeded 300,000 in 2011, with a total of 329,609 patients at the end of December 2016. In our Institute, the number of patients with destructive spondylosis is increasing with the increase in the number of dialysis patients in Japan. We had 14 Cases in the 1990s, and then 82 cases in the 2000s and have already had 131 cases in the 2010s. The purpose of this study was to investigate the incidence of dialysis-related amyloidosis (DRA) such as destructive spondyloarthropathy (DSA), dialysis amyloid arthropathy (DAA), and carpal tunnel syndrome (CTS). In addition, another purpose was to examine the risk factors of the DRA. DAA made its own assessment on radiographs based on stage. Survey items were patient’s basic data, laboratory data and X-ray view. Patientʼs basic data included such as sex, age, height, and weight and RDT-related factors such as kidney disease that led to RDT, age at start of RDT, RDT history, medical history (past and present), and history of surgery. The frequency of DRA was examined by medical history and radiological examination in 199 dialysis patients who obtained informed consent. The patients were divided into two groups according to the presence or absence of DRA, and risk factors of DRA were investigated from the medical history, basic data of patients, and blood tests. Of the 199 patients on regular dialysis therapy, 41 (20.6%) showed DRA. Based on the X-ray images, 21 patients (10.6%) showed DSA, while 22 patients (11.1%) showed DAA. Sixteen patients (8.0%) had CTS, determined through a history of surgery. Regarding overlap of conditions, 14 had both DSA and DAA, 3 had both DSA and CTS, and 2 had both DAA and CTS. There were statistically significant differences between the two groups in the cause of disease in Chronic glomerulonephritis and Diabetic Nephropathy, age at the start of RDT, period of RDT, body weight, blood platelet count, and blood Ca level. When multivariate analysis was performed on these items, statistical differences were recognized only during the dialysis period. In conclusion, long dialysis period was a risk factor for DRA.





Similar content being viewed by others
References
The Japanese Society for Dialysis Therapy An overview of regular dialysis treatment in Japan as of Dec. 31, 2016
Hoshino J, Yamagata K, Nishi S, Nakai S, Masakane I, Iseki K, Tsubakihara Y (2016) Significance of the decreased risk of dialysis-related amyloidosis now proven by results from Japanese nationwide surveys in 1998 and 2010. Nephrol Dial Transplant 31:595–602. https://doi.org/10.1093/ndt/gfv276
Chin M, Hase H, Miyamoto T, Tsuji Y (2006) Radiological grading of cervical destructive spondyloarthropathy in long-term hemodialysis patients. J Spinal Disord Tech 19:430–435
Roderick P, Roth M, Mindell J (2011) Prevalence of chronic kidney disease in England: findings from the 2009 Health Survey for England. J Epidemiol Community Health 65:A12–A12. https://doi.org/10.1136/jech.2011.143586.26
Lee Jon Choon E, Secretary NA, Choong Hui Lin L, et al (2014) Singapore renal registry annual registry report 1999–2013 (Preliminary). Health Promotion Board. National Registry of Diseases Office (NRDO)
Zijlstra M, Bix M, Simister NE, Loring JM, Raulet DH, Jaenisch R (1990) β2-Microglobulin deficient mice lack CD4-8+ cytolytic T cells. Nature 344:742–746. https://doi.org/10.1038/344742a0
Hughes EA, Hammond C, Cresswell P (1997) Misfolded major histocompatibility complex class I heavy chains are translocated into the cytoplasm and degraded by the proteasome. Immunology 94:1896–1901
Gejyo F, Homma N, Suzuki Y, Arakawa M (1986) Serum levels of beta 2-microglobulin as a new form of amyloid protein in patients undergoing long-term hemodialysis. N Engl J Med 314:585–586. https://doi.org/10.1056/NEJM198602273140920
Eichner T, Radford SE (2011) Understanding the complex mechanisms of β 2-microglobulin amyloid assembly. FEBS J 278:3868–3883. https://doi.org/10.1111/j.1742-4658.2011.08186.x
Kuntz D, Naveau B, Bardin T, Drueke T, Treves R, Dryll A (1984) Destructive spondylarthropathy in hemodialyzed patients. A new syndrome. Arthritis Rheum 27:369–375
Fiocchi O, Bedani PL, Orzincolo C, Storari A, Cavazzini PL, Malacarne F, Farinelli A (1989) Radiological features of dialysis amyloid spondyloarthropathy. Int J Artif Organs 12:216–222
Leone A, Sundaram M, Cerase A, Magnavita N, Tazza L, Marano P (2001) Destructive spondyloarthropathy of the cervical spine in long-term hemodialyzed patients: A five-year clinical radiological prospective study. Skeletal Radiol 30:431–441. https://doi.org/10.1007/s002560100389
Yamamoto T, Matsuyama Y, Tsuji T, Nakamura H, Yanase M, Ishiguro N (2005) Destructive spondyloarthropathy in hemodialysis patients comparison between patients with and those without destructive spondyloarthropathy. J Spinal Disord Tech 18:283–285
Naidich JB, Karmel MI, Mossey RT, Bluestone PA, Stein HL (1987) Osteoarthropathy of the hand and wrist in patients undergoing long-term hemodialysis. Radiology 164:205–209. https://doi.org/10.1148/radiology.164.1.3588905
Bardin T, Kuntz D (1987) The arthropathy of chronic haemodialysis. Clin Exp Rheumatol 5:379–386
Duncan IJ, Hurst NP, Sebben R, Milazzo SC, Disney A (1990) Premature development of erosive osteoarthritis of hands in patients with chronic renal failure. Ann Rheum Dis 49:378–382. https://doi.org/10.1136/ARD.49.6.378
Rubin LA, Fam AG, Rubenstein J, Campbell J, Saiphoo C (1984) Erosive azotemic osteoarthropathy. Arthritis Rheum 27:1086–1094. https://doi.org/10.1002/art.1780271002
Sundaram M, Wolverson MK, Heiberg E, Grider RD (1981) Erosive azotemic osteodystrophy. AJR Am J Roentgenol 136:363–367. https://doi.org/10.2214/ajr.136.2.363
Koch KM (1992) Dialysis-related amyloidosis. Kidney Int 41:1416–1429. https://doi.org/10.1038/ki.1992.207
Otsubo S, Kimata N, Okutsu I, Oshikawa K, Ueda S, Sugimoto H, Mitobe M, Uchida K, Otsubo K, Nitta K, Akiba T (2009) Characteristics of dialysis-related amyloidosis in patients on haemodialysis therapy for more than 30 years. Nephrol Dial Transplant 24:1593–1598. https://doi.org/10.1093/ndt/gfn706
Kessler M, Netter P, Azoulay E, Mayeux D, Pere P, Gaucher A (1992) Dialysis-associated arthropathy: a multicentre survey of 171 patients receiving haemodialysis for over 10 years. The Co-operative Group on Dialysis-associated Arthropathy. Br J Rheumatol 31:157–162
De Van Ypersele Strihou C, Jadoul M, Malghem J, Maldague B, Jamart J (1991) Effect of dialysis membrane and patient’s age on signs of dialysis-related amyloidosis. Kidney Int 39:1012–1019. https://doi.org/10.1038/ki.1991.128
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Tagami, A., Tomita, M., Adachi, S. et al. Epidemiological survey and risk factor analysis of dialysis-related amyloidosis including destructive spondyloarthropathy, dialysis amyloid arthropathy, and carpal tunnel syndrome. J Bone Miner Metab 38, 78–85 (2020). https://doi.org/10.1007/s00774-019-01028-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00774-019-01028-6