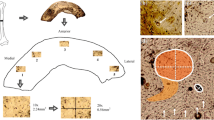
Abstract
Recent quantitative analyses of human bone microanatomy, as well as theoretical models that propose bone microstructure and gross anatomical associations, have started to reveal insights into biological links that may facilitate remodeling processes. However, relationships between bone size and the underlying cortical bone histology remain largely unexplored. The goal of this study is to determine the extent to which static indicators of bone remodeling and vascularity, measured using histomorphometric techniques, relate to femoral midshaft cortical width and robusticity. Using previously published and new quantitative data from 450 adult human male (n = 233) and female (n = 217) femora, we determine if these aspects of femoral size relate to bone microanatomy. Scaling relationships are explored and interpreted within the context of tissue form and function. Analyses revealed that the area and diameter of Haversian canals and secondary osteons, and densities of secondary osteons and osteocyte lacunae from the sub-periosteal region of the posterior midshaft femur cortex were significantly, but not consistently, associated with femoral size. Cortical width and bone robusticity were correlated with osteocyte lacunae density and scaled with positive allometry. Diameter and area of osteons and Haversian canals decreased as the width of cortex and bone robusticity increased, revealing a negative allometric relationship. These results indicate that microscopic products of cortical bone remodeling and vascularity are linked to femur size. Allometric relationships between more robust human femora with thicker cortical bone and histological products of bone remodeling correspond with principles of bone functional adaptation. Future studies may benefit from exploring scaling relationships between bone histomorphometric data and measurements of bone macrostructure.



Similar content being viewed by others
Notes
For the sake of clarity, and to ensure that our study follows standard histomorphometry nomenclature [11], we refer to the cortical distance between the endosteum and periosteum as ‘cortical width’ (defining transverse 2D measurements of diaphyseal cortex) rather than ‘cortical thickness’ (implying 3D measurements) [e.g., 24].
Abbreviations
- Ct.Wi:
-
Cortical width
- Ct.Wi.RI:
-
Cortical width robusticity index
- N.On:
-
Intact osteon density
- N.On.Fg:
-
Fragmentary osteon density
- OPD:
-
Osteon population density
- On.Ar:
-
Osteon area
- H.Ar:
-
Haversian canal area
- H.Dm:
-
Haversian canal diameter
- Ot.Dn:
-
Osteocyte lacunae density
- RMA:
-
Reduced major axis regression
References
Cambra-Moo O, Nacarino-Meneses C, Díaz-Güemes I, Enciso S, Gil OG, Rodríguez LL, Angel Rodríguez MÁ, Antonio BH (2015) Multidisciplinary characterization of the long-bone cortex growth patterns through sheep’s ontogeny. J Struct Biol 191:1–9
Miszkiewicz JJ (2016) Investigating histomorphometric relationships at the human femoral midshaft in a biomechanical context. J Bone Miner Metab 34:179–192
Skedros JG, Su SC, Bloebaum RD (1997) Biomechanical implications of mineral content and microstructural variations in cortical bone of horse, elk, and sheep calcanei. Anat Rec 249:297–316
Bourrin S, Ghaemmaghami F, Vico L, Chappard D, Gharib C, Alexandre C (1992) Effect of a five-week swimming program on rat bone: a histomorphometric study. Calcif Tissue Int 51:137–142
Britz HM, Thomas CDL, Clement JG, Cooper DML (2009) The relation of femoral osteon geometry to age, sex, height and weight. Bone 45:77–83
Dempster DW, Cosman F, Kurland ES, Zhou H, Nieves J, Woelfert L, Shane E, Plavetić K, Müller R, Bilezikian J, Lindsay R (2001) Effects of daily treatment with parathyroid hormone on bone microarchitecture and turnover in patients with osteoporosis: a paired biopsy study. J Bone Miner Res 16:1846–1853
Schlecht SH, Pinto DC, Agnew AM, Stout SD (2012) Brief communication: the effects of disuse on the mechanical properties of bone: what unloading tells us about the adaptive nature of skeletal tissue. Am J Phys Anthropol 149:599–605
Li XJ, Jee WSS, Ke HZ, Mori S, Akamine T (1991) Age-related changes of cancellous and cortical bone histomorphometry in female Sprague-Dawley rats. Cells Mater Suppl 1:25–35
Rauch F, Travers R, Parfitt AM, Glorieux FH (2000) Static and dynamic bone histomorphometry in children with osteogenesis imperfecta. Bone 26:581–589
Pitfield R, Miszkiewicz JJ, Mahoney P (2017) Cortical histomorphometry of the human humerus during ontogeny. Calcif Tissue Int 101:148–158
Dempster DW, Compston JE, Drezner MK, Glorieux FH, Kanis JA, Malluche H, Meunier PJ, Ott SM, Recker RR, Parfitt AM (2013) Standardized nomenclature, symbols, and units for bone histomorphometry: a 2012 update of the report of the ASBMR Histomorphometry Nomenclature Committee. J Bone Miner Res 28:2–17
Chavassieux PM, Arlot ME, Roux JP, Portero N, Daifotis A, Yates AJ, Hamdy NAT, Malice M, Freedholm D, Meunier PJ (2000) Effects of alendronate on bone quality and remodeling in glucocorticoid-induced osteoporosis: a histomorphometric analysis of transiliac biopsies. J Bone Miner Res 15:754–762
Athanasiou KA, Zhu CF, Lanctot DR, Agrawal CM, Wang X (2000) Fundamentals of biomechanics in tissue engineering of bone. Tissue Eng 6:361–381
Frost HM (1987) Bone “mass” and the “mechanostat”: a proposal. Anat Rec 219:1–9
Robling AG, Castillo AB, Turner CH (2006) Biomechanical and molecular regulation of bone remodeling. Annu Rev Biomed Eng 8:455–498
Lanyon LE, Goodship AE, Pye CJ, MacFie JH (1982) Mechanically adaptive bone remodeling. J Biomech 15:141–154
Moustafa A, Sugiyama T, Saxon LK, Zaman G, Sunters A, Armstrong VJ, Javaheri B, Lanyon LE, Price JS (2009) The mouse fibula as a suitable bone for the study of functional adaptation to mechanical loading. Bone 44:930–935
Stock JT, Shaw CN (2007) Which measures of diaphyseal robusticity are robust? A comparison of external methods of quantifying the strength of long bone diaphyses to cross-sectional geometric properties. Am J Phys Anthropol 134:412–423
Stout S, Crowder C (2011) Bone remodeling, histomorphology, and histomorphometry. In: Crowder C, Stout SD (eds) Bone histology: an anthropological perspective. CRC Press, Boca Raton, pp 1–21
Bromage TG, Juwayeyi YM, Katris JA, Gomez S, Ovsiy O, Goldstein J, Janal MN, Hu B, Schrenk F (2016) The scaling of human osteocyte lacuna density with body size and metabolism. C R Palevol 15:33–40
Klein-Nulend J, Bakker AD, Bacabac RG, Vatsa A, Weinbaum S (2012) Mechanosensation and transduction in osteocytes. Bone 54:182–190
Van Oers RFM, Ruimerman R, Van Rietbergen B, Hilbers PAJ, Huiskes R (2008) Relating osteon diameter to strain. Bone 43:476–482
Britz HM, Jokihaara J, Leppänen OV, Järvinen TL, Cooper DM (2012) The effects of immobilization on vascular canal orientation in rat cortical bone. J Anat 220:67–76
Miszkiewicz JJ, Mahoney P (2016) Ancient human bone microstructure in medieval England: comparisons between two socio-economic groups. Anat Rec 299:42–59
Skedros JG, Mason MW, Bloebaum RD (1994) Differences in osteonal micromorphology between tensile and compressive cortices of a bending skeletal system: indications of potential strain-specific differences in bone microstructure. Anat Rec 239:405–413
Wojda SJ, Weyland DR, Gray SK, Mcgee-Lawrence ME, Drummer TD, Donahue SW (2013) Black bears with longer disuse (hibernation) periods have lower femoral osteon population density and greater mineralization and intracortical porosity. Anat Rec 296:1148–1153
Sugawara Y, Kamioka H, Ishihara Y, Fujisawa N, Kawanabe N, Yamashiro T (2013) The early mouse 3D osteocyte network in the presence and absence of mechanical loading. Bone 52:189–196
Totland GK, Fjelldal PG, Kryvi H, Løkka G, Wargelius A, Sagstad A, Hansen T, Grotmol S (2011) Sustained swimming increases the mineral content and osteocyte density of salmon vertebral bone. J Anat 219:490–501
Martin RB (2002) Is all cortical bone remodeling initiated by microdamage? Bone 30:8–13
Kim YK, Kameo Y, Tanaka S, Adachi T (2017) Capturing microscopic features of bone remodeling into a macroscopic model based on biological rationales of bone adaptation. Biomech Model Mechanobiol 16(5):1697–1708
Goldman HM, Hampson NA, Guth JJ, Lin D, Jepsen KJ (2014) Intracortical remodeling parameters are associated with measures of bone robustness. Anat Rec 297:1817–1828
Jepsen KJ, Bigelow EM, Schlecht SH (2015) Women build long bones with less cortical mass relative to body size and bone size compared with men. Clin Orthop Relat Res 473:2530–2539
Tommasini SM, Nasser P, Jepsen KJ (2007) Sexual dimorphism affects tibia size and shape but not tissue-level mechanical properties. Bone 40:498–505
Schlecht SH, Jepsen KJ (2013) Functional integration of skeletal traits: an intraskeletal assessment of bone size, mineralization, and volume covariance. Bone 56:127–138
Buikstra JE, Ubelaker DH (1994) Standards for data collection from human skeletal remains. Arkansas Archaeology Survey, Fayetteville
Miszkiewicz JJ (2014) Ancient human bone histology and behaviour. University of Kent, PhD Thesis
Mahoney P, Miszkiewicz JJ, Chapple S, Le Luyer M, Schlecht SH, Stewart TJ, Griffiths RA, Deter C, Guatelli Steinberg D (2018) The biorhythm of human skeletal growth. J Anat 232(1):26–38
Hammer Ø, Harper DAT, Ryan PD (2001) PAST-palaeontological statistics, ver. 1.89. Palaeont Electr 4:1–9
Smith RJ (2009) Use and misuse of the reduced major axis for line-fitting. Am J Phys Anthropol 140:476–486
Taylor R (1990) Interpretation of the correlation coefficient: a basic review. J Diagn Med Sonogr 6:35–39
Breitling LP (2015) Calcium intake and bone mineral density as an example of non-linearity and threshold analysis. Osteoporos Int 26:1271–1281
Warton DI, Wright IJ, Falster DS, Westoby M (2006) Bivariate line-fitting methods for allometry. Biol Rev 81:259–291
Pearson OM, Lieberman DE (2004) The aging of Wolff’s “law”: ontogeny and responses to mechanical loading in cortical bone. Am J Phys Anthropol 125:63–99
LaMothe JM, Hamilton NH, Zernicke RF (2005) Strain rate influences periosteal adaptation in mature bone. Med Eng Phys 27:277–284
Thompson DW (1942) On growth and form. Cambridge University Press, Cambridge
Barak MM, Lieberman DE, Hublin JJ (2013) Of mice, rats and men: trabecular bone architecture in mammals scales to body mass with negative allometry. J Struct Biol 183:123–131
Thompson DD, Galvin CA (1983) Estimation of age at death by tibial osteon remodeling in an autopsy series. Forensic Sci Int 22:203–211
Nowlan NC, Jepsen KJ, Morgan EF (2011) Smaller, weaker, and less stiff bones evolve from changes in subsistence strategy. Osteoporos Int 22:1967–1980
Yerramshetty JS, Akkus O (2008) The associations between mineral crystallinity and the mechanical properties of human cortical bone. Bone 42:476–482
Currey JD (2003) The many adaptations of bone. J Biomech 36:1487–1495
Code of Ethics of the American Association of Physical Anthropologists (2003) http://physanth.org/documents/3/ethics.pdf. Accessed 24 Jan 2011
British Association for Biological Anthropology and Osteoarchaeology Code of Practice (2010) http://www.babao.org.uk/index/ethics-and-standards. Accessed 24 Jan 2011
Mays S, Elders J, Humphrey L, White W, and Marshall P (2013) Science and the dead: guidelines for the destructive sampling of archaeological human remains for scientific analysis. Advisory panel on the archaeology of burials in England. English Heritage
Acknowledgements
We thank the School of Anthropology and Conservation (University of Kent, UK) for research funding (JJM), The Royal Society for equipment funding (PM), Prof Kate Robson Brown (Bristol), Prof Richard Griffiths (Kent), and Prof Dr David Roberts (Kent) for research support, and Prof Stephen Haslett (ANU), Dr Simon Tollington (Kent), Alannah Pearson (ANU), Dr Julien Louys (ANU) for statistical advice. Invaluable feedback from the editor and two reviewers greatly improved our manuscript.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
About this article
Cite this article
Miszkiewicz, J.J., Mahoney, P. Histomorphometry and cortical robusticity of the adult human femur. J Bone Miner Metab 37, 90–104 (2019). https://doi.org/10.1007/s00774-017-0899-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00774-017-0899-3