Abstract
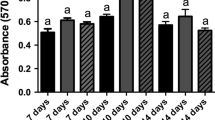
Antioxidant lycopene supplementation has been shown to decrease oxidative stress and have beneficial effects on bone health. However, it remains unclear whether lycopene exerts its beneficial effect on bone metabolism through mitigation of oxidative stress in vivo. The aim of this study was to investigate whether lycopene intake protects against bone loss by reducing oxidative stress in ovariectomized rats. Female Sprague–Dawley 6-week-old rats were ovariectomized and randomly divided into four groups according to the lycopene content of their diet: 0, 50, 100, and 200 ppm. The tibial bone mineral density (BMD) in the 50, 100, and 200 ppm groups was significantly higher than that in the 0 ppm group. Serum and urinary bone resorption marker levels were significantly lower in the 50, 100, and 200 ppm groups than in the 0 ppm group. There was no significant difference in systemic oxidative stress markers among all groups. However, systemic oxidative stress levels were inversely correlated with the tibial BMD. Our findings suggest that lycopene intake significantly inhibits bone loss by suppressing bone resorption in ovariectomized rats. Further studies are necessary to clarify the effect of lycopene on oxidative stress in local tissues such as bone tissue.




Similar content being viewed by others
References
Halliwell B (2011) Free radicals and antioxidants—quo vadis? Trends Pharmacol Sci 32:125–130
Valko M, Leibfritz D, Moncol J, Cronin MTD, Mazur M, Telser J (2007) Free radicals and antioxidants in normal physiological function and human disease. Int J Biochem Cell Biol 39:44–84
Wauquier F, Leotoing L, Coxam V, Guicheux J, Wittrant Y (2009) Oxidative stress in bone remodeling and diseases. Trends Mol Med 15:468–477
Fraser JHE, Helfrich MH, Wallace HM, Ralston SH (1996) Hydrogen peroxide, but not superoxide, stimulates bone resorption in mouse calvaria. Bone 19:223–226
Lee NK, Choi YG, Baik JY, Han SY, Jeong DW, Bae YS, Kim N, Lee SY (2005) A crucial role for reactive oxygen species in RANKL-induced osteoclast differentiation. Blood 106:852–859
Banifi G, Iorio EL, Corsi MM (2008) Oxidative stress, free radical and bone remodeling. Clin Chem Lab Med 46:1550–1555
Weitzmann MN, Pacifici R (2006) Estrogen deficiency and bone loss: an inflammatory tale. J Clin Invest 116:1186–1194
Almeida M, Han L, Martin-Milla M, Plotkin LI, Stewart SA, Roberson PK, Kousteni S, O’Brien CA, Bellido T, Parfitt AM, Weinstein RS, Jilka RL, Manolagas SC (2007) Skeletal involution by age-related oxidative stress and its acceleration by loss of sex steroids. J Biol Chem 282:27285–27297
Hernández I, Delgado JL, Díaz J, Quesada T, Teruel MJG, Lianos MC, Carbonell LF (2000) 17β-estradiol prevents oxidative stress and decreases blood pressure in ovariectomized rats. Am J Physiol Regulatory Integrative Comp Physiol 279:1599–1605
Topçuoglu A, Uzun H, Balci H, Karakus M, Çoban I, Altug T, Aydin S, Topçuoglu D, Çakatay U (2009) Effects of estrogens on oxidative protein damage in plasma and tissues in ovariectomized rats. Clin Invest Med 32:133–143
Bednarek-Tupikowska G, Tworowska U, Jedrychowska I, Radomska B, Tupikowski K, Bidzinska-Speichert B, Milewicz A (2006) Effects of oestradiol and oestroprogestin on erythrocyte antioxidative enzyme system activity in postmenopausal women. Clin Endocrinol 64:463–468
Di Mascio P, Kaiser S, Sies H (1989) Lycopene as the most efficient biological carotenoids singlet oxygen quencher. Arch Biochem Biophys 274:532–538
Mackinnon ES, Rao AV, Josse RG, Rao LG (2011) Supplementation with the antioxidant lycopene significantly decreases oxidative stress parameters and the bone resorption marker N-telopeptide of type I collagen in postmenopausal women. Osteoporosis Int 22:1091–1101
Rao LG, Krishnadev N, Banasikowska K, Rao AV (2003) Lycopene I––Effect on osteoclast: lycopene inhibits basal and parathyroid hormone-stimulated osteoclast formation and mineral resorption mediated by reactive oxygen species in rat bone marrow cultures. J Med Food 6:69–78
Liang H, Yu F, Tong Z, Zeng W (2012) Lycopene effects on serum mineral elements and bone strength in rats. Molecules 17:7093–7102
Cesarone MR, Belcaro G, Carratelli M, Cornelli U, De Sanctis MT, Incandela L, Barsotti A, Terranova R, Nicolaides A (1999) A simple test to monitor oxidative stress. Int Angiol 18:127–130
Alberti A, Bolognini L, Macciantelli D, Caratelli M (2000) The radical cation of N,N-diethyl-para-phenylenediamine: a possible indicator of oxidative stress in biological samples. Res Chem Intermed 26:253–267
Kotani K, Sakane N (2012) C-reactive protein and reactive oxygen metabolites in subjects with metabolic syndrome. J Int Med Res 40:1074–1081
Nagatomo F, Fujino H, Kondo H, Ishihara A (2012) Oxygen concentration-dependent oxidative stress levels in rats. Oxid Med Cell 2012:381763
Kim DJ, Takasuka N, Kim JM, Sekine K, Ota T, Asamoto M, Murakoshi M, Nishino H, Nir Z, Tsuda H (1997) Chemoprevention by lycopene of mouse lung neoplasia after combined initiation treatment with DEN, MNU and DMH. Cancer Lett 120:15–22
Kitade Y, Watanabe S, Masaki T, Nishioka M, Nishino H (2002) Inhibition of liver fibrosis in LEC rats by a carotenoid, lycopene, or a herbal medicine, Sho-saiko-to. Hepatol Res 22:196–205
Omi N, Katayama R, Asano K, Miyakawa N, Nishino H, Ezawa I (2011) The effectiveness of lycopene intake with treadmill running exercise training for bone mineral content increase in growing rats. Jpn Soc Exerc Sports Physiol 18:11–20
Porrini M, Riso P, Testolin G (1998) Absorption of lycopene from single or daily portion of raw and processed tomato. Br J Nutr 80:353–361
Ferreira ALA, Yeum KJ, Liu C, Smith D, Krinsky NI, Wang XD, Russell RM (2000) Tissue distribution of lycopene in ferrets and rats after lycopene supplementation. J Nutr 130:1256–1260
Omi N, Tsukahara N, Ezawa I (2001) Effect of milk on bone metabolism in growing male and female rats. J Home Econ Jpn 52:689–698
Ezawa I, Okada R, Nozaki Y, Ogata E (1979) Breaking-properties and ash contents of femur of growing rat fed a low calcium diet (in Japanese). Nihon Eiyou Shokuryou gakkaishi (J Jpn Soc Nutr Food Sci) 32:329–335
Omi N, Goseki M, Oida S, Sasaki S, Ezawa I (1994) The nutritional evaluation of globin on maintenance of bone metabolism in ovariectomized osteoporotic rats. J Nutr Sci Vitaminol 40:443–457
Robins SP, Woitge H, Hesley R, Ju J, Seyedin S, Seibel MJ (1994) Direct, enzyme-linked immunoassay for urinary deoxypyridinoline as a specific marker for measuring bone resorption. J Bone Miner Res 9:1643–1649
De Zwart LL, Meerman JH, Commandeur JN, Vermeulen NP (1999) Biomarkers of free radical damage applications in experimental animals and in humans. Free Radic Biol Med 26:202–226
Kim L, Rao AV, Rao LG (2003) Lycopene II—Effect on osteoblast: the carotenoid lycopene stimulates cell proliferation and alkaline phosphatase activity of SaOS-2 cells. J Med Food 6:79–86
Iimura Y, Agata U, Takeda S, Kobayashi Y, Yoshida S, Ezawa I, Omi N (2014) Lycopene intake facilitates the increase of bone mineral density in growing female rats. J Nutr Sci Vitaminol 60 (in press)
Dhem A, Goret-Nicaise M (1984) Effects of retinoic acid on rat bone. Food Chem Toxic 22:199–206
Kneissel M, Studer A, Cortesi R, Susa M (2005) Retinoid-induced bone thinning is caused by subperiosteal osteoclast activity in adult rodents. Bone 36:202–214
Lind T, Sundqvist A, Hu L, Pejler G, Andersson G, Jacobson A, Melhus H (2013) Vitamin A is a negative regulator of osteoblast mineralization. PLoS One 8:e82388
Maggio D, Polidori MC, Barabani M, Tufi A, Ruggiero C, Cecchetti R, Aisa MC, Stahl W, Cherubini A (2006) Low levels of carotenoids and retinol in involutional osteoporosis. Bone 38:244–248
Ferreira AL, Yeum KJ, Russell RM, Krinsky NI, Tang G (2004) Enzymatic and oxidative metabolites of lycopene. J Nutr Biochem 15:493–502
Sthal W, Sies H (1996) Lycopene: a biologically important carotenoid for humans? Arch Biochem Biophys 336:1–9
Lee CM, Boileau AC, Boileau TWM, Williams AW, Swanson KS, Heintz KA, Erdman JW Jr (1999) Review of animal model in carotenoids research. J Nutr 129:2271–2277
Al-Malki AL, Moselhy SS, Refai MY (2011) Synergistic effect of lycopene and tocopherol against oxidative stress and mammary tumorigenesis induced by 7,12-dimethyl(a)benzanthracene in female rats. Toxicol Ind Health 28:542–548
Moselhy SS, Almslmani MAB (2008) Chemopreventive effect of lycopene alone or with melatonin against the genesis of oxidative stress and mammary tumors induced by 7, 12 dimethyl(a)benzanthracene in Sprague Dawley female rats. Mol Cell Biochem 319:175–180
Çeribaşi AO, Türk G, Sönmez M, Sakin F, Ateşşahin A (2010) Toxic effect of cyclophosphamide on sperm morphology, testicular histology and blood oxidant–antioxidant balance, and protective roles of lycopene and ellagic acid. Basic Clin Pharmacol Toxicol 107:730–736
Lean JM, Davies JT, Fuller K, Jagger CJ, Kirstein B, Partington GA, Urry ZL, Chambers TJ (2003) A crucial role for thiol antioxidants in estrogen-deficiency bone loss. J Clin Invest 112:915–923
Pacifici R (1996) Estrogen, cytokines, and pathogenesis of postmenopausal osteoporosis. J Bone Miner Res 11:1043–1051
Jagger CJ, Lean JM, Davies JT, Chambers TJ (2005) Tumor necrosis factor-alpha mediates osteopenia caused by depletion of antioxidants. Endocrinology 146:113–118
Grassi F, Tell G, Robbie-Ryan M, Gao Y, Terauchi M, Yang X, Romanello M, Jones DP, Weitzmann MN, Pacifici R (2007) Oxidative stress causes bone loss in estrogen-deficient mice through enhanced bone marrow dendritic cell activation. Proc Natl Acad Sci USA 104:15087–15092
Novack DV (2011) Role of NF-κB in the skeleton. Cell Res 21:169–182
Gouranton E, Thabuis C, Riollet C, Malezet-Desmoulins C, El Yazidi C, Amiot MJ, Borel P, Landrier JF (2011) Lycopene inhibits proinflammatory cytokine and chemokine expression in adipose tissue. J Nutr Biochem 22:642–648
Palozza P, Simone R, Catalano A, Monego G, Barini A, Mele MC, Parrone N, Trombino S, Picci N, Ranelletti FO (2011) Lycopene prevention of oxysterol-induced proinflammatory cytokine cascade in human macrophages: inhibition of NF-κB nuclear binding and increase in PPARγ expression. J Nutr Biochem 22:259–268
Batra N, Kar R, Jiang JX (2012) Gap junctions and hemichannels in signal transmission, function and development of bone. Biochim Biophys Acta 1818:1909–1918
Acknowledgments
The authors would like to thank Kyoritsu Bussan Co., Ltd., Japan, for supplying the lycopene samples.
Conflict of interest
All authors have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Iimura, Y., Agata, U., Takeda, S. et al. The protective effect of lycopene intake on bone loss in ovariectomized rats. J Bone Miner Metab 33, 270–278 (2015). https://doi.org/10.1007/s00774-014-0596-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00774-014-0596-4