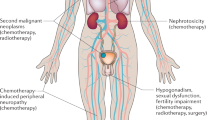
Abstract
Background
Modern oncological treatment has rendered testicular cancer (TC) a curative malignant disease but there is a risk of decreasing survival and reduced health-related quality of life (HR-QoL) related to long-term adverse health outcomes (AHOs).
Aim
To provide an overview on AHOs after oncological treatment of TC.
Material and methods
Summary of published studies and previous reviews.
Results
Relative survival rates decrease among TC survivors 25 years after diagnosis, mainly due to treatment-related second cancer and/or cardiovascular disease, the latter mediated by components of the metabolic syndrome. With increasing age cisplatin-induced ototoxicity becomes a clinical problem in patients, whereas reported peripheral neurotoxicity only exceptionally achieves major clinical relevance. Anxiety but not depression represents the dominating psychological problem. In most patients HR-QoL is good, but working ability may be reduced by very intensive treatment.
Conclusion
Awareness of long-term AHOs among TC survivors and health professionals can contribute to reduce long-term morbidity and mortality and to improve QoL by initiating early preventive and therapeutic measures. Radiotherapy should be avoided as much as possible during risk-adapted treatment of TC. Existing data indicate premature aging among strongly treated TC survivors but larger studies and longer follow-up in longitudinal studies are required to confirm these preliminary observations.



Similar content being viewed by others
References
Bandak M, Jorgensen N, Juul A, Vogelius IR, Lauritsen J, Kier MG et al (2016) Testosterone deficiency in testicular cancer survivors—a systematic review and meta-analysis. Andrology 4:382–388
Bower JE (2014) Cancer-related fatigue—mechanisms, risk factors, and treatments. Nat Rev Clin Oncol 11:597–609
Bower JE, Bak K, Berger A, Breitbart W, Escalante CP, Ganz PA, American Society of Clinical Oncology, - (2014) Screening, assessment, and management of fatigue in adult survivors of cancer: an American Society of Clinical oncology clinical practice guideline adaptation. J Clin Oncol 32:1840–1850
Brydoy M, Fossa SD, Klepp O, Bremnes RM, Wist EA, Wentzel-Larsen T, Dahl O (2005) Paternity following treatment for testicular cancer. J Natl Cancer Inst 97:1580–1588
Cappuccio F, Rossetti S, Cavaliere C, Iovane G, Taibi R, D’Aniello C et al (2018) Health-related quality of life and psychosocial implications in testicular cancer survivors. A literature review. Eur Rev Med Pharmacol Sci 22:645–661
Chovanec M, Zaid AM, Hanna N, El-Kouri N, Einhorn LH, Albany C (2017) Long-term toxicity of cisplatin in germ-cell tumor survivors. Ann Oncol 28:2670–2679
Classen J, Schmidberger H, Meisner C, Winkler C, Dunst J, Souchon R et al (2004) Para-aortic irradiation for stage I testicular seminoma: results of a prospective study in 675 patients. A trial of the German testicular cancer study group (GTCSG). Br J Cancer 90:2305–2311
Eberhard J, Ståhl O, Cohn-Cedermark G, Cavallin-Ståhl E, Giwercman Y, Rylander L et al (2009) Sexual function in men treated for testicular cancer. J Sex Med 6:1979–1989
Einhorn LH, Donohue J (1977) Cis-diamminedichloroplatinum, vinblastine, and bleomycin combination chemotherapy in disseminated testicular cancer. Ann Intern Med 87:293–298
Feldman DR, Ardeshir-Rouhani-Fard S, Monahan P, Sesso HD, Fung C, Williams AM et al (2018) Predicting cardiovascular disease among testicular cancer survivors after modern cisplatin-based chemotherapy: Application of the Framingham Risk Score. Clin Genitourin Cancer 4:e761–e769
Fossa SD, Aass N, Winderen M, Börmer OP, Olsen DR (2002) Long-term renal function after treatment for malignant germ-cell tumours. Ann Oncol 13:222–228
Frisina RD, Wheeler HE, Fossa SD, Kerns SL, Fung C, Sesso HD et al (2016) Comprehensive Audiometric Analysis of Hearing Impairment and Tinnitus After Cisplatin-Based Chemotherapy in Survivors of Adult-Onset Cancer. J Clin Oncol 34:2712–2720
Fung C, Fossa SD, Milano MT, Oldenburg J, Travis LB (2013) Solid tumors after chemotherapy or surgery for testicular nonseminoma: a population-based study. J Clin Oncol 31:3807–3814
Fung C, Fossa SD, Milano MT, Sahasrabudhe DM, Peterson DR, Travis LB (2015) Cardiovascular Disease Mortality After Chemotherapy or Surgery for Testicular Nonseminoma: A Population-Based Study. J Clin Oncol 33:3105–3115
Fung C, Dinh P Jr., Ardeshir-Rouhani-Fard S, Scha’er K, Fossa SD, Travis LB (2018) Toxicities associated with cisplatin-based chemotherapy and radiotherapy in long-term testicular cancer survivors. Hindawi Advances in Urology. https://doi.org/10.1155/2018/8671832
Hansen SW, Groth S, Daugaard G, Rossing N, Rørth M (1988) Long-term effects on renal function and blood pressure of treatment with cisplatin, vinblastine, and bleomycin in patients with germ cell cancer. J Clin Oncol 6:1728–1731
Haugnes HS, Bosl GJ, Boer H, Gietema JA, Brydøy M, Oldenburg J et al (2012) Long-term and late effects of germ cell testicular cancer treatment and implications for follow-up. J Clin Oncol 30:3752–3763
Haugnes HS, Stenklev NC, Brydøy M, Dahl O, Wilsgaard T, Laukli E, Fosså SD (2018) Hearing loss before and after cisplatin-based chemotherapy in testicular cancer survivors: a longitudinal study. Acta Oncol 31:1–9
Hershman DL, Lacchetti C, Dworkin RH, Lavoie Smith EM, Bleeker J, Cavaletti G et al (2014) Prevention and management of chemotherapy-induced peripheral neuropathy in survivors of adult cancers: American Society of Clinical Oncology clinical practice guideline. J Clin Oncol 32:1941–1967
Jankowska M (2011) Sexual functioning of testicular cancer survivors and their partners—A review of literature. Rep Pract Oncol Radiother 17:54–62
Kier MG, Hansen MK, Lauritsen J, Mortensen MS, Bandak M, Agerbaek M et al (2016) Second malignant neoplasms and cause of death in patients with germ cell cancer: A Danish Nationwide Cohort Study. Jama Oncol 2:1624–1627
Kvammen Ø, Myklebust TÅ, Solberg A, Møller B, Klepp OH, Fosså SD, Tandstad T (2016) Long-term Relative Survival after Diagnosis of Testicular Germ Cell Tumor. Cancer Epidemiol Biomarkers Prev 25:773–779
Lauritsen J, Kier MG, Bandak M, Mortensen MS, Thomsen FB, Mortensen J, Daugaard G (2016) Pulmonary Function in Patients With Germ Cell Cancer Treated With Bleomycin, Etoposide, and Cisplatin. J Clin Oncol 34:1492–1499
Loren AW, Mangu PB, Beck LN, Brennan L, Magdalinski AJ, Partridge AH et al (2013) Fertility preservation for patients with cancer: American Society of Clinical Oncology clinical practice guideline update. J Clin Oncol 31:2500–2510
Nord C, Olofsson SE, Glimelius I, Cedermark GC, Ekberg S, Cavallin-Ståhl E et al (2015) Sick leave and disability pension among Swedish testicular cancer survivors according to clinical stage and treatment. Acta Oncol 54:1770–1780
Skakkebaek NE, Rajpert-De Meyts E, Main KM (2001) Testicular dysgenesis syndrome: an increasingly common developmental disorder with environmental aspects. Hum Reprod 16:972–978
Smith AB, Rutherford C, Butow P, Olver I, Luckett T, Grimison P et al (2017) A systematic review of quantitative observational studies investigating psychological distress in testicular cancer survivors. Psychooncology. https://doi.org/10.1002/pon.4596 (Epub ahead of print)
Sprauten M, Darrah TH, Peterson DR, Campbell ME, Hannigan RE, Cvancarova M et al (2012) Impact of long-term serum platinum concentrations on neuro- and ototoxicity in Cisplatin-treated survivors of testicular cancer. J Clin Oncol 30:300–307
Sprauten M, Brydoy M, Haugnes HS, Cvancarova M, Bjøro T, Bjerner J et al (2014) Longitudinal serum testosterone, luteinizing hormone, and follicle-stimulating hormone levels in a population-based sample of long-term testicular cancer survivors. J Clin Oncol 32:571–578
Sprauten M, Haugnes HS, Brydøy M et al (2015) Increasing prevalence of chronic fatigue in 812 testicualr cancer survivors: A longitudinal study. Ann Oncol 10:2133–2140
Travis LB, Fosså SD, Schonfeld SJ, McMaster ML, Lynch CF, Storm H et al (2005) Second cancers among 40,576 testicular cancer patients: focus on long-term survivors. J Natl Cancer Inst 97:1354–1365
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
S.D. Fosså, C. Fung and A.A. Dahl declare that they have no competing interests.
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committees and with the 1975 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the mentioned studies.
Rights and permissions
About this article
Cite this article
Fosså, S.D., Fung, C. & Dahl, A.A. Late adverse outcomes after treatment of testicular cancer. Onkologe 24 (Suppl 2), 104–109 (2018). https://doi.org/10.1007/s00761-018-0442-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00761-018-0442-8