Abstract
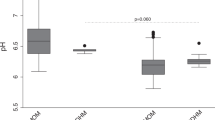
Postpartum depression (PPD) is the most common complication of childbearing with a 13 % prevalence rate, and there is no widespread approach for prevention. There is an appealing theoretical rationale for oral tyrosine to help prevent PPD. However, the effect of oral tyrosine on its total and free concentrations in breast milk and plasma of breastfeeding mothers is not known. Twenty-four healthy breastfeeding women were randomly assigned to 0, 2, 5, or 10 g of oral tyrosine. Free and total tyrosine in breast milk and free tyrosine in plasma were measured. Free tyrosine was also measured in 12 different infant formulas. Total tyrosine in breast milk did not rise, but there was a slight tendency towards a reduction (up to −12 %; repeated measures ANOVA (RMANOVA): p = 0.074). Maternal plasma tyrosine rose (RMANOVA: p < 0.005). In breast milk, 98 % of tyrosine was in proteins or peptides and 2 % was free. Free tyrosine levels in breast milk rose in each group (RMANOVA: p < 0.005), but levels were within the range found in common infant formulas. The negligible effect of oral tyrosine on its concentration in breast milk supports further development of oral tyrosine as part of a prevention strategy for PPD.




Similar content being viewed by others
References
(1997) World Medical Association declaration of Helsinki. Recommendations guiding physicians in biomedical research involving human subjects. JAMA 277(11):925–926
(2007) Antenatal and postnatal mental health: clinical management and service guidance. NICE clinical guidelines 45. National Institute for Health and Clinical Excellence, London
Agostoni C, Carratu B, Boniglia C, Lammardo AM, Riva E, Sanzini E (2000a) Free glutamine and glutamic acid increase in human milk through a three-month lactation period. J Pediatr Gastroenterol Nutr 31(5):508–512
Agostoni C, Carratu B, Boniglia C, Riva E, Sanzini E (2000b) Free amino acid content in standard infant formulas: comparison with human milk. J Am Coll Nutr 19:434–438
Alegria A, Barbera R, Farre R, Lagarda MJ, Lopez JC (1999) Amino acid contents of infant formulas. J Food Compos Anal 12:137–146
Amminger GP, Schafer MR, Papageorgiou K, Klier CM, Cotton SM, Harrigan SM, Mackinnon A, McGorry PD, Berger GE (2010) Long-chain omega-3 fatty acids for indicated prevention of psychotic disorders: a randomized, placebo-controlled trial. Arch Gen Psychiatry 67:146–154
Bacher I, Houle S, Xu X, Zawertailo L, Soliman A, Wilson AA, Selby P, George TP, Sacher J, Miler L, Kish SJ, Rusjan P, Meyer JH (2011) Monoamine oxidase A binding in the prefrontal and anterior cingulate cortices during acute withdrawal from heavy cigarette smoking. Arch Gen Psychiatry 68:817–826
Banderet LE, Lieberman HR (1989) Treatment with tyrosine, a neurotransmitter precursor, reduces environmental stress in humans. Brain Res Bull 22:759–762
Bener A, Galadari S, Gillet M, Osman N, Al-Taneiji H, Al-Kuwaiti MHH, Al-Sabosy MMA (2001) Fasting during the holy month of Ramadan does not change the composition of breast milk. Nutr Res 21:859–864
Carratú B, Boniglia C, Scalise F, Ambruzzi AM, Sanzini E (2003) Nitrogenous components of human milk: non-protein nitrogen, true protein and free amino acids. Food Chem 81:357–362
Comtois KA, Schiff MA, Grossman DC (2008) Psychiatric risk factors associated with postpartum suicide attempt in Washington State, 1992–2001. Am J Obstet Gynecol 199(120):e1–e5
Deijen JB, Orlebeke JF (1994) Effect of tyrosine on cognitive function and blood pressure under stress. Brain Res Bull 33:319–323
Dennis CL, Dowswell T (2013) Psychosocial and psychological interventions for preventing postpartum depression. Cochrane Database Syst Rev 2, CD001134
Emmett PM, Rogers IS (1997) Properties of human milk and their relationship with maternal nutrition. Early Hum Dev 49(Suppl):S7–S28
Glaeser BS, Melamed E, Growdon JH, Wurtman RJ (1979) Elevation of plasma tyrosine after a single oral dose of L-tyrosine. Life Sci 25:265–271
Goodman JH (2004) Postpartum depression beyond the early postpartum period. J Obstet Gynecol Neonatal Nurs 33:410–420
Growdon JH, Melamed E, Logue M, Hefti F, Wurtman RJ (1982) Effects of oral L-tyrosine administration on CSF tyrosine and homovanillic acid levels in patients with Parkinson’s disease. Life Sci 30:827–832
Horowitz JA, Goodman J (2004) A longitudinal study of maternal postpartum depression symptoms. Res Theory Nurs Pract 18:149–163
Johnson S, Stockmeier CA, Meyer JH, Austin MC, Albert PR, Wang J, May WL, Rajkowska G, Overholser JC, Jurjus G, Dieter L, Johnson C, Sittman DB, Ou XM (2011) The reduction of R1, a novel repressor protein for monoamine oxidase A, in major depressive disorder. Neuropsychopharmacology 36:2139–2148
Kumar R, Robson KM (1984) A prospective study of emotional disorders in childbearing women. Br J Psychiatry 144:35–47
Lapiz-Bluhm MD, Soto-Pina AE, Hensler JG, Morilak DA (2009) Chronic intermittent cold stress and serotonin depletion induce deficits of reversal learning in an attentional set-shifting test in rats. Psychopharmacology (Berl) 202:329–341
Lemons JA, Reyman D, Moye L (1983) Amino acid composition of preterm and term breast milk during early lactation. Early Hum Dev 8:323–329
Lonnerdal B (1986) Effects of maternal dietary intake on human milk composition. J Nutr 116:499–513
Mahoney MM, Ramanathan C, Smale L (2007) Tyrosine hydroxylase positive neurons and their contacts with vasoactive intestinal polypeptide-containing fibers in the hypothalamus of the diurnal murid rodent, Arvicanthis niloticus. J Chem Neuroanat 33:131–139
Melamed E, Glaeser B, Growdon JH, Wurtman RJ (1980) Plasma tyrosine in normal humans: effects of oral tyrosine and protein-containing meals. J Neural Transm 47:299–306
Meyer JH, Ginovart N, Boovariwala A, Sagrati S, Hussey D, Garcia A, Young T, Praschak-Rieder N, Wilson AA, Houle S (2006) Elevated monoamine oxidase A levels in the brain: an explanation for the monoamine imbalance of major depression. Arch Gen Psychiatry 63:1209–1216
Meyer JH, Wilson AA, Sagrati S, Miler L, Rusjan P, Bloomfield PM, Clark M, Sacher J, Voineskos AN, Houle S (2009) Brain monoamine oxidase A binding in major depressive disorder: relationship to selective serotonin reuptake inhibitor treatment, recovery, and recurrence. Arch Gen Psychiatry 66:1304–1312
Miller LJ, LaRusso EM (2011) Preventing postpartum depression. Psychiatr Clin N Am 34:53–65
Naqvi F, Haider S, Batool Z, Perveen T, Haleem DJ (2012) Sub-chronic exposure to noise affects locomotor activity and produces anxiogenic and depressive like behavior in rats. Pharmacol Rep 64:64–69
Nelson DL, Herbet A, Glowinski J, Hamon M (1979) [3H]Harmaline as a specific ligand of MAO A–II. Measurement of the turnover rates of MAO A during ontogenesis in the rat brain. J Neurochem 32:1829–1836
Nott PN (1987) Extent, timing and persistence of emotional disorders following childbirth. Br J Psychiatry 151:523–527
O’Brien C, Mahoney C, Tharion WJ, Sils IV, Castellani JW (2007) Dietary tyrosine benefits cognitive and psychomotor performance during body cooling. Physiol Behav 90:301–307
O’Hara M (1994) Postpartum depression: causes and consequences. Springer-Verlag, New York
O’Hara MW, Swain AM (1996) Rates and risk of postpartum depression: a meta-analysis. Int Rev Psychiatry 8:37–54
Pardridge WM (1977) Kinetics of competitive inhibition of neutral amino acid transport across the blood–brain barrier. J Neurochem 28:103–108
Philipps LH, O’Hara MW (1991) Prospective study of postpartum depression: 4 1/2-year follow-up of women and children. J Abnorm Psychol 100:151–155
Rakicioglu N, Samur G, Topcu A, Topcu AA (2006) The effect of Ramadan on maternal nutrition and composition of breast milk. Pediatr Int 48:278–283
Sacher J, Wilson AA, Houle S, Rusjan P, Hassan S, Bloomfield PM, Stewart DE, Meyer JH (2010) Elevated brain monoamine oxidase A binding in the early postpartum period. Arch Gen Psychiatry 67:468–474
Sacher J, Rekkas PV, Wilson AA, Houle S, Romano L, Hamidi J, Rusjan P, Fan I, Stewart DE, Meyer JH (2014) Relationship of monoamine oxidase a distribution volume to postpartum depression and postpartum crying. Neuropsychopharmacology (in press)
Saura J, Kettler R, Da Prada M, Richards JG (1992) Quantitative enzyme radioautography with 3H-Ro 41–1049 and 3H-Ro 19–6327 in vitro: localization and abundance of MAO-A and MAO-B in rat CNS, peripheral organs, and human brain. J Neurosci 12:1977–1999
Shurtleff D, Thomas JR, Schrot J, Kowalski K, Harford R (1994) Tyrosine reverses a cold-induced working memory deficit in humans. Pharmacol Biochem Behav 47:935–941
Stewart DE (1988) Prophylactic lithium in postpartum affective psychosis. J Nerv Ment Dis 176:485–489
Stone EA (1975) Stress and catecholamines. In: Friedhof AJ (ed) Catecholamines and behavior. Plenum Press, New York, pp 31–72
Svanberg U, Gebre-Medhin M, Ljungqvist B, Olsson M (1977) Breast milk composition in Ethiopian and Swedish mothers. III. Amino acids and other nitrogenous substances. Am J Clin Nutr 30:499–507
Tumilty L, Davison G, Beckmann M, Thatcher R (2011) Oral tyrosine supplementation improves exercise capacity in the heat. Eur J Appl Physiol 111:2941–2950
Van Praag HM (1990) Catecholamine precursor research in depression: the practical and scientific yield. In: Richardson MA (ed) Amino acids in psychiatric disease. American Psychiatric Press, Washington, pp 77–97
Ventura AK, San Gabriel A, Hirota M, Mennella JA (2012) Free amino acid content in infant formula. Nutrition Food Sci 42:271–278
Warner R, Appleby L, Whitton A, Faragher B (1996) Demographic and obstetric risk factors for postnatal psychiatric morbidity. Br J Psychiatry 168:607–611
Wisner KL, Parry BL, Piontek CM (2002) Clinical practice. Postpartum depression. N Engl J Med 347:194–199
Wurtman JJ, Fernstrom JD (1979) Free amino acid, protein, and fat contents of breast milk from Guatemalan mothers consuming a corn-based diet. Early Hum Dev 3:67–77
Yamawaki N, Yamada M, Kan-no T, Kojima T, Kaneko T, Yonekubo A (2005) Macronutrient, mineral and trace element composition of breast milk from Japanese women. J Trace Elem Med Biol 19:171–181
Yeghiayan SK, Luo S, Shukitt-Hale B, Lieberman HR (2001) Tyrosine improves behavioral and neurochemical deficits caused by cold exposure. Physiol Behav 72:311–316
Young SN (1996) Behavioral effects of dietary neurotransmitter precursors: basic and clinical aspects. Neurosci Biobehav Rev 20:313–323
Funding
This research received project support from the Canadian Institutes of Health Research, Dr. Meyer’s Canada Research Chair, and Yekta Dowlati received salary support from the Ontario Mental Health Foundation (OMHF).
Conflict of interest
Yekta Dowlati is assessing the effect of dietary supplements on mood in early postpartum. Jeffrey H. Meyer has received operating grant funds for other studies from Eli Lilly, Lundbeck, GlaxoSmithKline, and SK Life Science in the past 5 years. Dr. Meyer has been a consultant to several of these companies as well as Takeda, Sepracor, Trius, Mylan, and Teva. None of these companies participated in the design or execution of this study or writing of the manuscript. Dr. Meyer is developing natural health products to treat high MAO-A states and is applying for patents for such supplements. Dr. Meyer is applying for patents to implement measures of utilizing MAO to diagnose or treat mood disorders. It is likely that companies which make medications affecting monoamine receptor or monoamine oxidase binding will seek collaborations with Dr. Meyer in the future. Donna E. Stewart is a member of the Scientific Advisory Committee of the Duloxetine Pregnancy Registry and received travel funds for Ranbaxy Pharma in the past 12 months. She was also an author on an up-to-date chapter for depression in pregnancy. Arun V. Ravindran has received grant support from, has served on advisory boards for, and has participated in sponsored lectures in the past 3 years for AstraZeneca, Eli Lilly & Co., Pfizer Canada, Bristol-Myers Squibb, Janssen Ortho, and Cephalon. Meir Steiner does not have any conflict of interest. Maxim Maheux does not have any conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dowlati, Y., Ravindran, A.V., Maheux, M. et al. No effect of oral tyrosine on total tyrosine levels in breast milk: implications for dietary supplementation in early postpartum. Arch Womens Ment Health 17, 541–548 (2014). https://doi.org/10.1007/s00737-014-0441-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00737-014-0441-8