Abstract
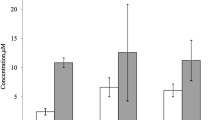
Alzheimer's disease is characterized by the presence of senile plaques composed of β-amyloid peptide (Aβ) aggregates with toxic effects that are still not fully understood. Recently, it was discovered that Aβ(1–42) fibrils possess catalytic activity on acetylcholine hydrolysis. Catalytic amyloids are an emerging and exciting field of research. In this study, we examined the catalytic activity of the fibrils formed by Aβ(1–40), the most abundant Aβ variant, on acetylcholine hydrolysis. Our findings reveal that Aβ(1–40) fibrils exhibit moderate enzymatic activity, indicating that natural peptide aggregates could serve as biocatalysts and provide new insights into the potential role of Aβ in neurological disorders.


Similar content being viewed by others
Availability of data and materials
Not applicable.
Code availability (software application or custom code)
Not applicable.
References
Arad E, Jelinek R (2022) Catalytic Amyloids. Trends Chem 4(10):907–917. https://doi.org/10.1016/j.trechm.2022.07.001
Arad E, Leshem AB, Rapaport H, Jelinek R (2021) β-Amyloid fibrils catalyze neurotransmitter degradation. Chem Catalysis 1(4):908–922. https://doi.org/10.1016/j.checat.2021.07.005
Association A (2023) 2023 Alzheimer’s disease facts and figures. Alzheimer’s Dementia 19(4):1598–1695. https://doi.org/10.1002/alz.13016
Bachurin SO, Makhaeva GF, Shevtsova EF, Boltneva NP, Kovaleva NV, Lushchekina SV, Rudakova EV, Dubova LG, Vinogradova DV, Sokolov VB, Aksinenko AY, Fisenko VP, Richardson RJ, Aliev G (2019) Conjugates of methylene blue with γ-carboline derivatives as new multifunctional agents for the treatment of neurodegenerative diseases. Sci Rep 9(1):4873. https://doi.org/10.1038/s41598-019-41272-4
Bajda M, Więckowska A, Hebda M, Guzior N, Sotriffer CA, Malawska B (2013) Structure-based search for new inhibitors of cholinesterases. Int J Mol Sci 14(3):5608–5632. https://doi.org/10.3390/ijms14035608
Bertini I, Gonnelli L, Luchinat C, Mao J, Nesi A (2011) A new structural model of Aβ40 fibrils. J Am Chem Soc 133(40):16013–16022. https://doi.org/10.1021/ja2035859
Biancalana M, Koide S (2010) Molecular mechanism of Thioflavin-T binding to amyloid fibrils. Biochem Biophys Acta 1804(7):1405–1412. https://doi.org/10.1016/j.bbapap.2010.04.001
Brinkmalm G, Hong W, Wang Z, Liu W, O’Malley TT, Sun X, Frosch MP, Selkoe DJ, Portelius E, Zetterberg H, Blennow K, Walsh DM (2019) Identification of neurotoxic cross-linked amyloid-β dimers in the Alzheimer’s brain. Brain 142(5):1441–1457. https://doi.org/10.1093/brain/awz066
Cerofolini L, Ravera E, Bologna S, Wiglenda T, Böddrich A, Purfürst B, Benilova I, Korsak M, Gallo G, Rizzo D, Gonnelli L, Fragai M, De Strooper B, Wanker EE, Luchinat C (2020) Mixing Aβ(1–40) and Aβ(1–42) peptides generates unique amyloid fibrils. Chem Commun (camb) 56(62):8830–8833. https://doi.org/10.1039/d0cc02463e
Chen X, Wehle S, Kuzmanovic N, Merget B, Holzgrabe U, König B, Sotriffer CA, Decker M (2014) Acetylcholinesterase inhibitors with photoswitchable inhibition of β-amyloid aggregation. ACS Chem Neurosci 5(5):377–389. https://doi.org/10.1021/cn500016p
Díaz-Caballero M, Navarro S, Nuez-Martínez M, Peccati F, Rodríguez-Santiago L, Sodupe M, Teixidor F, Ventura S (2021) pH-responsive self-assembly of amyloid fibrils for dual hydrolase-oxidase reactions. ACS Catal 11(2):595–607. https://doi.org/10.1021/acscatal.0c03093
Eisenthal R, Danson MJ, Hough DW (2007) Catalytic efficiency and kcat/KM: a useful comparator? Trends Biotechnol 25(6):247–249. https://doi.org/10.1016/j.tibtech.2007.03.010
Ellman GL, Courtney KD, Andres V Jr, Feather-Stone RM (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7(2):88–95. https://doi.org/10.1016/0006-2952(61)90145-9
Gu L, Guo Z (2021) Alzheimer’s Aβ42 and Aβ40 form mixed oligomers with direct molecular interactions. Biochem Biophys Res Commun 534:292–296. https://doi.org/10.1016/j.bbrc.2020.11.092
Heerde T, Bansal A, Schmidt M, Fändrich M (2023) Cryo-EM structure of a catalytic amyloid fibril. Sci Rep 13(1):4070. https://doi.org/10.1038/s41598-023-30711-y
Hudson SA, Ecroyd H, Kee TW, Carver JA (2009) The thioflavin T fluorescence assay for amyloid fibril detection can be biased by the presence of exogenous compounds. FEBS J 276(20):5960–5972. https://doi.org/10.1111/j.1742-4658.2009.07307.x
Lewczuk P, Wiltfang J, Kornhuber J, Verhasselt A (2021) Distributions of Aβ42 and Aβ42/40 in the cerebrospinal fluid in view of the probability theory. Diagnostics 11(12):2372. https://doi.org/10.3390/diagnostics11122372
Lin Y, Im H, Diem LT, Ham S (2019) Characterizing the structural and thermodynamic properties of Aβ42 and Aβ40. Biochem Biophys Res Commun 510(3):442–448. https://doi.org/10.1016/j.bbrc.2019.01.124
Liu PP, Xie Y, Meng XY, Kang JS (2019) History and progress of hypotheses and clinical trials for Alzheimer’s disease. Signal Transduct Target Ther 4:29. https://doi.org/10.1038/s41392-019-0063-8
Makam P, Yamijala SSRKC, Tao K, Shimon LJW, Eisenberg DS, Sawaya MR, Wong BM, Gazit E (2019) Non-proteinaceous hydrolase comprised of a phenylalanine metallo-supramolecular amyloid-like structure. Nat Catal 2(11):977–985. https://doi.org/10.1038/s41929-019-0348-x
Nicoll A, Allemann R (2004) Nucleophilic and general acid catalysis at physiological pH by a designed miniature esterase. Org Biomol Chem 2(15):2175–2180. https://doi.org/10.1039/B404730C
Qiu T, Liu Q, Chen YX, Zhao YF, Li YM (2015) Aβ42 and Aβ40: similarities and differences. J Pept Sci 21(7):522–529. https://doi.org/10.1002/psc.2789
Rahman MM, Lendel C (2021) Extracellular protein components of amyloid plaques and their roles in Alzheimer’s disease pathology. Mol Neurodegener 16(1):59. https://doi.org/10.1186/s13024-021-00465-0
Rufo CM, Moroz YS, Moroz OV, Stöhr J, Smith TA, Hu X, DeGrado WF, Korendovych IV (2014) Short peptides self-assemble to produce catalytic amyloids. Nat Chem 6(4):303–309. https://doi.org/10.1038/nchem.1894
Sanchis I, Spinelli R, Dias J, Brazzolotto X, Rietmann A, Aimaretti F, Siano AS (2023) inhibition of human cholinesterases and invitro β-amyloid aggregation by rationally designed peptides. ChemMedChem. https://doi.org/10.1002/cmdc.202200691
Sarkhel B, Chatterjee A, Das D (2020) Covalent catalysis by cross β amyloid nanotubes. J Am Chem Soc 142(9):4098–4103. https://doi.org/10.1021/jacs.9b13517
Smith DH, Stewart W (2018) Traumatic brain injury: a platform for studies in Aβ processing: Commentary on: “Rapid Aβ oligomer and protofibril accumulation in traumatic brain injury.” Brain Pathol 28(4):463–465. https://doi.org/10.1111/bpa.12534
Wang L, Eom K, Kwon T (2021) Different aggregation pathways and structures for Aβ40 and Aβ42 peptides. Biomolecules 11(2):198. https://doi.org/10.3390/biom11020198
Wildburger NC, Esparza TJ, LeDuc RD, Fellers RT, Thomas PM, Cairns NJ, Kelleher NL, Bateman RJ, Brody DL (2017) Diversity of amyloid-beta proteoforms in the Alzheimer’s disease brain. Sci Rep 7(1):9520. https://doi.org/10.1038/s41598-017-10422-x
Zhang C, Xue X, Luo Q, Li Y, Yang K, Zhuang X, Jiang Y, Zhang J, Liu J, Zou G, Liang XJ (2014) Self-assembled Peptide nanofibers designed as biological enzymes for catalyzing ester hydrolysis. ACS Nano 8(11):11715–11723. https://doi.org/10.1021/nn5051344
Zozulia O, Dolan MA, Korendovych IV (2018) Catalytic peptide assemblies. Chem Soc Rev 47:3621–3639. https://doi.org/10.1039/C8CS00080H
Funding
This work was financially supported by the Argentinian National Scientific and Technical Research Council (CONICET) of the Ministry of Science, Technology and Innovation under grants PICT-2017-0035, PIP 2017-2019 GI. 112 201701 00462 CO.
Author information
Authors and Affiliations
Contributions
IS and AS: Conceptualization; IS and RS: Methodology; investigation and result analysis: IS (Synthesis and assays), RS (ThT assay); IS: Writing—original draft preparation; IS, RS and AS: Writing—review and editing; AS: Funding acquisition; AS: Supervision.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare no competing interests.
Additional information
Handling editor: S. Beninati.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sanchis, I., Spinelli, R. & Siano, A. Acetylcholine hydrolytic activity of fibrillated β-amyloid (1–40) peptide. Amino Acids 55, 1991–1997 (2023). https://doi.org/10.1007/s00726-023-03349-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-023-03349-3