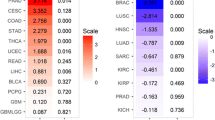
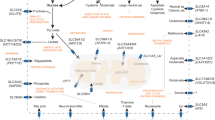
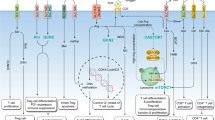
Abstract
Amino acids are essential for the survival of all living organisms and living cells. Amino acid transporters mediate the transport and absorption of amino acids, and the dysfunction of these proteins can induce human diseases. Cationic amino acid transporters (CAT family, SLC7A1-4, and SLC7A14) are considered to be a group of transmembrane transporters, of which SLC7A1-3 are essential for arginine transport in mammals. Numerous studies have shown that CAT family-mediated arginine transport is involved in signal crosstalk between malignant tumor cells and immune cells, especially T cells. The modulation of extracellular arginine concentration has entered a number of clinical trials and achieved certain therapeutic effects. Here, we review the role of CAT family on tumor cells and immune infiltrating cells in malignant tumors and explore the therapeutic strategies to interfere with extracellular arginine concentration, to elaborate its application prospects. CAT family members may be used as biomarkers for certain cancer entities and might be included in new ideas for immunotherapy of malignant tumors.

Similar content being viewed by others
Data availability
Data sharing does not apply to this paper, because no corresponding experimental data were generated during the current study. Some of the data supporting the article can be found in the hyperlink to the manuscript.
References
Abdelmagid SA, Rickard JA, Mcdonald WJ et al (2011) CAT-1-mediated arginine uptake and regulation of nitric oxide synthases for the survival of human breast cancer cell lines. J Cell Biochem 112(4):1084–1092
Abou-Alfa GK, Qin S, Ryoo BY et al (2018) Phase III randomized study of second line ADI-PEG 20 plus best supportive care versus placebo plus best supportive care in patients with advanced hepatocellular carcinoma. Ann Oncol 29(6):1402–1408
Agrawal V, Woo JH, Mauldin JP et al (2012) Cytotoxicity of human recombinant arginase I (Co)-PEG5000 in the presence of supplemental L-citrulline is dependent on decreased argininosuccinate synthetase expression in human cells. Anticancer Drugs 23(1):51–64
Albritton LM, Tseng L, Scadden D et al (1989) A putative murine ecotropic retrovirus receptor gene encodes a multiple membrane-spanning protein and confers susceptibility to virus infection. Cell 57(4):659–666
Al-Koussa H, Al-Haddad M, Abi-Habib R et al (2019) Human recombinant arginase I [HuArgI (Co)-PEG5000]-induced arginine depletion inhibits colorectal cancer cell migration and invasion. Int J Mol Sci 20(23)
Bai L, Sato H, Kubo Y et al (2019) CAT1/SLC7A1 acts as a cellular receptor for bovine leukemia virus infection. FASEB J 33(12):14516–14527
Bian Y, Li W, Kremer DM et al (2020) Cancer SLC43A2 alters T cell methionine metabolism and histone methylation. Nature 585(7824):277–282
Bowles TL, Kim R, Galante J et al (2008) Pancreatic cancer cell lines deficient in argininosuccinate synthetase are sensitive to arginine deprivation by arginine deiminase. Int J Cancer 123(8):1950–1955
Brin E, Wu K, Lu HT et al (2017) PEGylated arginine deiminase can modulate tumor immune microenvironment by affecting immune checkpoint expression, decreasing regulatory T cell accumulation and inducing tumor T cell infiltration. Oncotarget 8(35):58948–58963
Chang KY, Chiang NJ, Wu SY et al (2021) Phase 1b study of pegylated arginine deiminase (ADI-PEG 20) plus Pembrolizumab in advanced solid cancers. Oncoimmunology 10(1):1943253
Chedid V, Vijayvargiya P, Camilleri M (2018) Elobixibat for the treatment of constipation. Expert Rev Gastroenterol Hepatol 12(10):951–960
Chen CL, Hsu SC, Ann DK et al (2021) Arginine signaling and cancer metabolism. Cancers 13(14)
Cheng PN, Lam TL, Lam WM et al (2007) Pegylated recombinant human arginase (rhArg-peg5,000mw) inhibits the in vitro and in vivo proliferation of human hepatocellular carcinoma through arginine depletion. Cancer Res 67(1):309–317
Cheon DJ, Walts AE, Beach JA et al (2015) Differential expression of argininosuccinate synthetase in serous and non-serous ovarian carcinomas. J Pathol Clin Res 1(1):41–53
Cimen Bozkus C, Elzey BD, Crist SA et al (2015) Expression of cationic amino acid transporter 2 is required for myeloid-derived suppressor cell-mediated control of T cell immunity. J Immunol 195(11):5237–5250
Closs EI, Boissel JP, Habermeier A et al (2006) Structure and function of cationic amino acid transporters (CATs). J Membr Biol 213(2):67–77
Closs EI, Simon A, Vékony N et al (2004) Plasma membrane transporters for arginine. J Nutr 134(10 Suppl):2752S-2759S (discussion 65S–67S)
Coburn LA, Singh K, Asim M et al (2019) Loss of solute carrier family 7 member 2 exacerbates inflammation-associated colon tumorigenesis. Oncogene 38(7):1067–1079
Delage B, Luong P, Maharaj L et al (2012) Promoter methylation of argininosuccinate synthetase-1 sensitises lymphomas to arginine deiminase treatment, autophagy and caspase-dependent apoptosis. Cell Death Dis 3(7):e342
Du F, Zheng F, Han Y et al (2022) Novel immune-related gene signature for risk stratification and prognosis of survival in ER (+) and/or PR (+) and HER2 (-) breast cancer. Front Pharmacol 13:820437
Eckert MA, Coscia F, Chryplewicz A et al (2019) Proteomics reveals NNMT as a master metabolic regulator of cancer-associated fibroblasts. Nature 569(7758):723–728
El-Mais N, Fakhoury I, Al Haddad M et al (2021) Human recombinant arginase I [HuArgI(Co)-PEG5000]-induced arginine depletion inhibits pancreatic cancer cell migration and invasion through autophagy. Pancreas 50(8):1187–1194
Ensor CM, Holtsberg FW, Bomalaski JS et al (2002) Pegylated arginine deiminase (ADI-SS PEG20,000 mw) inhibits human melanomas and hepatocellular carcinomas in vitro and in vivo. Cancer Res 62(19):5443–5450
Fan A, Zhang Y, Cheng J et al (2022) A novel prognostic model for prostate cancer based on androgen biosynthetic and catabolic pathways. Front Oncol 12:950094
Feun LG, Marini A, Walker G et al (2012) Negative argininosuccinate synthetase expression in melanoma tumours may predict clinical benefit from arginine-depleting therapy with pegylated arginine deiminase. Br J Cancer 106(9):1481–1485
Fotiadis D, Kanai Y, Palacín M (2013) The SLC3 and SLC7 families of amino acid transporters. Mol Aspects Med 34(2–3):139–158
Fultang L, Vardon A, De Santo C et al (2016) Molecular basis and current strategies of therapeutic arginine depletion for cancer. Int J Cancer 139(3):501–509
Garibsingh RA, Schlessinger A (2019) Advances and challenges in rational drug design for SLCs. Trends Pharmacol Sci 40(10):790–800
Geiger R, Rieckmann JC, Wolf T et al (2016) L-Arginine modulates T cell metabolism and enhances survival and anti-tumor activity. Cell 167(3):829–42.e13
Giffen KP, Li Y, Liu H et al (2022) Mutation of SLC7A14 causes auditory neuropathy and retinitis pigmentosa mediated by lysosomal dysfunction. Sci Adv 8(14):eabk0942
Gong W, Chen Y, Zhang Y (2021) Prognostic and clinical significance of Solute Carrier Family 7 Member 1 in ovarian cancer. Transl Cancer Res 10(2):602–612
He X, Lin H, Yuan L et al (2017) Combination therapy with L-arginine and α-PD-L1 antibody boosts immune response against osteosarcoma in immunocompetent mice. Cancer Biol Ther 18(2):94–100
He W, Zhang J, Liu B et al (2020) S119N mutation of the E3 ubiquitin ligase SPOP suppresses SLC7A1 degradation to regulate hepatoblastoma progression. Mol Ther Oncolytics 19:149–162
Heys SD, Segar A, Payne S et al (1997) Dietary supplementation with L-arginine: modulation of tumour-infiltrating lymphocytes in patients with colorectal cancer. Br J Surg 84(2):238–241
Heys SD, Ogston K, Miller I et al (1998) Potentiation of the response to chemotherapy in patients with breast cancer by dietary supplementation with L-arginine: results of a randomised controlled trial. Int J Oncol 12(1):221–225
Hosokawa H, Sawamura T, Kobayashi S et al (1997) Cloning and characterization of a brain-specific cationic amino acid transporter. J Biol Chem 272(13):8717–8722
Huang Y (2007) Pharmacogenetics/genomics of membrane transporters in cancer chemotherapy. Cancer Metastasis Rev 26(1):183–201
Huang Y, Anderle P, Bussey KJ et al (2004) Membrane transporters and channels: role of the transportome in cancer chemosensitivity and chemoresistance. Cancer Res 64(12):4294–4301
Huang H, Zhou P, Wei J et al (2021) In vivo CRISPR screening reveals nutrient signaling processes underpinning CD8(+) T cell fate decisions. Cell 184(5):1245–61.e21
Ito K, Groudine M (1997) A new member of the cationic amino acid transporter family is preferentially expressed in adult mouse brain. J Biol Chem 272(42):26780–26786
Januchowski R, Zawierucha P, Andrzejewska M et al (2013) Microarray-based detection and expression analysis of ABC and SLC transporters in drug-resistant ovarian cancer cell lines. Biomed Pharmacother 67(3):240–245
Ji JX, Cochrane DR, Tessier-Cloutier B et al (2020) Arginine depletion therapy with ADI-PEG20 limits tumor growth in argininosuccinate synthase-deficient ovarian cancer, including small-cell carcinoma of the ovary, hypercalcemic type. Clin Cancer Res 26(16):4402–4413
Jiang YQ, Wang ZX, Zhong M et al (2021) Investigating mechanisms of response or resistance to immune checkpoint inhibitors by analyzing cell-cell communications in tumors before and after programmed cell death-1 (PD-1) targeted therapy: an integrative analysis using single-cell RNA and bulk-RNA sequencing data. Oncoimmunology 10(1):1908010
Jiang S, Zou J, Dong J et al (2023) Lower SLC7A2 expression is associated with enhanced multidrug resistance, less immune infiltrates and worse prognosis of NSCLC. Cell Commun Signal 21(1):9
Jin ZB, Huang XF, Lv JN et al (2014) SLC7A14 linked to autosomal recessive retinitis pigmentosa. Nat Commun 5:3517
Kelly MP, Jungbluth AA, Wu BW et al (2012) Arginine deiminase PEG20 inhibits growth of small cell lung cancers lacking expression of argininosuccinate synthetase. Br J Cancer 106(2):324–332
Khoury O, Ghazale N, Stone E et al (2015) Human recombinant arginase I (Co)-PEG5000 [HuArgI (Co)-PEG5000]-induced arginine depletion is selectively cytotoxic to human glioblastoma cells. J Neurooncol 122(1):75–85
Kishikawa T, Otsuka M, Tan PS et al (2015) Decreased miR122 in hepatocellular carcinoma leads to chemoresistance with increased arginine. Oncotarget 6(10):8339–8352
Kwong-Lam F, Chi-Fung CG (2013) Vincristine could partly suppress stromal support to T-ALL blasts during pegylated arginase I treatment. Exp Hematol Oncol 2(1):11
Lam TL, Wong GK, Chong HC et al (2009) Recombinant human arginase inhibits proliferation of human hepatocellular carcinoma by inducing cell cycle arrest. Cancer Lett 277(1):91–100
Lemos H, Huang L, Prendergast GC et al (2019) Immune control by amino acid catabolism during tumorigenesis and therapy. Nat Rev Cancer 19(3):162–175
Lorentzen CL, Martinenaite E, Kjeldsen JW et al (2022) Arginase-1 targeting peptide vaccine in patients with metastatic solid tumors - a phase I trial. Front Immunol 13:1023023
Lowman XH, Hanse EA, Yang Y et al (2019) p53 promotes cancer cell adaptation to glutamine deprivation by upregulating Slc7a3 to increase arginine uptake. Cell Rep 26(11):3051–60.e4
Lukey MJ, Katt WP, Cerione RA (2017) Targeting amino acid metabolism for cancer therapy. Drug Discov Today 22(5):796–804
Mccord N, Ayuk P, Mcmahon M et al (2006) System y+ arginine transport and NO production in peripheral blood mononuclear cells in pregnancy and preeclampsia. Hypertension 47(1):109–115
Menjivar RE, Nwosu ZC, Du W et al (2023) Arginase 1 is a key driver of immune suppression in pancreatic cancer. eLife 12
Miraki-Moud F, Ghazaly E, Ariza-Mcnaughton L et al (2015) Arginine deprivation using pegylated arginine deiminase has activity against primary acute myeloid leukemia cells in vivo. Blood 125(26):4060–4068
Missiaen R, Anderson NM, Kim LC et al (2022) GCN2 inhibition sensitizes arginine-deprived hepatocellular carcinoma cells to senolytic treatment. Cell Metab 34(8):1151–67.e7
Mussai F, Egan S, Higginbotham-Jones J et al (2015) Arginine dependence of acute myeloid leukemia blast proliferation: a novel therapeutic target. Blood 125(15):2386–2396
Nakanishi T (2007) Drug transporters as targets for cancer chemotherapy. Cancer Genomics Proteomics 4(3):241–254
Nakaya M, Xiao Y, Zhou X et al (2014) Inflammatory T cell responses rely on amino acid transporter ASCT2 facilitation of glutamine uptake and mTORC1 kinase activation. Immunity 40(5):692–705
Nasreddine G, El-Sibai M, Abi-Habib RJ (2020) Cytotoxicity of [HuArgI (co)-PEG5000]-induced arginine deprivation to ovarian Cancer cells is autophagy dependent. Invest New Drugs 38(1):10–19
Okita K, Hara Y, Okura H et al (2021) Antitumor effects of novel mAbs against cationic amino acid transporter 1 (CAT1) on human CRC with amplified CAT1 gene. Cancer Sci 112(2):563–574
Patil MD, Bhaumik J, Babykutty S et al (2016) Arginine dependence of tumor cells: targeting a chink in cancer’s armor. Oncogene 35(38):4957–4972
Payne SH, Loomis WF (2006) Retention and loss of amino acid biosynthetic pathways based on analysis of whole-genome sequences. Eukaryot Cell 5(2):272–276
Peng JB, Zhuang L, Berger UV et al (2001) CaT1 expression correlates with tumor grade in prostate cancer. Biochem Biophys Res Commun 282(3):729–734
Rodriguez-Ruiz ME, Buqué A, Hensler M et al (2019) Apoptotic caspases inhibit abscopal responses to radiation and identify a new prognostic biomarker for breast cancer patients. Oncoimmunology 8(11):e1655964
Rothenberg ME, Doepker MP, Lewkowich IP et al (2006) Cationic amino acid transporter 2 regulates inflammatory homeostasis in the lung. Proc Natl Acad Sci 103(40):14895–14900
Roy DG, Chen J, Mamane V et al (2020) Methionine metabolism shapes T helper cell responses through regulation of epigenetic reprogramming. Cell Metab 31(2):250–66.e9
Shen L, Qian C, Cao H et al (2018) Upregulation of the solute carrier family 7 genes is indicative of poor prognosis in papillary thyroid carcinoma. World J Surg Oncol 16(1):235
Siska PJ, Rathmell JC (2015) T cell metabolic fitness in antitumor immunity. Trends Immunol 36(4):257–264
Steggerda SM, Bennett MK, Chen J et al (2017) Inhibition of arginase by CB-1158 blocks myeloid cell-mediated immune suppression in the tumor microenvironment. J Immunother Cancer 5(1):101
Sun T, Bi F, Liu Z et al (2020) SLC7A2 serves as a potential biomarker and therapeutic target for ovarian cancer. Aging 12(13):13281–13296
Szefel J, Danielak A, Kruszewski WJ (2019) Metabolic pathways of L-arginine and therapeutic consequences in tumors. Adv Med Sci 64(1):104–110
Szlosarek PW, Grimshaw MJ, Wilbanks GD et al (2007) Aberrant regulation of argininosuccinate synthetase by TNF-alpha in human epithelial ovarian cancer. Int J Cancer 121(1):6–11
Tang K, Zhang H, Deng J et al (2023) Ammonia detoxification promotes CD8(+) T cell memory development by urea and citrulline cycles. Nat Immunol 24(1):162–173
Tanios R, Bekdash A, Kassab E et al (2013) Human recombinant arginase I(Co)-PEG5000 [HuArgI(Co)-PEG5000]-induced arginine depletion is selectively cytotoxic to human acute myeloid leukemia cells. Leuk Res 37(11):1565–1571
Tian X, Shi C, Liu S et al (2023) Methylation related genes are associated with prognosis of patients with head and neck squamous cell carcinoma via altering tumor immune microenvironment. J Dent Sci 18(1):57–64
Vasquez-Dunddel D, Pan F, Zeng Q et al (2013) STAT3 regulates arginase-I in myeloid-derived suppressor cells from cancer patients. J Clin Investig 123(4):1580–1589
Vékony N, Wolf S, Boissel JP et al (2001) Human cationic amino acid transporter hCAT-3 is preferentially expressed in peripheral tissues. Biochemistry 40(41):12387–12394
Verrey F, Closs EI, Wagner CA et al (2004) CATs and HATs: the SLC7 family of amino acid transporters. Pflugers Arch 447(5):532–542
Werner A, Amann E, Schnitzius V et al (2016) Induced arginine transport via cationic amino acid transporter-1 is necessary for human T-cell proliferation. Eur J Immunol 46(1):92–103
Werner A, Pieh D, Echchannaoui H et al (2019) Cationic amino acid transporter-1-mediated arginine uptake is essential for chronic lymphocytic leukemia cell proliferation and viability. Front Oncol 9:1268
Wolf S, Janzen A, Vékony N et al (2002) Expression of solute carrier 7A4 (SLC7A4) in the plasma membrane is not sufficient to mediate amino acid transport activity. Biochem J 364(Pt 3):767–775
Woodard MH, Dunn WA, Laine RO et al (1994) Plasma membrane clustering of system y+ (CAT-1) amino acid transporter as detected by immunohistochemistry. Am J Physiol 266(5 Pt 1):E817–E824
Xia S, Wu J, Zhou W et al (2021) SLC7A2 deficiency promotes hepatocellular carcinoma progression by enhancing recruitment of myeloid-derived suppressors cells. Cell Death Dis 12(6):570
Yan L, He J, Liao X et al (2022a) A comprehensive analysis of the diagnostic and prognostic value associated with the SLC7A family members in breast cancer. Gland Surg 11(2):389–411
Yan Y, Chen C, Li Z et al (2022b) Extracellular arginine is required but the arginine transporter CAT3 (Slc7a3) is dispensable for mouse normal and malignant hematopoiesis. Sci Rep 12(1):21832
Yang R, Wang Z, Li J et al (2021) The identification of the metabolism subtypes of skin cutaneous melanoma associated with the tumor microenvironment and the immunotherapy. Front Cell Dev Biol 9:707677
Yau T, Cheng PNM, Chiu J et al (2022) A phase 1 study of pegylated recombinant arginase (PEG-BCT-100) in combination with systemic chemotherapy (capecitabine and oxaliplatin)[PACOX] in advanced hepatocellular carcinoma patients. Invest New Drugs 40(2):314–321
Yeramian A, Martin L, Serrat N et al (2006) Arginine transport via cationic amino acid transporter 2 plays a critical regulatory role in classical or alternative activation of macrophages. J Immunol 176(10):5918–5924
You S, Zhu X, Yang Y et al (2022) SLC7A1 overexpression is involved in energy metabolism reprogramming to induce tumor progression in epithelial ovarian cancer and is associated with immune-infiltrating cells. J Oncol 2022:5864826
Acknowledgements
Thanks all the authors for their contributions to this study, which was partially supported by the 2020 Annual Science and Technology Project of Qingdao West Coast New Area (Grant No. 2020-53).
Author information
Authors and Affiliations
Contributions
All authors contributed to the writing of this review.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Additional information
Handling editor: E. Closs.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
You, S., Han, X., Xu, Y. et al. Research progress on the role of cationic amino acid transporter (CAT) family members in malignant tumors and immune microenvironment. Amino Acids 55, 1213–1222 (2023). https://doi.org/10.1007/s00726-023-03313-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-023-03313-1