Abstract
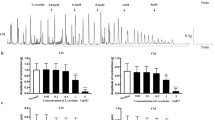
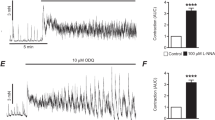
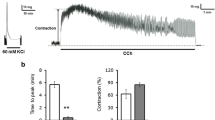
Arginine (Arg), as a basic amino acid, has been reported to be involved in regulation of gut motility. However, the evidence is limited and the underlying mechanism is not fully understood. Our study was conducted to investigate the effects of L-Arg on spontaneous contraction of the longitudinal muscle strip (LMS) of the rat colon and the relevant mechanisms. An organ bath system was used to detect the contractile force of the LMS. Whole-cell voltage-clamp techniques were applied to observe alterations in the currents of large conductance Ca2+-activated K+ (KCa) channels, voltage-dependent potassium (KV) channels, and L-type Ca2+ channels (LTCCs) in smooth muscle cells (SMCs) of the colon. We found that L-Arg within the physiological concentration had no effect on contraction of LMS, while 1 mM L-Arg significantly increased both the amplitude and frequency of LMS contractility. And the increase in force was mucosa-dependent, whereas changes in frequency as well as in amplitude were inhibited by atropine. In addition, L-Arg (1 mM) activated the LTTCs and inhibited both KCa channels and KV channels on SMCs. Thus, L-Arg above the physiological concentration exerted an excitatory effect on colonic LM contraction, and stimulation by L-Arg was mediated by ACh. In addition, LTCCs, KCa channels, and KV channels on SMCs were involved in the action of L-Arg.








Similar content being viewed by others
Data availability
The datasets used or analyzed during the current study are available from the corresponding author on reasonable request.
References
Adams DJ, Barakeh J, Laskey R, Van Breemen C (1989) Ion channels and regulation of intracellular calcium in vascular endothelial cells. FASEB J 3(12):2389–2400. https://doi.org/10.1096/fasebj.3.12.2477294
Azzena GB, Mancinelli R (1999) Nitric oxide regenerates the normal colonic peristaltic activity in mdx dystrophic mouse. Neurosci Lett 261(1–2):9–12. https://doi.org/10.1016/s0304-3940(98)00993-8
Bolton TB, Prestwich SA, Zholos AV, Gordienko DV (1999) Excitation-contraction coupling in gastrointestinal and other smooth muscles. Annu Rev Physiol 61:85–115. https://doi.org/10.1146/annurev.physiol.61.1.85
Calver A, Collier J, Vallance P (1993) Nitric oxide and cardiovascular control. Exp Physiol 78(3):303–326. https://doi.org/10.1113/expphysiol.1993.sp003687
Chuman H, Sugimoto T, Nao-I N (2017) Vasodilatory effect of L-arginine on isolated rabbit and human posterior ciliary arteries in vitro and increased optic disc blood flow in vivo. Graefes Arch Clin Exp Ophthalmol 255(12):2381–2388. https://doi.org/10.1007/s00417-017-3824-5
Fischer H, Becker JC, Boknik P et al (1999) Expression of constitutive nitric oxide synthase in rat and human gastrointestinal tract. Biochim Biophys Acta 1450(3):414–422. https://doi.org/10.1016/s0167-4889(99)00049-x
Fukui H, Xu X, Miwa H (2018) Role of Gut Microbiota-Gut Hormone Axis in the Pathophysiology of Functional Gastrointestinal Disorders. J Neurogastroenterol Motil 24(3):367–386. https://doi.org/10.5056/jnm18071
Groneberg D, Voussen B, Friebe A (2016) Integrative Control of Gastrointestinal Motility by Nitric Oxide. Curr Med Chem 23(24):2715–2735. https://doi.org/10.2174/0929867323666160812150907
Hall S, Turcato S, Clapp L (1996) Abnormal activation of K+ channels underlies relaxation to bacterial lipopolysaccharide in rat aorta. Biochem Biophys Res Commun 224(1):184–190. https://doi.org/10.1006/bbrc.1996.1005
Hayashi K, Shibata C, Nagao M et al (2010) Intracolonic capsaicin stimulates colonic motility and defecation in conscious dogs. Surgery 47(6):789–797. https://doi.org/10.1016/j.surg.2009.11.019
Hong SK, Maltz BE, Coburn LA et al (2010) Increased serum levels of L-arginine in ulcerative colitis and correlation with disease severity. Inflamm Bowel Dis 16(1):105–111. https://doi.org/10.1002/ibd.21035
Kakuda DK, Sweet MJ, Mac Leod CL, Hume DA, Markovich D (1999) CAT2-mediated L-arginine transport and nitric oxide production in activated macrophages. Biochem J 340:549–553
Lu P, Luo H, Quan X et al (2016) The role of substance P in the maintenance of colonic hypermotility induced by repeated stress in rats. Neuropeptides 56:75–82. https://doi.org/10.1016/j.npep.2016.01.006
Miyoshi H, Nakaya Y, Moritoki H (1994) Nonendothelial-derived nitric oxide activates the ATP-sensitive K+ channel of vascular smooth muscle cells. FEBS Lett 345(1):47–49. https://doi.org/10.1016/0014-5793(94)00417-x
Montgomery LE, Tansey EA, Johnson CD, Roe SM, Quinn JG (2016) Autonomic modification of intestinal smooth muscle contractility. Adv Physiol Educ 40(1):104–109. https://doi.org/10.1152/advan.00038.2015
Nakato J, Ho YY, Omae R et al (2017) l-Ornithine and l-lysine stimulate gastrointestinal motility via transient receptor potential vanilloid 1. Mol Nutr Food Res. https://doi.org/10.1002/mnfr.201700230
Parnell MM, Chin-Dusting JP, Starr J, Kaye DM (2004) In vivo and in vitro evidence for ACh-stimulated L-arginine uptake. Am J Physiol Heart Circ Physiol 287(1):H395–H400. https://doi.org/10.1152/ajpheart.01094.2003
Peng J, Li YJ (2010) The vanilloid receptor TRPV1: role in cardiovascular and gastrointestinal protection. Eur J Pharmacol 627(1–3):1–7. https://doi.org/10.1016/j.ejphar.2009.10.053
Scheurer U, Drack E, Halter F (1994) Substance P activates rat colonic motility via excitatory and inhibitory neural pathways and direct action on muscles. J Pharmacol Exp Ther 271(1):7–13
Shibata C, Sasaki I, Naito H, Ueno T, Matsuno S (1999) Intragastric capsaicin stimulates motility of upper gut and proximal colon via distinct pathways in conscious dogs. Dig Dis Sci 44(6):1083–1089. https://doi.org/10.1023/a:1026651518373
Shimamura K, Zhou M, Toba M et al (2003) Effects of L-arginine on spontaneous contraction of the rat portal vein. Pflugers Arch 446(1):30–35. https://doi.org/10.1007/s00424-003-1007-1
Singh K, Coburn LA, Barry DP, Boucher JL, Chaturvedi R, Wilson KT (2012) L-arginine uptake by cationic amino acid transporter 2 is essential for colonic epithelial cell restitution. Am J Physiol Gastrointest Liver Physiol 302(9):G1061–G1073. https://doi.org/10.1152/ajpgi.00544.2011
Smith PA, Sakura H, Coles B, Gummerson N, Proks P, Ashcroft FM (1997) Electrogenic arginine transport mediates stimulus-secretion coupling in mouse pancreatic beta-cells. J Physiol 499(3):625–635. https://doi.org/10.1113/jphysiol.1997.sp021955
Swiderski K, Bindon R, Trieu J et al (2020) Spatiotemporal Mapping Reveals Regional Gastrointestinal Dysfunction in mdx Dystrophic Mice Ameliorated by Oral L-arginine Supplementation. J Neurogastroenterol Motil 26(1):133–146. https://doi.org/10.5056/jnm19029
Taché Y, Martinez V, Million M, Rivier J (1999) Corticotropin-releasing factor and the brain-gut motor response to stress. Can J Gastroenterol 13:18–25. https://doi.org/10.1155/1999/375916
Teramoto N (2006) Physiological roles of ATP-sensitive K+ channels in smooth muscle. J Physiol 572(Pt 3):617–624. https://doi.org/10.1113/jphysiol.2006.105973
Vogalis F, Publicover NG, Hume JR, Sanders KM (1991) Relationship between calcium current and cytosolic calcium in canine gastric smooth muscle cells. Am J Physiol 260(5 Pt 1):C1012–C1018. https://doi.org/10.1152/ajpcell.1991.260.5.C1012
Ward SM, Sanders KM (1992) Upstroke component of electrical slow waves in canine colonic smooth muscle due to nifedipine-resistant calcium current. J Physiol 455:321–337. https://doi.org/10.1113/jphysiol.1992.sp019304
Wilson LB, Engbretson J (2000) Dorsal horn administration of L-arginine accentuates the pressor response evoked by activation of muscle mechanoreceptors. Auton Neurosci 86(1–2):135–139. https://doi.org/10.1016/S1566-0702(00)00207-1
Wu GY (2009) Amino acids: metabolism, functions, and nutrition. Amino Acids 37(1):1–17. https://doi.org/10.1007/s00726-009-0269-0
Yan L, Tang Q, Quan X et al (2019) Effects of exendin-4 on colonic motility in rats and its underlying mechanism. Neurogastroenterol Motil 1(2):e13482. https://doi.org/10.1111/nmo.13482
Acknowledgements
This work was supported by the Natural Science Foundation of Shaanxi Province [Grant No. 2021JQ-411] and the National Natural Science Foundation of China [Grant No. 82100567].
Author information
Authors and Affiliations
Contributions
XQ and WC performed research and drafted the paper and contributed equally to this study. SG and NZ analyzed the data. JW and HL revised the manuscript for important intellectual content. FD designed the study and approved the final version to be published. All authors agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All persons designated as authors qualify for authorship, and all those who qualify for authorship are listed.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest.
Additional information
Handling editor: N. Le Floch.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Quan, X., Chen, W., Gao, S. et al. Effects and underlying mechanisms of L-arginine on spontaneous muscle contraction of rat colon. Amino Acids 55, 731–741 (2023). https://doi.org/10.1007/s00726-023-03264-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-023-03264-7