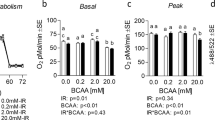
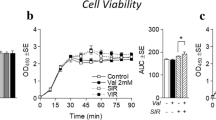
Abstract
Those with insulin resistance often display increased circulating branched-chain amino acids (BCAA), which has been largely attributable to reduced BCAA catabolic capacity. Metabolic stimuli such as exercise activates AMP-activated kinase (AMPK), which promotes the metabolism of BCAA and induction/activation of BCAA catabolic enzymes. Though much attention has been paid to BCAA catabolic machinery, few studies have assessed the effect of AMPK activation on the predominant BCAA transporter, L-type amino acid transporter 1 (LAT1). This study assessed the effect of AMPK activation on LAT1 expression via common chemical AMPK activators in a cell model of skeletal muscle. C2C12 myotubes were treated with either 1 mM AICAR, 1 mM Metformin, or filter-sterilized water (control) for 24 h with either low- (5 mM) or high-glucose (25 mM) media. LAT1 and pAMPK protein content were measured via western blot. BCAA media content was measured using liquid chromatography-mass spectrometry. AICAR treatment significantly increased pAMPK and reduced LAT1 expression. Collectively, pAMPK and LAT1 displayed a significant inverse relationship independent of glucose levels. During low-glucose experiments, AICAR-treated cells had higher BCAA media content compared to other groups, and an inverse relationship between LAT1 and BCAA media content was observed, however, these effects were not consistently observed during high-glucose conditions. Further investigation with AICAR with and without concurrent LAT1 inhibition (via JPH203) also revealed reduced BCAA utilization in AICAR-treated cells regardless of LAT1 inhibition (which also independently reduced BCAA utilization). pAMPK activation via AICAR (but not Metformin) may reduce LAT1 expression and BCAA uptake in a glucose-dependent manner.






Similar content being viewed by others
Availability of data and materials
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Abbreviations
- AMPK:
-
AMP-activated protein kinase
- BCAA:
-
Branched-chain amino acid
- BCAT2:
-
Branched-chain amino acid transaminase 2
- BCKDH:
-
Branched-chain alpha-keto acid dehydrogenase
- BCKDK:
-
Branched-chain alpha-keto acid dehydrogenase kinase
- LAT1:
-
L-type amino acid transporter 1
- mTOR:
-
Mammalian/mechanistic target of rapamycin
- PGC-1α:
-
Peroxisome proliferator-activated receptor gamma coactivator 1-alpha
References
Arany Z, Neinast M (2018) Branched chain amino acids in metabolic disease. Curr Diab Rep 18(10):76. https://doi.org/10.1007/s11892-018-1048-7
Bröer A, Gauthier-Coles G, Rahimi F, van Geldermalsen M, Dorsch D, Wegener A, Bröer S (2019) Ablation of the ASCT2 (SLC1A5) gene encoding a neutral amino acid transporter reveals transporter plasticity and redundancy in cancer cells. J Biol Chem 294(11):4012–4026. https://doi.org/10.1074/jbc.RA118.006378
Cormerais Y, Massard PA, Vucetic M, Giuliano S, Tambutté E, Durivault J, Pouyssegur J (2018) The glutamine transporter ASCT2 (SLC1A5) promotes tumor growth independently of the amino acid transporter LAT1 (SLC7A5). J Biol Chem 293(8):2877–2887. https://doi.org/10.1074/jbc.RA117.001342
Crossland H, Smith K, Idris I, Phillips BE, Atherton PJ, Wilkinson DJ (2020) Exploring mechanistic links between extracellular BCAA & muscle insulin resistance: an in vitro approach. Am J Physiol Cell Physiol. https://doi.org/10.1152/ajpcell.00377.2020
Gannon NP, Schnuck JK, Vaughan RA (2018) BCAA metabolism and insulin sensitivity - dysregulated by metabolic status? Mol Nutr Food Res. https://doi.org/10.1002/mnfr.201700756
Gauthier-Coles G, Vennitti J, Zhang Z, Comb WC, Xing S, Javed K, Bröer S (2021) Quantitative modelling of amino acid transport and homeostasis in mammalian cells. Nat Commun 12(1):5282. https://doi.org/10.1038/s41467-021-25563-x
Hatazawa Y, Tadaishi M, Nagaike Y, Morita A, Ogawa Y, Ezaki O, Miura S (2014) PGC-1alpha-mediated branched-chain amino acid metabolism in the skeletal muscle. PLoS ONE 9(3):e91006. https://doi.org/10.1371/journal.pone.0091006
Hatazawa Y, Qian K, Gong DW, Kamei Y (2018) PGC-1alpha regulates alanine metabolism in muscle cells. PLoS ONE 13(1):e0190904. https://doi.org/10.1371/journal.pone.0190904
Hinkle JS, Rivera CN, Vaughan RA (2021) AICAR stimulates mitochondrial biogenesis and BCAA catabolic enzyme expression in C2C12 myotubes. Biochimie. https://doi.org/10.1016/j.biochi.2021.11.004
Holeček M (2020) Why are branched-chain amino acids increased in starvation and diabetes? Nutrients 12(10):3087. https://doi.org/10.3390/nu12103087
Honda K, Ono K, Mori T, Kochi H (1991) Both induction and activation of the branched-chain 2-oxo acid dehydrogenase complex in primary-cultured rat hepatocytes by clofibrate. J Biochem 109(6):822–827
Kim J, Yang G, Kim Y, Ha J (2016) AMPK activators: mechanisms of action and physiological activities. Exp Mol Med 48:e224. https://doi.org/10.1038/emm.2016.16
Kobayashi R, Murakami T, Obayashi M, Nakai N, Jaskiewicz J, Fujiwara Y, Harris RA (2002) Clofibric acid stimulates branched-chain amino acid catabolism by three mechanisms. Arch Biochem Biophys 407(2):231–240
Lee S, Gulseth HL, Langleite TM, Norheim F, Olsen T, Refsum H, Drevon CA (2021) Branched-chain amino acid metabolism, insulin sensitivity and liver fat response to exercise training in sedentary dysglycaemic and normoglycaemic men. Diabetologia 64(2):410–423. https://doi.org/10.1007/s00125-020-05296-0
Li R, Liu P, Tian Y, Hua Y, Gao Y, He H, Huang Y (2016a) A novel liquid chromatography tandem mass spectrometry method for simultaneous determination of branched-chain amino acids and branched-chain α-keto acids in human plasma. Amino Acids 48(6):1523–1532. https://doi.org/10.1007/s00726-016-2212-5
Li Y, Wei H, Li F, Chen S, Duan Y, Guo Q, Yin Y (2016b) Supplementation of branched-chain amino acids in protein-restricted diets modulates the expression levels of amino acid transporters and energy metabolism associated regulators in the adipose tissue of growing pigs. Anim Nutr 2(1):24–32. https://doi.org/10.1016/j.aninu.2016.01.003
Lian K, Du C, Liu Y, Zhu D, Yan W, Zhang H, Tao L (2015) Impaired adiponectin signaling contributes to disturbed catabolism of branched-chain amino acids in diabetic mice. Diabetes 64(1):49–59. https://doi.org/10.2337/db14-0312
Luo JQ, Chen DW, Yu B (2013) Upregulation of amino acid transporter expression induced by L-leucine availability in L6 myotubes is associated with ATF4 signaling through mTORC1-dependent mechanism. Nutrition 29(1):284–290. https://doi.org/10.1016/j.nut.2012.05.008
Lynch CJ, Adams SH (2014) Branched-chain amino acids in metabolic signalling and insulin resistance. Nat Rev Endocrinol 10(12):723–736. https://doi.org/10.1038/nrendo.2014.171
Mahmassani ZS, Son K, Pincu Y, Munroe M, Drnevich J, Chen J, Boppart MD (2017) α7β1 Integrin regulation of gene transcription in skeletal muscle following an acute bout of eccentric exercise. Am J Physiol Cell Physiol 312(5):C638–C650. https://doi.org/10.1152/ajpcell.00106.2016
Newgard CB (2012) Interplay between lipids and branched-chain amino acids in development of insulin resistance. Cell Metab 15(5):606–614. https://doi.org/10.1016/j.cmet.2012.01.024
Okunushi K, Furihata T, Morio H, Muto Y, Higuchi K, Kaneko M, Anzai N (2020) JPH203, a newly developed anti-cancer drug, shows a preincubation inhibitory effect on L-type amino acid transporter 1 function. J Pharmacol Sci 144(1):16–22. https://doi.org/10.1016/j.jphs.2020.06.006
Poncet N, Mitchell FE, Ibrahim AF, McGuire VA, English G, Arthur JS, Taylor PM (2014) The catalytic subunit of the system L1 amino acid transporter (slc7a5) facilitates nutrient signalling in mouse skeletal muscle. PLoS ONE 9(2):e89547. https://doi.org/10.1371/journal.pone.0089547
Rivera ME, Lyon ES, Vaughan RA (2020) Effect of Metformin on myotube BCAA catabolism. J Cell Biochem 121(1):816–827. https://doi.org/10.1002/jcb.29327
Rivera M, Rivera C, Vaughan R (2021) Branched-chain amino acids at supraphysiological but not physiological levels reduce myotube insulin sensitivity. Diabetes/metab Res Rev. https://doi.org/10.1002/dmrr.3490
Roberson PA, Mobley CB, Romero MA, Haun CT, Osburn SC, Mumford PW, Roberts MD (2020) LAT1 protein content increases following 12 weeks of resistance exercise training in human skeletal muscle. Front Nutr 7:628405. https://doi.org/10.3389/fnut.2020.628405
Shimomura Y, Murakami T, Nakai N, Nagasaki M, Harris RA (2004) Exercise promotes BCAA catabolism: effects of BCAA supplementation on skeletal muscle during exercise. J Nutr 134(6 Suppl):1583s–1587s
Sun H, Wang Y (2019) A new branch connecting thermogenesis and diabetes. Nat Metab 1(9):845–846. https://doi.org/10.1038/s42255-019-0112-1
Walker DK, Drummond MJ, Dickinson JM, Borack MS, Jennings K, Volpi E, Rasmussen BB (2014) Insulin increases mRNA abundance of the amino acid transporter SLC7A5/LAT1 via an mTORC1-dependent mechanism in skeletal muscle cells. Physiol Rep 2(3):e00238. https://doi.org/10.1002/phy2.238
Wu H, Deng X, Shi Y, Su Y, Wei J, Duan H (2016) PGC-1alpha, glucose metabolism and type 2 diabetes mellitus. J Endocrinol 229(3):R99-r115. https://doi.org/10.1530/joe-16-0021
Yamamoto Y, Sawa R, Wake I, Morimoto A, Okimura Y (2017) Glucose-mediated inactivation of AMP-activated protein kinase reduces the levels of L-type amino acid transporter 1 mRNA in C2C12 cells. Nutr Res 47:13–20. https://doi.org/10.1016/j.nutres.2017.08.003
Yoneshiro T, Wang Q, Tajima K, Matsushita M, Maki H, Igarashi K, Kajimura S (2019) BCAA catabolism in brown fat controls energy homeostasis through SLC25A44. Nature 572(7771):614–619. https://doi.org/10.1038/s41586-019-1503-x
Funding
Support for this work was provided by the Department of Exercise Science within the Congdon School of Health Sciences. Additional support was provided by the High Point University Undergraduate Research and Creative Works Small Project Support Grant (F-21-02), the Congdon School of Health Sciences Summer Undergraduate Research Fellowship (SURF), and the Wanek School of Undergraduate Sciences Summer Research Program in the Sciences (SuRPS). We would like also to thank the Department of Physical Therapy (Congdon School of Health Sciences) for the use of shared lab space and equipment. Instrumentation support was provided by the Shimadzu Partnership for Academics, Research and Quality of Life (SPARQ) Program.
Author information
Authors and Affiliations
Contributions
CNR, RMW, ZAB, SAM, and AJW conducted experiments and assisted with manuscript preparation. RAV conceived the study, conducted, and oversaw experiments, performed all statistical analyses, and oversaw manuscript preparation. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Authors and contributors declare no conflict of interest.
Ethical approval
Not applicable. This research falls outside of human or animal studies and institutional ethical approval was not required.
Additional information
Handling editor: S. Broeer.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rivera, C.N., Watne, R.M., Brown, Z.A. et al. Effect of AMPK activation and glucose availability on myotube LAT1 expression and BCAA utilization. Amino Acids 55, 275–286 (2023). https://doi.org/10.1007/s00726-022-03224-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-022-03224-7