Abstract
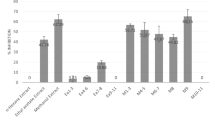
Chia (Salvia hispanica) seed peptides have drawn attention because of their antioxidant, antihypertensive and anti-inflammatory activities, making them ideal candidates for development of cosmeceutical skin products. However, there are no preceding reports that address their aging-related enzyme inhibitory activities. The aim of this study was to investigate the in vitro and in silico inhibitory activity of chia seed peptides towards the main aging-related enzymes. Enzyme-inhibition activity of < 3 kDa chia seed peptides towards collagenase, hyaluronidase, tyrosinase, and elastase was evaluated. Further fractions were obtained by size exclusion chromatography (SEC) and re-tested for enzyme inhibitory activity. Peptide sequences were identified from the most effective fraction and used for in silico analysis. The < 3 kDa peptides exhibited inhibitory activities towards elastase (65.32%, IC50 = 0.43 mg/mL), tyrosinase (58.74%, IC50 = 0.66 mg/mL), hyaluronidase (26.96%, IC50 = 1.28 mg/mL), and collagenase (28.90%, IC50 = 1.41 mg/mL). They showed mixed-type inhibition patterns towards elastase and hyaluronidase, while a non-competitive inhibition pattern was observed towards collagenase and tyrosinase. Fraction II obtained by SEC, showed higher enzyme inhibitory activity. Seven peptides were identified in this fraction (APHWYTN, DQNPRSF, GDAHWAY, GDAHWTY, GDAHWVY, GFEWITF, and KKLKRVYV), which according to in silico analysis, possess 19–29 enzyme–peptide pair interactions towards elastase and three peptide sequences shared homology sequence (GDAHW). These results demonstrate that peptides from chia seeds may contribute in the improvement of skin health by offering protection against aging-related enzymes by preventing degradation of the protein matrix on the skin; however, further in vivo studies are needed to evaluate its actual capability.






Similar content being viewed by others
References
Aguilar-Toalá JE, Hernández-Mendoza A, González-Córdova AF, Vallejo-Cordoba B, Liceaga AM (2019) Potential role of natural bioactive peptides for development of cosmeceutical skin products. Peptides 122:170170. https://doi.org/10.1016/j.peptides.2019.170170
Alamgir ANM (2017) Classification of drugs, nutraceuticals, functional food, and cosmeceuticals; proteins, peptides, and enzymes as drugs. In: Alamgir ANM (ed) Therapeutic use of medicinal plants and their extracts: Pharmacognosy, vol 1. Springer International Publishing, Cham, pp 125–175
Azmi N, Hashim P, Hashim DM, Halimoon N, Majid NMN (2014) Anti-elastase, anti-tyrosinase and matrix metalloproteinase-1 inhibitory activity of earthworm extracts as potential new anti-aging agent. Asian Pac J Trop Biomed 4(Suppl 1):S348–S352
Clemente A (2000) Enzymatic protein hydrolysates in human nutrition. Trends Food Sci Technol 11:254–256
Coelho MS, Soares-Freitas RAM, Arêas JAG, Gandra EA, MdlM S-M (2018) Peptides from chia present antibacterial activity and inhibit cholesterol synthesis. Plant Foods Hum Nutr 73(2):101–107
Cotabarren J, Rosso AM, Tellechea M, García-Pardo J, Rivera JL, Obregón WD, Parisi MG (2019) Adding value to the chia (Salvia hispanica L.) expeller: production of bioactive peptides with antioxidant properties by enzymatic hydrolysis with Papain. Food Chem 274:848–856
Crown OO, Olayeriju OS, Kolawole AO, Akinmoladun AC, Olaleye MT, Akindahunsi AA (2017) Mobola plum seed methanolic extracts exhibit mixed type inhibition of angiotensin I-converting enzyme in vitro. Asian Pac J Trop Biomed 7(12):1079–1084
Dorni AIC, Amalraj A, Gopi S, Varma K, Anjana SN (2017) Novel cosmeceuticals from plants—an industry guided review. J Appl Res Med Aromat Plants 7:1–26
Engelking LR (2015) Enzyme kinetics, Chapter 6. In: Engelking LR (ed) Textbook of veterinary physiological chemistry, 3rd edn. Academic Press, Boston, pp 32–38
Facino RM, Carini M, Stefani R, Aldini G, Saibene L (1995) Anti-elastase and anti-hyaluronidase activities of saponins and sapogenins from Hedera helix, Aesculus hippocastanum, and Ruscus aculeatus: Factors contributing to their efficacy in the treatment of venous insufficiency. Arch Pharm 328(10):720–724
FDA (2015) Select Committee on GRAS Substances (SCOGS) Opinion: Enzymatically hydrolyzed casein, enzymatically hydrolyzed protein, acid hydrolyzed protein, soy sauces, yeast autolyzates, GRAS Substances (SCOGS) Database
García-Mora P, Martín-Martínez M, Angeles Bonache M, González-Múniz R, Peñas E, Frias J, Martinez-Villaluenga C (2017) Identification, functional gastrointestinal stability and molecular docking studies of lentil peptides with dual antioxidant and angiotensin I converting enzyme inhibitory activities. Food Chem 221:464–472
Grancieri M, Martino HSD, Gonzalez de Mejia E (2019) Digested total protein and protein fractions from chia seed (Salvia hispanica L.) had high scavenging capacity and inhibited 5-LOX, COX-1-2, and iNOS enzymes. Food Chem 289:204–214
Hall F, Liceaga A (2020) Effect of microwave-assisted enzymatic hydrolysis of cricket (Gryllodes sigillatus) protein on ACE and DPP-IV inhibition and tropomyosin-IgG binding. J Funct Foods 64:103634. https://doi.org/10.1016/j.jff.2019.103634
Hall F, Johnson PE, Liceaga A (2018) Effect of enzymatic hydrolysis on bioactive properties and allergenicity of cricket (Gryllodes sigillatus) protein. Food Chem 262:39–47
Hong GP, Min SG, Jo YJ (2019) Anti-oxidative and anti-aging activities of porcine by-product collagen hydrolysates produced by commercial proteases: effect of hydrolysis and ultrafiltration. Molecules 24(6):1104. https://doi.org/10.3390/molecules24061104
Huang MCJ, Tang J (2015) Probiotics in personal care products. Microbiol Discov 3(1):5. https://doi.org/10.7243/2052-6180-3-5
Kubglomsong S, Theerakulkait C, Reed RL, Yang L, Maier CS, Stevens JF (2018) Isolation and identification of tyrosinase-inhibitory and copper-chelating peptides from hydrolyzed rice-bran-derived albumin. J Agr Food Chem 66(31):8346–8354
Kurcinski M, Jamroz M, Blaszczyk M, Kolinski A, Kmiecik S (2015) CABS-dock web server for the flexible docking of peptides to proteins without prior knowledge of the binding site. Nucleic Acids Res 43:419–424
Lemes AC, Sala L, Ores JdC, Braga ARC, Egea MB, Fernandes KF (2016) A review of the latest advances in encrypted bioactive peptides from protein-rich waste. Int J Mol Sci 17(6):950. https://doi.org/10.3390/ijms17060950
Lima TN, Pedriali Moraes CA (2018) Bioactive peptides: applications and relevance for cosmeceuticals. Cosmetics 5(1):21. https://doi.org/10.3390/cosmetics5010021
Limbert G, Masen MA, Pond D, Graham HK, Sherratt MJ, Jobanputra R, McBride A (2019) Biotribology of the ageing skin—Why we should care. Biotribology 17:75–90
Lintner K, Mas-Chamberlin C, Mondon P, Peschard O, Lamy L (2009) Cosmeceuticals and active ingredients. Clin Dermatol 27(5):461–468
Liu C, Fang L, Min W, Liu J, Li H (2018) Exploration of the molecular interactions between angiotensin-I-converting enzyme (ACE) and the inhibitory peptides derived from hazelnut (Corylus heterophylla Fisch.). Food Chem 245:471–480
Liu Y, Zheng L, Xu J, Sun-waterhouse D, Sun B, Su G, Zhao M (2020) Identification of novel peptides with high stability against in vitro hydrolysis from bovine elastin hydrolysates and evaluation of their elastase inhibitory activity. Int J Food Sci Technol 55(1):99–108
Lorencini M, Brohem CA, Dieamant GC, Zanchin NIT, Maibach HI (2014) Active ingredients against human epidermal aging. Ageing Res Rev 15:100–115
Morganti P (2008) Reflections on cosmetics, cosmeceuticals, and nutraceuticals. Clin Dermatol 26(4):318–320
Mukherjee PK, Maity N, Nema NK, Sarkar BK (2011) Bioactive compounds from natural resources against skin aging. Phytomedicine 19(1):64–73
Nakchum L, Kim SM (2016) Preparation of squid skin collagen hydrolysate as an antihyaluronidase, antityrosinase, and antioxidant agent. Prep Biochem Biotech 46(2):123–130
Ramos-e-Silva M, Celem LR, Ramos-e-Silva S, Fucci-da-Costa AP (2013) Anti-aging cosmetics: facts and controversies. Clin Dermatol 31(6):750–758
Roskoski R (2015) Michaelis-Menten Kinetics, Reference Module in Biomedical Sciences. Elsevier, Amsterdam
Schaafsma G (2009) Safety of protein hydrolysates, fractions thereof and bioactive peptides in human nutrition. Eur J Clin Nutr 63:1161. https://doi.org/10.1038/ejcn.2009.56
Shin KO, Park HS (2019) Antiaging cosmeceuticals in korea and open innovation in the era of the 4th industrial revolution: from research to business. Sustainability 11(3):898. https://doi.org/10.3390/su11030898
Shin JW, Kwon SH, Choi JY, Na JI, Huh CH, Choi HR, Park KC (2019) Molecular mechanisms of dermal aging and antiaging approaches. Int J Mol Sci 20(9):2126. https://doi.org/10.3390/ijms20092126
Sun L, Warren FJ, Netzel G, Gidley MJ (2016) 3 or 3′-Galloyl substitution plays an important role in association of catechins and theaflavins with porcine pancreatic α-amylase: the kinetics of inhibition of α-amylase by tea polyphenols. J Funct Foods 26:144–156
Tabassum N, Hamdani M (2014) Plants used to treat skin diseases. Pharmacogn Rev 8(15):52–60
Taddese S, Weiss AS, Neubert RHH, Schmelzer CEH (2008) Mapping of macrophage elastase cleavage sites in insoluble human skin elastin. Matrix Biol 27(5):420–428
Taofiq O, González-Paramás AM, Martins A, Barreiro MF, Ferreira ICFR (2016) Mushrooms extracts and compounds in cosmetics, cosmeceuticals and nutricosmetics—a review. Ind Crop Prod 90:38–48
Tobin DJ (2017) Introduction to skin aging. J Tissue Viability 26(1):37–46
Urbizo-Reyes UC, San Martin-González MF, Garcia-Bravo J, López Malo A, Liceaga AM (2019) Physicochemical characteristics of chia seed (Salvia hispanica) protein hydrolysates produced using ultrasonication followed by microwave-assisted hydrolysis. Food Hydrocoll 97:105187. https://doi.org/10.1016/j.foodhyd.2019.105187
Vasconcelos A, Azoia NG, Carvalho AC, Gomes AC, Güebitz G, Cavaco-Paulo A (2011) Tailoring elastase inhibition with synthetic peptides. Eur J Pharmacol 666(1):53–60
Venkatesan J, Anil S, Kim SK, Shim MS (2017) Marine fish proteins and peptides for cosmeceuticals: a review. Mar Drugs 15(5):143. https://doi.org/10.3390/md15050143
Verma RP, Hansch C (2007) Matrix metalloproteinases (MMPs): chemical–biological functions and (Q)SARs. Bioorganic Med Chem 15(6):2223–2268
Wu Y, Choi MH, Li J, Yang H, Shin HJ (2016) Mushroom cosmetics: the present and future. Cosmetics 3(3):22. https://doi.org/10.3390/cosmetics3030022
Acknowledgements
The authors would like to thank Uriel C. Urbizo Reyes (Protein Chemistry and Bioactive Peptides Laboratory, Department of Food Sciences) for the technical support. The authors would also like to thank Dr. Emma Doud with the Indiana University Proteomics at Indiana University School of Medicine for her assistance with the proteomics analysis in this study. In addition, the authors express their appreciation to Patrick J. Polowsky (Agricultural Sciences Education and Communication, Purdue University) for the design of the illustration (Fig. 6) used in this article.
Funding
The present work was supported by the USDA-NIFA, Hatch Act formula funds (project 1019794) in the College of Agriculture, Purdue University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants performed by any of the authors.
Additional information
Handling editor: M. S. Palma.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Aguilar-Toalá, J.E., Liceaga, A.M. Identification of chia seed (Salvia hispanica L.) peptides with enzyme inhibition activity towards skin-aging enzymes. Amino Acids 52, 1149–1159 (2020). https://doi.org/10.1007/s00726-020-02879-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-020-02879-4