Abstract
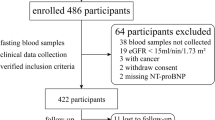
In heart failure (HF), metabolic disturbances represent functional perturbations in peripheral tissues and also predict patient outcomes. This study developed a simplified essential amino acid-based profile and tested whether it could improve prognostication. Plasma essential amino acids and lipidomics were measured on 1084 participants. The initial cohort included 94 normal controls and 599 patients hospitalized due to acute/decompensated HF. The validation cohort included 391 HF patients. Patients were followed for composite events (death/HF related re-hospitalization) and were categorized into three groups: high risk type 1 (leucine ≥145 μM and phenylalanine ≥ 88.9 μM), high risk type 2 (leucine < 81.2 μM), and low risk (other). Types 1 and 2 were associated with higher event rates [hazard ratio (95% confidence intervals) = 1.88 (1.27–2.79) and 7.71 (4.97–11.9), respectively, p < 0.001]. Compared to the low-risk group, both types of high-risk patients were older and had lower blood pressure and estimated glomerular filtration rates, but higher B-type natriuretic peptides (BNP). In addition, type 1 was associated with more incompletely metabolized lipids in the blood; type 2 patients had lower body mass indexes, rates of using guideline-based medications, and levels of cholesterol, hemoglobin, and albumin. The prognostic value of types 1 and 2 remained significant after adjusting for age, BNP and other risk factors. The value of using high-risk types for prognosis was confirmed in the validation cohort. In conclusion, simplified essential amino acid-based profiling identified two high-risk populations and provided metabolic information and prognostic value additive to traditional risk factors.

Similar content being viewed by others
Abbreviations
- BNP:
-
B-type natriuretic peptide
- CI:
-
Confidence interval
- eGFR:
-
Estimated glomerular filtration rate
- HR:
-
Hazard ratio
- HF:
-
Heart failure
- Leu:
-
Leucine
- LVEF:
-
Left ventricular ejection fraction
- Phe:
-
Phenylalanine
- ROC:
-
Receiver operating characteristic curve
- UPLC:
-
Ultra-performance liquid chromatography
References
Ahmad T, Kelly JP et al (2016) Prognostic implications of long-chain acylcarnitines in heart failure and reversibility with mechanical circulatory support. J Am Coll Cardiol 67(3):291–299
Aquilani R, La Rovere MT et al (2012) Preserved muscle protein metabolism in obese patients with chronic heart failure. Int J Cardiol 160(2):102–108
Cheng ML, Wang CH et al (2015) Metabolic disturbances identified in plasma are associated with outcomes in patients with heart failure: diagnostic and prognostic value of metabolomics. J Am Coll Cardiol 65(15):1509–1520
Chioncel O, Collins SP et al (2016) Natriuretic peptide-guided management in heart failure. J Cardiovasc Med 17(8):556–568
Frank MP, Powers RW (2007) Simple and rapid quantitative high-performance liquid chromatographic analysis of plasma amino acids. J Chromatogr B Analyt Technol Biomed Life Sci 852(1–2):646–649
Griffin JL, Atherton H et al (2011) Metabolomics as a tool for cardiac research. Nat Rev Cardiol 8(11):630–643
Hakuno D, Hamba Y et al (2015) Plasma amino acid profiling identifies specific amino acid associations with cardiovascular function in patients with systolic heart failure. PLoS One 10(2):e0117325
Hunter WG, Kelly JP et al (2016) Metabolic dysfunction in heart failure: diagnostic, prognostic, and pathophysiologic insights from metabolomic profiling. Curr Heart Fail Rep 13(3):119–131
Lavie CJ, Berra K et al (2013) Formal cardiac rehabilitation and exercise training programs in heart failure: evidence for substantial clinical benefits. J Cardiopulm Rehabil Prev 33(4):209–211
Liu Z, Barrett EJ (2002) Human protein metabolism: its measurement and regulation. Am J Physiol Endocrinol Metab 283(6):E1105–E1112
Maisel AS, Clopton P et al (2004) Impact of age, race, and sex on the ability of B-type natriuretic peptide to aid in the emergency diagnosis of heart failure: results from the breathing not properly (BNP) multinational study. Am Heart J 147(6):1078–1084
Nagabhushan VS, Narasinga Rao BS (1978) Studies on 3-methylhistidine metabolism in children with protein-energy malnutrition. Am J Clin Nutr 31(8):1322–1327
Nishijima Y, Sridhar A et al (2011) Tetrahydrobiopterin depletion and NOS2 uncoupling contribute to heart failure-induced alterations in atrial electrophysiology. Cardiovasc Res 91(1):71–79
Pappa-Louisi A, Nikitas P et al (2007) Optimization of separation and detection of 6-aminoquinolyl derivatives of amino acids by using reversed-phase liquid chromatography with on line UV, fluorescence and electrochemical detection. Anal Chim Acta 593(1):92–97
Rajadurai J, Tse HF et al (2017) Understanding the epidemiology of heart failure to improve management practices: An Asia-Pacific Perspective. J Card Fail 23(4):327–339
Rehman SU, Mueller T et al (2008) Characteristics of the novel interleukin family biomarker ST2 in patients with acute heart failure. J Am Coll Cardiol 52(18):1458–1465
Reid C (2006) Frequency of under- and overfeeding in mechanically ventilated ICU patients: cause and possible consequences. J Hum Nutr Diet 19(1):13–22
Savarese G, Lund LH (2017) Global public health burden of heart failure. Card Fail Rev 3(1):7–11
Schooneman MG, Vaz FM et al (2013) Acylcarnitines: reflecting or inflicting insulin resistance? Diabetes 62(1):1–8
Sperry BW, Ruiz G et al (2015) Hospital readmission in heart failure, a novel analysis of a longstanding problem. Heart Fail Rev 20(3):251–258
Wang CH, Yang NI et al (2016) Estimating systemic fibrosis by combining galectin-3 and ST2 provides powerful risk stratification value for patients after acute decompensated heart failure. Cardiol J 23(5):563–572
Wang CH, Cheng ML et al (2017) Metabolic profile provides prognostic value better than galectin-3 in patients with heart failure. J Cardiol 70(1):92–98
Ziolo MT, Maier LS et al (2004) Myocyte nitric oxide synthase 2 contributes to blunted beta-adrenergic response in failing human hearts by decreasing Ca2+ transients. Circulation 109(15):1886–1891
Acknowledgement
The authors thank Cardiology Section, Department of Internal Medicine, Chang Gung Memorial Hospital, Keeling, Taiwan for providing samples from patients and normal controls. We also thank Healthy Aging Research Center, Chang Gung University from the Featured Areas Research Center Program within the Framework of the Higher Education Sprout Project by the Ministry of Education in Taiwan.
Funding
This study was supported in part by the Ministry of Science and Technology of Taiwan (MOST105-2314-B-182-046-MY2, 107-2314-B-182-071-MY2); Chang Gung Memorial Hospital (CMRPG2C0313, G2E0351, G2G0601, G2G0581); and the Ministry of Education of Taiwan (EMRPD1G0251, EMRPD1H0401).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. This article does not contain any studies with animals performed by any of the authors.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Handling Editor: D. Tsikas.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, CH., Cheng, ML. & Liu, MH. Simplified plasma essential amino acid-based profiling provides metabolic information and prognostic value additive to traditional risk factors in heart failure. Amino Acids 50, 1739–1748 (2018). https://doi.org/10.1007/s00726-018-2649-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-018-2649-9