Abstract
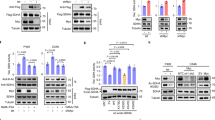
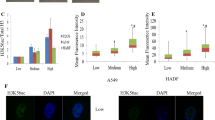
Carnosine (β-alanyl-l-histidine) affects a plethora of signaling pathways and genes in different biological systems. Although known as a radical scavenger, not all of these effects can simply be ascribed to its chemical nature. As previous experiments pointed towards the possibility that carnosine affects epigenetic regulation via histone acetylation, we investigated this hypothesis using the glioblastoma cell lines U87 and T98G in which carnosine’s anti-neoplastic effect is accompanied by increased expression of pyruvate dehydrogenase kinase 4. Viability and expression of PDK4 was analyzed after incubation in carnosine and different histone deacetylase inhibitors (HDACi) using cell-based assays and qRT-PCR. In addition, chromatin immunoprecipitation (ChIP) experiments were performed and the global influence of carnosine on histone H3 acetylation was analyzed by Western blot. Carnosine as well as the HDACi used increased expression of PDK4. In addition, all compounds reduced cell viability, although differences were observed with regard to magnitude and required concentrations. ChIP analysis revealed increased acetylation of histone H3 in the PDK4 promoter of U87 and T98G cells (~ 1.3- and ~ 1.7-fold, respectively) 6 h after the addition of carnosine (50 mM) followed by increased expression of PDK4 mRNA. Western blots did not detect a general increase of H3 acetylation at a genome-wide scale under the influence of carnosine. Our experiments for the first time demonstrate that carnosine influences epigenetic regulation via increased histone acetylation.





Similar content being viewed by others
References
Araki M, Nozaki Y, Motojima K (2007) Transcriptional regulation of metabolic switching PDK4 gene under various physiological conditions. Yakugaku Zasshi 127:153–162
Baran EJ (2000) Metal complexes of carnosine. Biochemistry (Mosc.) 65:789–797
Barrès R, Yan J, Egan B, Treebak JT, Rasmussen M, Fritz T, Caidahl K, Krook A, O’Gorman DJ, Zierath JR (2012) Acute exercise remodels promoter methylation in human skeletal muscle. Cell Metab 15:405–411. https://doi.org/10.1016/j.cmet.2012.01.001
Blancquaert L, Everaert I, Missinne M, Baguet A, Stegen S, Volkaert A, Petrovic M, Vervaet C, Achten E, de Maeyer M, de Henauw S, Derave W (2017) Effects of histidine and β-alanine Supplementation on human muscle carnosine storage. Med Sci Sports Exerc 49:602–609. https://doi.org/10.1249/MSS.0000000000001213
Boldyrev AA, Aldini G, Derave W (2013) Physiology and pathophysiology of carnosine. Physiol Rev 93:1803–1845. https://doi.org/10.1152/physrev.00039.2012
Chavez-Blanco A, Segura-Pacheco B, Perez-Cardenas E, Taja-Chayeb L, Cetina L, Candelaria M, Cantu D, Gonzalez-Fierro A, Garcia-Lopez P, Zambrano P, Perez-Plasencia C, Cabrera G, Trejo-Becerril C, Angeles E, Duenas-Gonzalez A (2005) Histone acetylation and histone deacetylase activity of magnesium valproate in tumor and peripheral blood of patients with cervical cancer. A phase I study. Mol Cancer 4:22. https://doi.org/10.1186/1476-4598-4-22
Cohen I, Poręba E, Kamieniarz K, Schneider R (2011) Histone modifiers in cancer: friends or foes? Genes Cancer 2:631–647. https://doi.org/10.1177/1947601911417176
Gardner MLG, Illingworth KM, Kelleher J, Wood D (1991) Intestinal-absorption of the intact peptide carnosine in man, and comparison with intestinal permeability to lactulose. J Physiol 439:411–422
Gaunitz F, Heise K (2003) HTS compatible assay for antioxidative agents using primary cultured hepatocytes. Assay Drug Dev Technol 1:469–477
Gaunitz F, Hipkiss AR (2012) Carnosine and cancer: a perspective. Amino Acids 43:135–142. https://doi.org/10.1007/s00726-012-1271-5
Gulewitsch W, Amiradzibi S (1900) Ueber das Carnosin, eine neue organische Base des Fleischextraktes. Ber Dtsch Chem Ges 33:1902–1903
Han H, Li W, Shen H, Zhang J, Zhu Y, Li Y (2016) microRNA-129-5p, a c-Myc negative target, affects hepatocellular carcinoma progression by blocking the Warburg effect. J Mol Cell Biol. https://doi.org/10.1093/jmcb/mjw010
He Y, Cai C, Tang D, Sun S, Li H (2014) Effect of histone deacetylase inhibitors trichostatin A and valproic acid on hair cell regeneration in zebrafish lateral line neuromasts. Front Cell Neurosci 8:382. https://doi.org/10.3389/fncel.2014.00382
Hipkiss AR, Gaunitz F (2014) Inhibition of tumour cell growth by carnosine: some possible mechanisms. Amino Acids 46:327–337
Howitz KT, Bitterman KJ, Cohen HY, Lamming DW, Lavu S, Wood JG, Zipkin RE, Chung P, Kisielewski A, Zhang L-L, Scherer B, Sinclair DA (2003) Small molecule activators of sirtuins extend Saccharomyces cerevisiae lifespan. Nature 425:191–196. https://doi.org/10.1038/nature01960
Iovine B, Iannella ML, Nocella F, Pricolo MR, Baldi MR, Bevilacqua MA (2011) Carnosine inhibits KRas-mediated HCT-116 proliferation by affecting ATP and ROS production. Cancer Lett 315:122–128. https://doi.org/10.1016/j.canlet.2011.07.021
Iovine B, Oliviero G, Garofalo M, Orefice M, Nocella F, Borbone N, Piccialli V, Centore R, Mazzone M, Piccialli G, Bevilacqua MA (2014) The anti-proliferative effect of l-carnosine correlates with a decreased expression of hypoxia inducible factor 1 alpha in human colon cancer cells. PLoS ONE 9:e96755. https://doi.org/10.1371/journal.pone.0096755
Ito-Kato E, Suzuki N, Maeno M, Takada T, Tanabe N, Takayama T, Ito K, Otsuka K (2004) Effect of carnosine on runt-related transcription factor-2/core binding factor alpha-1 and Sox9 expressions of human periodontal ligament cells. J Periodontal Res 39:199–204
Kohen R, Yamamoto Y, Cundy KC, Ames BN (1988) Antioxidant activity of carnosine, homocarnosine, and anserine present in muscle and brain. Proc Natl Acad Sci USA 85:3175–3179
Kulebyakin K, Karpova L, Lakonsteva E, Krasavin M, Boldyrev A (2012) Carnosine protects neurons against oxidative stress and modulates the time profile of MAPK cascade signaling. Amino Acids 43:91–96. https://doi.org/10.1007/s00726-011-1135-4
Kuo M-H, Allis CD (1998) Roles of histone acetyltransferases and deacetylases in gene regulation. BioEssays 20:615–626
Kwon H-S, Harris RA (2004) Mechanisms responsible for regulation of pyruvate dehydrogenase kinase 4 gene expression. Adv Enzyme Regul 44:109–121. https://doi.org/10.1016/j.advenzreg.2003.11.020
Kwon H-S, Huang B, Ho Jeoung N, Wu P, Steussy CN, Harris RA (2006) Retinoic acids and trichostatin A (TSA), a histone deacetylase inhibitor, induce human pyruvate dehydrogenase kinase 4 (PDK4) gene expression. Biochim Biophys Acta 1759:141–151. https://doi.org/10.1016/j.bbaexp.2006.04.005
Letzien U, Oppermann H, Meixensberger J, Gaunitz F (2014) The antineoplastic effect of carnosine is accompanied by induction of PDK4 and can be mimicked by L-histidine. Amino Acids. https://doi.org/10.1007/s00726-014-1664-8
Mackay HJ, Hirte H, Colgan T, Covens A, MacAlpine K, Grenci P, Wang L, Mason J, Pham P-A, Tsao M-S, Pan J, Zwiebel J, Oza AM (2010) Phase II trial of the histone deacetylase inhibitor belinostat in women with platinum resistant epithelial ovarian cancer and micropapillary (LMP) ovarian tumours. Eur J Cancer 46:1573–1579. https://doi.org/10.1016/j.ejca.2010.02.047
McFarland GA, Holliday R (1994) Retardation of the senescence of cultured human diploid fibroblasts by carnosine. Exp Cell Res 212:167–175
Oppermann H, Schnabel L, Meixensberger J, Gaunitz F (2016) Pyruvate attenuates the anti-neoplastic effect of carnosine independently from oxidative phosphorylation. Oncotarget 7:85848–85860. https://doi.org/10.18632/oncotarget.13039
Smith EC (1938) The buffering of muscle in rigor; protein, phosphate and carnosine. J Physiol 92:336–343
Venturelli S, Berger A, Böcker A, Busch C, Weiland T, Noor S, Leischner C, Schleicher S, Mayer M, Weiss TS, Bischoff SC, Lauer UM, Bitzer M (2013) Resveratrol as a pan-HDAC inhibitor alters the acetylation status of histone corrected proteins in human-derived hepatoblastoma cells. PLoS ONE 8:e73097. https://doi.org/10.1371/journal.pone.0073097
Wang J-P, Yang Z-T, Liu C, He Y-H, Zhao S-S (2013) l-carnosine inhibits neuronal cell apoptosis through signal transducer and activator of transcription 3 signaling pathway after acute focal cerebral ischemia. Brain Res 1507:125–133. https://doi.org/10.1016/j.brainres.2013.02.032
Way JM, Harrington WW, Brown KK, Gottschalk WK, Sundseth SS, Mansfield TA, Ramachandran RK, Willson TM, Kliewer SA (2001) Comprehensive messenger ribonucleic acid profiling reveals that peroxisome proliferator-activated receptor gamma activation has coordinate effects on gene expression in multiple insulin-sensitive tissues. Endocrinology 142:1269–1277. https://doi.org/10.1210/endo.142.3.8037
Xu G, Wang J, Wu Z, Qian L, Dai L, Wan X, Tan M, Zhao Y, Wu Y (2014) SAHA regulates histone acetylation, butyrylation, and protein expression in neuroblastoma. J Proteome Res 13:4211–4219. https://doi.org/10.1021/pr500497e
Zhang Z, Miao L, Wu X, Liu G, Peng Y, Xin X, Jiao B, Kong X (2014) Carnosine inhibits the proliferation of human gastric carcinoma cells by retarding Akt/mTOR/p70S6 K signaling. J Cancer 5:382–389. https://doi.org/10.7150/jca.8024
Acknowledgements
We like to thank Flamma (Flamma s.p.a. Chignolo d’Isola, Italy (http://www.flammagroup.com)) for the generous supply with very high quality carnosine for all of our experiments. In addition, we like to thank Dr. Hans-Heinrich Foerster from the Genolytic GmbH (Leipzig, Germany) for genotyping and confirmation of cell identity. We also thank Berthold Technologies (Bad Wildbad, Germany) for providing us a Mithras LB 940 Multimode Microplate Reader and last but not least Mr. Rainer Baran-Schmidt for technical assistance.
Author information
Authors and Affiliations
Contributions
AA and HO performed the experiments presented in the manuscript. CS performed the experiments presented in Supplement 3. HO, JM and FG supervised the experiments. All authors read and contributed to writing the manuscript. FG supervised and planned the experiments, analyzed the data together with AA and HO and wrote the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no potential conflict of interest.
Additional information
Handling Editor: W. Derave.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Oppermann, H., Alvanos, A., Seidel, C. et al. Carnosine influences transcription via epigenetic regulation as demonstrated by enhanced histone acetylation of the pyruvate dehydrogenase kinase 4 promoter in glioblastoma cells. Amino Acids 51, 61–71 (2019). https://doi.org/10.1007/s00726-018-2619-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-018-2619-2