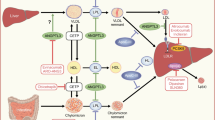
Abstract
Reduced availability of nitric oxide (NO) in the vasculature is a major factor contributing to the impaired action of insulin on blood flow and, therefore, insulin resistance in obese and diabetic subjects. Available evidence shows that vascular insulin resistance plays an important role in the pathogenesis of cardiovascular disease, the leading cause of death in developed nations. Interestingly, increased concentrations of l-leucine in the plasma occur in obese humans and other animals with vascular dysfunction. Among branched-chain amino acids, l-leucine is unique in inhibiting NO synthesis from l-arginine in endothelial cells and may modulate cardiovascular homeostasis in insulin resistance. Results of recent studies indicate that l-leucine is an activator of glutamine:fructose-6-phosphate aminotransferase (GFAT), which is the first and a rate-controlling enzyme in the synthesis of glucosamine (an inhibitor of endothelial NO synthesis). Through stimulating the mammalian target of rapamycin signaling pathway and thus protein synthesis, l-leucine may enhance GFAT protein expression, thereby inhibiting NO synthesis in endothelial cells. We propose that reducing circulating levels of l-leucine or endothelial GFAT activity may provide a potentially novel strategy for preventing and/or treating cardiovascular disease in obese and diabetic subjects. Such means may include dietary supplementation with either α-ketoglutarate to enhance the catabolism of l-leucine in the small intestine and other tissues or with N-ethyl-l-glutamine to inhibit GFAT activity in endothelial cells. Preventing leucine-induced activation of GFAT by nutritional supplements or pharmaceutical drugs may contribute to improved cardiovascular function by enhancing vascular NO synthesis.






Similar content being viewed by others
Abbreviations
- BCAA:
-
Branched-chain amino acids
- BCAT:
-
Branched-chain amino acid transaminase
- BCKAD:
-
Branched-chain α-ketoacid dehydrogenase
- BH4:
-
(6R)-5,6,7,8-Tetrahydro-l-biopterin
- EDCF:
-
Endothelium-derived constricting factor
- EDRF:
-
Endothelium-derived relaxing factor
- FAD:
-
Flavin adenine dinucleotide
- FMN:
-
Flavin mononucleotide
- GFAT:
-
Glutamine:fructose-6-phosphate aminotransferase
- KIC:
-
α-Ketoisocaproate
- mTOR:
-
Mammalian target of rapamycin
- NADPH:
-
Nicotinamide adenine dinucleotide phosphate
- NO:
-
Nitric oxide
- NOS:
-
Nitric oxide synthase
References
Alderton WK, Cooper CE, Knowles RG (2001) Nitric oxide synthases: structure, function and inhibition. Biochem J 357:593–615
Assaad H, Yao K, Tekwe CD et al (2014a) Analysis of energy expenditure in diet-induced obese rats. Front Biosci 19:967–985
Assaad H, Zhou L, Carroll RJ et al (2014b) Rapid publication-ready MS-Word tables for one-way ANOVA. SpringerPlus 3:474
Baron AD, Brechtel G (1993) Insulin differentially regulates systemic and skeletal muscle vascular resistance. Am J Physiol 265:E61–E67
Baron AD, Brechtel-Hook G, Johnson A et al (1993) Skeletal muscle blood flow. A possible link between insulin resistance and blood pressure. Hypertension 21:129–135
Baron AD, Steinberg HO, Chaker H et al (1995) Insulin-mediated skeletal muscle vasodilation contributes to both insulin sensitivity and responsiveness in lean humans. J Clin Invest 96:786–792
Bouloumie A, Bauersachs J, Linz W et al (1997) Endothelial dysfunction coincides with an enhanced nitric oxide synthase expression and superoxide anion production. Hypertension 30:934–941
Boveris A, Costa LE, Poderoso JJ et al (2000) Regulation of mitochondrial respiration by oxygen and nitric oxide. Ann N Y Acad Sci 899:121–135
Buga GM, Wei LH, Bauer PM et al (1998) N G-hydroxy-l-arginine and nitric oxide inhibit Caco-2 tumor cell proliferation by distinct mechanisms. Am J Physiol 275:R1256–R1264
Buse MG, Robinson KA, Gettys TW et al (1997) Increased activity of the hexosamine synthesis pathway in muscles of insulin-resistant ob/ob mice. Am J Physiol 272:E1080–E1088
Caballero AE, Arora S, Saouaf R et al (1999) Microvascular and macrovascular reactivity is reduced in subjects at risk for type 2 diabetes. Diabetes 48:1856–1862
Cayatte AJ, Palacino JJ, Horten K et al (1994) Chronic inhibition of nitric oxide production accelerates neointima formation and impairs endothelial function in hypercholesterolemic rabbits. Arterioscler Thromb 14:753–759
Cersosimo E, DeFronzo RA (2006) Insulin resistance and endothelial dysfunction: the road map to cardiovascular diseases. Diabetes Metab Res Rev 22:423–436
Chen L, Li P, Wang J et al (2009) Catabolism of nutritionally essential amino acids in developing porcine enterocytes. Amino Acids 37:143–152
Chevalier S, Burgess SC, Malloy CR et al (2006) The greater contribution of gluconeogenesis to glucose production in obesity is related to increased whole-body protein catabolism. Diabetes 55:675–681
Dai ZL, Wu ZL, Yang Y et al (2013) Nitric oxide and energy metabolism in mammals. BioFactors 39:383–391
Dai ZL, Wu ZL, Jia SC et al (2014) Analysis of amino acid composition in proteins of animal tissues and foods as pre-column o-phthaldialdehyde derivatives by HPLC with fluorescence detection. J Chromatogr B 964:116–127
DeFronzo RA (1988) Lilly lecture 1987. The triumvirate: beta-cell, muscle, liver. A collusion responsible for NIDDM. Diabetes 37:667–687
Dillon EL (2013) Nutritionally essential amino acids and metabolic signaling in aging. Amino Acids 45:431–441
Ding Y, Vaziri ND, Coulson R et al (2000) Effects of simulated hyperglycemia, insulin, and glucagon on endothelial nitric oxide synthase expression. Am J Physiol Endocrinol Metab 279:E11–E17
Du M, Islam MM, Lin L et al (1997) Promotion of proliferation of murine BALB/C3T3 fibroblasts mediated by nitric oxide at lower concentrations. Biochem Mol Biol Int 41:625–631
Dudzinski DM, Igarashi J, Greif D et al (2006) The regulation and pharmacology of endothelial nitric oxide synthase. Annu Rev Pharmacol Toxicol 46:235–276
Duncan C, Dougall H, Johnston P et al (1995) Chemical generation of nitric oxide in the mouth from the enterosalivary circulation of dietary nitrate. Nat Med 1:546–551
Felig P, Marliss E, Cahill GF Jr (1969) Plasma amino acid levels and insulin secretion in obesity. N Engl J Med 281:811–816
Felig P, Marliss E, Ohman JL, Cahill GF Jr (1970) Plasma amino acid levels in diabetic ketoacidosis. Diabetes 19:727–729
Fiehn O, Garvey WT, Newman JW et al (2010) Plasma metabolomic profiles reflective of glucose homeostasis in non-diabetic and type 2 diabetic obese African-American women. PLoS One 5:e15234
Förstermann U, Sessa WC (2012) Nitric oxide synthases: regulation and function. Eur Heart J 33:829–837
Fu WJ, Haynes TE, Kohli R et al (2005) Dietary l-arginine supplementation reduces fat mass in Zucker diabetic fatty rats. J Nutr 135:714–721
Fulton D, Gratton JP, McCabe TJ et al (1999) Regulation of endothelium-derived nitric oxide production by the protein kinase Akt. Nature 399:597–601
Garg UC, Hassid A (1990) Nitric oxide-generating vasodilators inhibit mitogenesis and proliferation of BALB/C 3T3 fibroblasts by a cyclic GMP-independent mechanism. Biochem Biophys Res Commun 171:474–479
Garvey WT, Birnbaum MJ (1993) Cellular insulin action and insulin resistance. Baillieres Clin Endocrinol Metab 7:785–873
Harper AE, Miller RH, Block KP (1984) Branched-chain amino acid metabolism. Annu Rev Nutr 4:409–454
Hawkins M, Barzilai N, Chen W et al (1996) Increased hexosamine availability similarly impairs the action of insulin and IGF-1 on glucose disposal. Diabetes 45:1734–1743
Henry RR (2003) Insulin resistance: from predisposing factor to therapeutic target in type 2 diabetes. Clin Ther 25(Suppl B):B47–B63
Hill JO, Peters JC, Catenacci VA et al (2008) International strategies to address obesity. Obes Rev 9(Suppl 1):41–47
Hoang HH, Padgham SV, Meininger CJ (2013) l-Arginine, tetrahydrobiopterin, nitric oxide and diabetes. Curr Opin Clin Nutr Metab Care 16:76–82
Hu G, Qiao Q, Tuomilehto J et al (2004) Prevalence of the metabolic syndrome and its relation to all-cause and cardiovascular mortality in nondiabetic European men and women. Arch Intern Med 164:1066–1076
Huffman KM, Shah SH, Stevens RD et al (2009) Relationships between circulating metabolic intermediates and insulin action in overweight to obese, inactive men and women. Diabetes Care 32:1678–1683
Ishii M, Shimizu S, Nagai T et al (2001) Stimulation of tetrahydrobiopterin synthesis induced by insulin: possible involvement of phosphatidylinositol 3-kinase. Int J Biochem Cell Biol 33:65–73
Jobgen WS, Fried SK, Fu WJ et al (2006) Regulatory role for the arginine-nitric oxide pathway in metabolism of energy substrates. J Nutr Biochem 17:571–588
Jobgen WJ, Meininger CJ, Jobgen SC et al (2009) Dietary l-arginine supplementation reduces white-fat gain and enhances skeletal muscle and brown fat masses in diet-induced obese rats. J Nutr 139:230–237
Kakoki M, Kim HS, Edgell CJ et al (2006) Amino acids as modulators of endothelium-derived nitric oxide. Am J Physiol Renal Physiol 291:F297–F304
Karbach S, Wenzel P, Waisman A, Munzel T, Daiber A (2014) eNOS uncoupling in cardiovascular diseases–the role of oxidative stress and inflammation. Curr Pharm Des 20:3579–3594
Kepka-Lenhart D, Mistry SK, Wu G et al (2000) Arginase I: a limiting factor for nitric oxide and polyamine synthesis by activated macrophages? Am J Physiol Regul Integrative Comp Physiol 279:R2237–R2242
Kuhlencordt PJ, Gyurko R, Han F et al (2001) Accelerated atherosclerosis, aortic aneurysm formation, and ischemic heart disease in apolipoprotein E/endothelial nitric oxide synthase double-knockout mice. Circulation 104:448–454
Laakso M, Edelman SV, Brechtel G et al (1990) Decreased effect of insulin to stimulate skeletal muscle blood flow in obese man. A novel mechanism for insulin resistance. J Clin Invest 85:1844–1852
Laakso M, Edelman SV, Brechtel G et al (1992) Impaired insulin-mediated skeletal muscle blood flow in patients with NIDDM. Diabetes 41:1076–1083
Lahteenvuo J, Rosenzweig A (2012) Effects of aging on angiogenesis. Circ Res 110:1252–1264
Lei J, Feng DY, Zhang YL et al (2012) Nutritional and regulatory role of branched-chain amino acids in lactation. Front Biosci 17:2725–2739
Lei J, Feng DY, Zhang YL et al (2013a) Hormonal regulation of leucine catabolism in mammary epithelial cells. Amino Acids 45:531–541
Lei J, Vodovotz Y, Tzeng E, Billiar TR (2013b) Nitric oxide, a protective molecule in the cardiovascular system. Nitric Oxide 35:175–185
Li H, Meininger CJ, Hawker JR et al (2001) Regulatory role of arginase I and II in nitric oxide, polyamine, and proline syntheses in endothelial cells. Am J Physiol Endocrinol Metab 280:E75–E82
Li H, Meininger CJ, Hawker JR et al (2002) Activities of arginase I and II are limiting for endothelial cell proliferation. Am J Physiol Regul Integr Comp Physiol 282:R64–R69
Li C, Najafi H, Daikhin Y et al (2003) Regulation of leucine-stimulated insulin secretion and glutamine metabolism in isolated rat islets. J Biol Chem 278:2853–2858
Li XL, Bazer FW, Gao H et al (2009a) Amino acids and gaseous signaling. Amino Acids 37:65–78
Li P, Knabe DA, Kim SW et al (2009b) Lactating porcine mammary tissue catabolizes branched-chain amino acids for glutamine and aspartate synthesis. J Nutr 139:1502–1509
Mark AL (2009) Cardiovascular side effects of antiobesity drugs: a yellow flag in the race to safe pharmacotherapy for obesity. Circulation 120:719–721
Mather KJ, Verma S, Anderson TJ (2001) Improved endothelial function with metformin in type 2 diabetes mellitus. J Am Coll Cardiol 37:1344–1350
Meininger CJ, Wu G (2002) Regulation of endothelial cell proliferation by nitric oxide. Methods Enzymol 352:280–295
Meininger CJ, Wu G (2011) Tetrahydrobiopterin: important endothelial mediator independent of endothelial nitric oxide synthase. Hypertension 58:145–147
Meininger CJ, Kelly KA, Hatakeyama K et al (2004) Tetrahydrobiopterin deficiency occurs in both type I and type II diabetes mellitus: role of insulin and GTP-CH. In: Thony B, Blau N (eds) Pterins, Folates, and Neurotransmitters in Molecular Medicine. SPS Verlagsgesellschaft mbh, Heilbronn, pp 83–87
Mels CM, Schutte AE, Schutte R et al (2013) The link between vascular deterioration and branched chain amino acids in a population with high glycated haemoglobin: the SABPA study. Amino Acids 45:1405–1413
Mimura T, Yamada C, Swendseid ME (1968) Influence of dietary protein levels and hydrocortisone administration on the branched-chain amino acid transaminase activity in rat tissues. J Nutr 95:493–497
Morris SJ, Shore AC, Tooke JE (1995) Responses of the skin microcirculation to acetylcholine and sodium nitroprusside in patients with NIDDM. Diabetologia 38:1337–1344
Nagase S, Aoyagi K, Hirayama A et al (1996) Decreased serum antioxidant activity of hemodialysis patients demonstrated by methylguanidine synthesis and microsomal lipid peroxidation. Nephron 74:555–560
Nagase S, Aoyagi K, Hirayama A et al (1997a) Favorable effect of hemodialysis on decreased serum antioxidant activity in hemodialysis patients demonstrated by electron spin resonance. J Am Soc Nephrol 8:1157–1163
Nagase S, Takemura K, Ueda A et al (1997b) A novel nonenzymatic pathway for the generation of nitric oxide by the reaction of hydrogen peroxide and D- or L-arginine. Biochem Biophys Res Commun 233:150–153
Natali A, Baldeweg S, Toschi E et al (2004) Vascular effects of improving metabolic control with metformin or rosiglitazone in type 2 diabetes. Diabetes Care 27:1349–1357
Nathan C, Xie QW (1994) Nitric oxide synthases: roles, tolls, and controls. Cell 78:915–918
Newgard CB, An J, Bain JR et al (2009) A branched-chain amino acid-related metabolic signature that differentiates obese and lean humans and contributes to insulin resistance. Cell Metab 9:311–326
Orellana RA, Suryawan A, Kimball SR et al (2008) Insulin signaling in skeletal muscle and liver of neonatal pigs during endotoxemia. Pediatr Res 64:505–510
Patel MJ, Wypij DM, Rose DA et al (1995) Secretion of cyclic GMP by cultured epithelial and fibroblast cell lines in response to nitric oxide. J Pharmacol Exp Ther 273:16–25
Patti ME, Virkamaki A, Landaker EJ et al (1999) Activation of the hexosamine pathway by glucosamine in vivo induces insulin resistance of early postreceptor insulin signaling events in skeletal muscle. Diabetes 48:1562–1571
Payne WJ, Liu MY, Bursakov SA et al (1997) Microbial and plant metabolism of NO. BioFactors 6:47–52
Pervin S, Singh R, Hernandez E et al (2007) Nitric oxide in physiological concentrations targets the translational machinery to increase the proliferation of human breast cancer cells: involvement of mammalian target of rapamycin/eIF4E pathway. Cancer Res 67:289–299
Prudente S, Morini E, Trischitta V (2009) Insulin signaling regulating genes: effect on T2DM and cardiovascular risk. Nat Rev Endocrinol 5:682–693
Romero MJ, Platt DH, Tawfik HE et al (2008) Diabetes-induced coronary vascular dysfunction involves increased arginase activity. Circ Res 102:95–102
Roy D, Perreault M, Marette A (1998) Insulin stimulation of glucose uptake in skeletal muscles and adipose tissues in vivo is NO dependent. Am J Physiol 274:E692–E699
Rubio AR, Morales-Segura MA (2004) Nitric oxide, an iceberg in cardiovascular physiology: far beyond vessel tone control. Arch Med Res 35:1–11
Santulli G, Totary-Jain H (2013) Tailoring mTOR-based therapy: molecular evidence and clinical challenges. Pharmacogenomics 14:1517–1526
Satoh N, Ogawa Y, Usui T et al (2003) Antiatherogenic effect of pioglitazone in type 2 diabetic patients irrespective of the responsiveness to its antidiabetic effect. Diabetes Care 26:2493–2499
Schachter D, Sang JC (2002) Aortic leucine-to-glutamate pathway: metabolic route and regulation of contractile responses. Am J Physiol Heart Circ Physiol 282:H1135–H1148
Shankar RR, Wu Y, Shen HQ et al (2000) Mice with gene disruption of both endothelial and neuronal nitric oxide synthase exhibit insulin resistance. Diabetes 49:684–687
Shaul PW (2002) Regulation of endothelial nitric oxide synthase: location, location, location. Annu Rev Physiol 64:749–774
She P, Van Horn C, Reid T et al (2007) Obesity-related elevations in plasma leucine are associated with alterations in enzymes involved in branched-chain amino acid metabolism. Am J Physiol Endocrinol Metab 293:E1552–E1563
Steinberg HO, Baron AD (2002) Vascular function, insulin resistance and fatty acids. Diabetologia 45:623–634
Steinberg HO, Brechtel G, Johnson A et al (1994) Insulin-mediated skeletal muscle vasodilation is nitric oxide dependent. A novel action of insulin to increase nitric oxide release. J Clin Invest 94:1172–1179
Stuehr DJ (1997) Structure–function aspects in the nitric oxide synthases. Annu Rev Pharmacol Toxicol 37:339–359
Tai ES, Tan ML, Stevens RD et al (2010) Insulin resistance is associated with a metabolic profile of altered protein metabolism in Chinese and Asian-Indian men. Diabetologia 53:757–767
Tekwe CD, Lei J, Yao K et al (2012) Oral administration of α-ketoglutarate or interferon-τ reduces adiposity in diet-induced obese rats. FASEB J 26(819):45
Tekwe CD, Lei J, Yao K et al (2013) Oral administration of interferon tau enhances oxidation of energy substrates and reduces adiposity in Zucker diabetic fatty rats. BioFactors 39:552–563
Thomae KR, Nakayama DK, Billiar TR et al (1995) The effect of nitric oxide on fetal pulmonary artery smooth muscle growth. J Surg Res 59:337–343
Thomas DD, Ridnour LA, Isenberg JS et al (2008) The chemical biology of nitric oxide: implications in cellular signaling. Free Radic Biol Med 45:18–31
Tischler ME, Goldberg AL (1980) Leucine degradation and release of glutamine and alanine by adipose tissue. J Biol Chem 255:8074–8081
Tremblay F, Krebs M, Dombrowski L et al (2005) Overactivation of S6 kinase 1 as a cause of human insulin resistance during increased amino acid availability. Diabetes 54:2674–2684
Vallance P, Leiper J (2002) Blocking NO synthesis: how, where and why? Nat Rev Drug Discov 1:939–950
Van Gaal LF, Mertens IL, De Block CE (2006) Mechanisms linking obesity with cardiovascular disease. Nature 444:875–880
Vincent MA, Barrett EJ, Lindner JR et al (2003) Inhibiting NOS blocks microvascular recruitment and blunts muscle glucose uptake in response to insulin. Am J Physiol Endocrinol Metab 285:E123–E129
Vincent MA, Clerk LH, Lindner JR et al (2004) Microvascular recruitment is an early insulin effect that regulates skeletal muscle glucose uptake in vivo. Diabetes 53:1418–1423
Wu G (2013) Amino Acids: Biochemistry and Nutrition. CRC Press, Boca Raton
Wu G (2014) Dietary requirements of synthesizable amino acids by animals: a paradigm shift in protein nutrition. J Anim Sci Biotechnol 5:34
Wu G, Meininger CJ (2000) Arginine nutrition and cardiovascular function. J Nutr 130:2626–2629
Wu G, Meininger CJ (2002) Regulation of nitric oxide synthesis by dietary factors. Annu Rev Nutr 22:61–86
Wu G, Meininger CJ (2008) Analysis of citrulline, arginine, and methylarginines using high-performance liquid chromatography. Methods Enzymol 440:177–189
Wu G, Meininger CJ (2009) Nitric oxide and vascular insulin resistance. BioFactors 35:21–27
Wu G, Morris SM Jr (1998) Arginine metabolism: nitric oxide and beyond. Biochem J 336:1–17
Wu G, Majumdar S, Zhang J et al (1994) Insulin stimulates glycolysis and pentose cycle activity in bovine microvascular endothelial cells. Comp Biochem Physiol C 108:179–185
Wu G, Haynes TE, Li H et al (2001a) Glutamine metabolism to glucosamine is necessary for glutamine inhibition of endothelial nitric oxide synthesis. Biochem J 353:245–252
Wu G, Haynes TE, Yan W et al (2001b) Presence of glutamine:fructose-6-phosphate amidotransferase for glucosamine-6-phosphate synthesis in endothelial cells: effects of hyperglycaemia and glutamine. Diabetologia 44:196–202
Wu G, Collins JK, Perkins-Veazie P et al (2007) Dietary supplementation with watermelon pomace juice enhances arginine availability and ameliorates the metabolic syndrome in Zucker diabetic fatty rats. J Nutr 137:2680–2685
Wu ZL, Satterfield MC, Bazer FW et al (2012) Regulation of brown adipose tissue development and white fat reduction by L-arginine. Curr Opin Clin Nutr Metab Care 15:529–538
Wu G, Bazer FW, Dai ZL et al (2014) Amino acid nutrition in animals: protein synthesis and beyond. Annu Rev Anim Biosci 2:387–417
Wullschleger S, Loewith R, Hall MN (2006) TOR signaling in growth and metabolism. Cell 124:471–484
Yang Y, Nie W, Yuan J et al (2010) Genistein activates endothelial nitric oxide synthase in broiler pulmonary arterial endothelial cells by an Akt-dependent mechanism. Exp Mol Med 42:768–776
Yang Y, Meininger C, Kelly K, Wu ZL, Bazer F, Wu G (2013a) l-Leucine inhibits nitric oxide synthesis in endothelial cells. FASEB J 27(1127):3
Yang Y, Wu ZL, Kelly K et al (2013b) N-ethyl-l-glutamine enhances the synthesis of nitric oxide by endothelial cells. Amino Acids 45:605
Yki-Jarvinen H (2003) Insulin resistance and endothelial dysfunction. Best Pract Res Clin Endocrinol Metab 17:411–430
Zecchin HG, Priviero FB, Souza CT et al (2007) Defective insulin and acetylcholine induction of endothelial cell-nitric oxide synthase through insulin receptor substrate/Akt signaling pathway in aorta of obese rats. Diabetes 56:1014–1024
Zeng G, Quon MJ (1996) Insulin-stimulated production of nitric oxide is inhibited by wortmannin. Direct measurement in vascular endothelial cells. J Clin Invest 98:894–898
Zhang Z, Naughton D, Winyard PG et al (1998) Generation of nitric oxide by a nitrite reductase activity of xanthine oxidase: a potential pathway for nitric oxide formation in the absence of nitric oxide synthase activity. Biochem Biophys Res Commun 249:767–772
Zhang L, Vincent MA, Richards SM et al (2004) Insulin sensitivity of muscle capillary recruitment in vivo. Diabetes 53:447–453
Zweier JL, Wang P, Samouilov A et al (1995) Enzyme-independent formation of nitric oxide in biological tissues. Nat Med 1:804–809
Acknowledgments
Work in our laboratories was supported by the National Basic Research Program of China (Grant 2013CB127302), the National Natural Science Foundation of China (No. 31172217, 31272451 and 31272450), American Heart Association (10GRNT4480020), and Texas A&M AgriLife Research (H-8200). Ying Yang was supported by a Fellowship from the Chinese Scholarship Council and National Natural Science Foundation of China (No. 31172225 and 31372327).
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Handling Editor: F. Galli.
Rights and permissions
About this article
Cite this article
Yang, Y., Wu, Z., Meininger, C.J. et al. l-Leucine and NO-mediated cardiovascular function. Amino Acids 47, 435–447 (2015). https://doi.org/10.1007/s00726-014-1904-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-014-1904-y