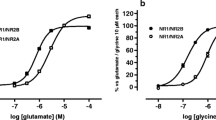
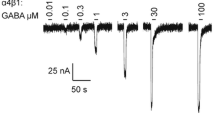
Abstract
GABAA receptors are members of the ligand-gated ion channel superfamily that mediate inhibitory neurotransmission in the central nervous system. They are thought to be composed of 2 alpha (α), 2 beta (β) subunits and one other such as a gamma (γ) or delta (δ) subunit. The potency of GABA is influenced by the subunit composition. However, there are no reported systematic studies that evaluate GABA potency on a comprehensive number of subunit combinations expressed in Xenopus oocytes, despite the wide use of this heterologous expression system in structure–function studies and drug discovery. Thus, the aim of this study was to conduct a systematic characterization of the potency of GABA at 43 human recombinant GABAA receptor combinations expressed in Xenopus oocytes using the two-electrode voltage clamp technique. The results show that the α-subunits and to a lesser extent, the β-subunits influence GABA potency. Of the binary and ternary combinations with and without the γ2L subunit, the α6/γ2L-containing receptors were the most sensitive to GABA, while the β2- or β3-subunit conferred higher sensitivity to GABA than receptors containing the β1-subunit with the exception of the α2β1γ2L and α6β1γ2L subtypes. Of the δ-subunit containing GABAA receptors, α4/δ-containing GABAA receptors displayed highest GABA sensitivity, with mid-nanomolar concentrations activating α4β1δ and α4β3δ receptors. At α4β2δ, GABA had low micromolar activity.





Similar content being viewed by others
References
Absalom N, Eghorn LF, Villumsen IS, Karim N, Bay T, Olsen JV, Knudsen GM, Brauner-Osborne H, Frolund B, Clausen RP, Chebib M, Wellendorph P (2012) Alpha4betadelta GABA(A) receptors are high-affinity targets for gamma-hydroxybutyric acid (GHB). Proc Natl Acad Sci USA 109(33):13404–13409
Atack J, Wafford KA, Tye SJ, Cook SM, Sohal B, Pike A, Sur C, Melillo D, Bristow L, Bromidge F, Ragan I, Kerby J, Street L, Carling R, Castro JL, Whiting P, Dawson GR, McKernan RM (2005) The benzodiazepine binding site of GABAA receptors as a target for the development of novel anxiolytics. Expert Opin Investig Drugs 14:601–618
Barnard EA, Skolnick P, Olsen RW, Mohler H, Sieghart W, Biggio G, Braestrup C, Bateson AN, Langer SZ (1998) International Union of Pharmacology. XV. Subtypes of gamma-aminobutyric acidA receptors: classification on the basis of subunit structure and receptor function. Pharmacol Rev 50(2):291–313
Belelli D, Harrison NL, Maguire J, Macdonald RL, Walker MC, Cope DW (2009) Extrasynaptic GABAA receptors: form, pharmacology, and function. J Neurosci 29(41):12757–12763
Boileau AJ, Baur R, Sharkey LM, Sigel E, Czajkowski C (2002) The relative amount of cRNA coding for gamma2 subunits affects stimulation by benzodiazepines in GABA(A) receptors expressed in Xenopus oocytes. Neuropharmacology 43(4):695–700
Bonnert TP, McKernan RM, Farrar S, Bourdelles Bl, Heavens RP, Smith DW, Hewson L, Rigby MR, Sirinathsinghji DJS, Brown N, Wafford KA, Whiting PJ (1999) θ, a Novel γ-aminobutyric acid type A receptor subunit. Proc Natl Acad Sci 96(17):9891–9896
Brandon NJ, Delmas P, Kittler JT, McDonald BJ, Sieghart W, Brown DA, Smart TG, Moss SJ (2000) GABAA receptor phosphorylation and functional modulation in cortical neurons by a protein kinase C-dependent pathway. J Biol Chem 275(49):38856–38862
Ducic I, Caruncho HJ, Wei Jian Z, Vicini S, Costa E (1995) GABA gating of Cl− channels in recombinant GABAA receptors. J Pharm Exp Ther 272(1):438–445
Hadley SH, Amin J (2007) Rat alpha6beta2delta GABAA receptors exhibit two distinct and separable agonist affinities. J Physiol 581(Pt 3):1001–1018
Hall BJ, Chebib M, Hanrahan JR, Johnston GAR (2005) 6-Methylflavanone, a more efficacious positive allosteric modulator of [gamma]-aminobutyric acid (GABA) action at human recombinant [alpha]2[beta]2[gamma]2L than at [alpha]1[beta]2[gamma]2L and [alpha]1[beta]2 GABAA receptors expressed in Xenopus oocytes. Eur J Pharmacol 512(2–3):97–104
Harpsoe K, Ahring PK, Christensen JK, Jensen ML, Peters D, Balle T (2011) Unraveling the high- and low-sensitivity agonist responses of nicotinic acetylcholine receptors. J Neurosci 31(30):10759–10766
Hosie AM, Dunne EL, Harvey RJ, Smart TG (2003) Zinc-mediated inhibition of GABA(A) receptors: discrete binding sites underlie subtype specificity. Nat Neurosci 6(4):362–369
Hutcheon B, Fritschy JM, Poulter MO (2004) Organization of GABA receptor alpha-subunit clustering in the developing rat neocortex and hippocampus. Eur J Neurosci 19(9):2475–2487
Karim N, Gavande N, Wellendorph P, Johnston GAR, Hanrahan JR, Chebib M (2011) 3-Hydroxy-2′-methoxy-6-methylflavone: a potent anxiolytic with a unique selectivity profile at GABA(A) receptor subtypes. Biochem Pharmacol 82(12):1971–1983
Karim N, Wellendorph P, Absalom N, Bang LH, Jensen ML, Hansen MM, Lee HJ, Johnston GAR, Hanrahan JR, Chebib M (2012) Low nanomolar GABA effects at extrasynaptic alpha4beta1/beta3delta GABA(A) receptor subtypes indicate a different binding mode for GABA at these receptors. Biochem Pharmacol 84(4):549–557
Kaur KH, Baur R, Sigel E (2009) Unanticipated structural and functional properties of delta-subunit-containing GABAA receptors. J Biol Chem 284(12):7889–7896
Kleingoor C, Wieland HA, Korpi ER, Seeburg PH, Kettenmann H (1993) Current potentiation by diazepam but not GABA sensitivity is determined by a single histidine residue. NeuroReport 4(2):187–190
Knoflach F, Benke D, Wang Y, Scheurer L, Luddens H, Hamilton BJ, Carter DB, Mohler H, Benson JA (1996) Pharmacological modulation of the diazepam-insensitive recombinant γ-aminobutyric acid(A) receptors α4β2γ2 and α6β2γ2. Mol Pharmacol 50(5):1253–1261
Korpi ER, Mihalek RM, Sinkkonen ST, Hauer B, Hevers W, Homanics GE, Sieghart W, Luddens H (2002) Altered receptor subtypes in the forebrain of GABA(A) receptor delta subunit-deficient mice: recruitment of gamma 2 subunits. Neuroscience 109(4):733–743
Meera P, Wallner M, Otis TS (2011) Molecular basis for the high THIP/gaboxadol sensitivity of extrasynaptic GABA(A) receptors. J Neurophysiol 106(4):2057–2064
Mortensen M, Patel B, Smart TG (2012) GABA potency at GABA(A) receptors found in synaptic and extrasynaptic zones. Front Cell Neurosci 6:1–10
Nusser Z, Sieghart W, Somogyi P (1998) Segregation of different GABAA receptors to synaptic and extrasynaptic membranes of cerebellar granule cells. J Neurosci 18(5):1693–1703
Olsen RW, Sieghart W (2008) International Union of Pharmacology. LXX. Subtypes of gamma-aminobutyric acid(A) receptors: classification on the basis of subunit composition, pharmacology, and function. Update. Pharmacol Rev 60(3):243–260
Palma E, Trettel F, Fucile S, Renzi M, Miledi R, Eusebi F (2003) Microtransplantation of membranes from cultured cells to Xenopus oocytes: a method to study neurotransmitter receptors embedded in native lipids. Proc Natl Acad Sci USA 100(5):2896–2900
Pirker S, Schwarzer C, Wieselthaler A, Sieghart W, Sperk G (2000) GABAA receptors: immunocytochemical distribution of 13 subunits in the adult rat brain. Neuroscience 101(4):815–850
Rudolph U, Crestani F, Mohler H (2001) GABAA receptor subtypes: dissecting their pharmacological functions. Trends Pharmacol Sci 22:188–194
Sieghart W (2006) Structure, pharmacology, and function of GABAA receptor subtypes. Adv Pharmacol 54:231–263
Sigel E, Kaur KH, Luscher BP, Baur R (2009) Use of concatamers to study GABAA receptor architecture and function: application to delta-subunit-containing receptors and possible pitfalls. Biochem Soc Trans 37(Pt 6):1338–1342
Sinkkonen ST, Hanna MC, Kirkness EF, Korpi ER (2000) GABA(A) receptor ε and θ subunits display unusual structural variation between species and are enriched in the rat locus ceruleus. J Neurosci 20(10):3588–3595
Smart TG, Moss SJ, Xie X, Huganir RL (1991) GABAA receptors are differentially sensitive to zinc: dependence on subunit composition. Br J Pharmacol 103(4):1837–1839
Sperk G, Schwarzer C, Tsunashima K, Fuchs K, Sieghart W (1997) GABA(A) receptor subunits in the rat hippocampus I: immunocytochemical distribution of 13 subunits. Neuroscience 80(4):987–1000
Storustovu SI, Ebert B (2006) Pharmacological characterization of agonists at delta-containing GABAA receptors: functional selectivity for extrasynaptic receptors is dependent on the absence of gamma2. J Pharmacol Exp Ther 316(3):1351–1359
Wagoner KR, Czajkowski C (2010) Stoichiometry of expressed alpha(4)beta(2)delta gamma-aminobutyric acid type A receptors depends on the ratio of subunit cDNA transfected. J Biol Chem 285(19):14187–14194
Wei W, Zhang N, Peng Z, Houser CR, Mody I (2003) Perisynaptic localization of delta subunit-containing GABA(A) receptors and their activation by GABA spillover in the mouse dentate gyrus. J Neurosci 23(33):10650–10661
Wisden W, Laurie DJ, Monyer H, Seeburg PH (1992) The distribution of 13 GABAA receptor subunit mRNAs in the rat brain: telencephalon, diencephalon, mesencephalon. J Neurosci 12(3):1040–1062
Wu J, Liu Q, Yu K, Hu J, Kuo YP, Segerberg M, St John PA, Lukas RJ (2006) Roles of nicotinic acetylcholine receptor beta subunits in function of human alpha4-containing nicotinic receptors. J Physiol 576(Pt 1):103–118
Acknowledgments
We are grateful to Dr. Paul Whiting (Merck, Sharpe and Dohme Research Laboratories, Harlow, UK) and Dr. Bjarke Ebert (H. Lundbeck A/S Valby, Denmark) for the gift of cDNA for GABAA subunits. We are very grateful to the Department of Pharmacology, The University of Sydney, for managing and maintaining the Xenopus laevis colony. MC acknowledges travel support from the Drug Research Academy, the Faculty of Pharmaceutical Sciences, The University of Copenhagen, Denmark, and the Australian Academy of Sciences. PW acknowledges support from the Alfred Benzon Foundation, Denmark. NK acknowledges The University of Malakand, Pakistan (Faculty Development Programme Scholarship) and the John Lamberton Scholarship. The funding sources solely provided financial support and were not involved in any part of the conduct of the research.
Conflicts of interest
The authors have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Karim, N., Wellendorph, P., Absalom, N. et al. Potency of GABA at human recombinant GABAA receptors expressed in Xenopus oocytes: a mini review. Amino Acids 44, 1139–1149 (2013). https://doi.org/10.1007/s00726-012-1456-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-012-1456-y