Abstract
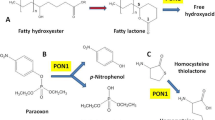
Paraoxonase 1 (PON1), a component of high-density lipoprotein (HDL), is a calcium-dependent multifunctional enzyme that connects metabolisms of lipoproteins and homocysteine (Hcy). Both PON1 and Hcy have been implicated in human diseases, including atherosclerosis and neurodegeneration. The involvement of Hcy in disease could be mediated through its interactions with PON1. Due to its ability to reduce oxidative stress, PON1 contributes to atheroprotective functions of HDL in mice and humans. Although PON1 has the ability to hydrolyze a variety of substrates, only one of them—Hcy-thiolactone—is known to occur naturally. In humans and mice, Hcy-thiolactonase activity of PON1 protects against N-homocysteinylation, which is detrimental to protein structure and function. PON1 also protects against neurotoxicity associated with hyperhomocysteinemia in mouse models. The links between PON1 and Hcy in relation to pathological states such as coronary artery disease, stroke, diabetic mellitus, kidney failure and Alzheimer’s disease that emerge from recent studies are the topics of this review.

Similar content being viewed by others
References
Adkins S, Gan KN et al (1993) Molecular basis for the polymorphic forms of human serum paraoxonase/arylesterase: glutamine or arginine at position 191, for the respective A or B allozymes. Am J Hum Genet 52(3):598–608
Amine K, Atouk A et al (2011) Paraoxonase-1 (PON1) activity in patients with coronary artery diseases and in diabetic patients. Ann Biol Clin (Paris) 69(6):671–677
Anderson J, Muhlestein J et al (2000) Plasma homocysteine predicts mortality independently of traditional risk factors and C-reactive protein in patients with angiographically defined coronary artery disease. Circulation 102(11):1227–1232
Angayarkanni N, Barathi S et al (2008) Serum PON1 arylesterase activity in relation to hyperhomocysteinaemia and oxidative stress in young adult central retinal venous occlusion patients. Eye (Lond) 22(7):969–974
Atamer A, Kocyigit Y et al (2008) Effect of oxidative stress on antioxidant enzyme activities, homocysteine and lipoproteins in chronic kidney disease. J Nephrol 21(6):924–930
Ates O, Azizi S et al (2009) Decreased serum paraoxonase 1 activity and increased serum homocysteine and malondialdehyde levels in age-related macular degeneration. Tohoku J Exp Med 217(1):17–22
Aviram M, Billecke S et al (1998) Paraoxonase active site required for protection against LDL oxidation involves its free sulfhydryl group and is different from that required for its arylesterase/paraoxonase activities: selective action of human paraoxonase allozymes Q and R. Arterioscler Thromb Vasc Biol 18(10):1617–1624
Aydin M, Gokkusu C et al (2009) Association of genetic variants in Methylenetetrahydrofolate Reductase and Paraoxonase-1 genes with homocysteine, folate and vitamin B12 in coronary artery disease. Mol Cell Biochem 325(1–2):199–208
Barathi S, Angayarkanni N et al (2010) Homocysteinethiolactone and paraoxonase: novel markers of diabetic retinopathy. Diabetes Care 33(9):2031–2037
Bayrak A, Bayrak T et al (2011) Serum PON-1 activity but not Q192R polymorphism is related to the extent of atherosclerosis. J Atheroscler Thromb 19(4):376–384
Bayrak T, Bayrak A et al (2010) Purification and kinetic properties of rabbit liver paraoxonase 1. J Chromatogr B Analyt Technol Biomed Life Sci 878(21):1791–1795
Bayrak A, Bayrak T et al (2011) Differential hydrolysis of homocysteine thiolactone by purified human serum (192)Q and (192)R PON1 isoenzymes. J Chromatogr B Analyt Technol Biomed Life Sci 879(1):49–55
Beltowski J, Wojcicka G et al (2010) Modulation of paraoxonase 1 and protein N-homocysteinylation by leptin and the synthetic liver X receptor agonist T0901317 in the rat. J Endocrinol 204(2):191–198
Ben-David M, Elias M et al (2012) Catalytic versatility and backups in enzyme active sites: the case of serum paraoxonase 1. J Mol Biol 418(3–4):181–196
Bhattacharyya T, Nicholls SJ et al (2008) Relationship of paraoxonase 1 (PON1) gene polymorphisms and functional activity with systemic oxidative stress and cardiovascular risk. JAMA 299(11):1265–1276
Blatter MC, James RW et al (1993) Identification of a distinct human high-density lipoprotein subspecies defined by a lipoprotein-associated protein, K-45. Identity of K-45 with paraoxonase. Eur J Biochem 211(3):871–879
Borowczyk K, Shih DM et al (2012a) Metabolism and neurotoxicity of homocysteine thiolactone in mice: evidence for a protective role of paraoxonase 1. J Alzheimers Dis 30(2):225–231
Borowczyk K, Tisończyk J et al (2012b) Metabolism and neurotoxicity of homocysteine thiolactone in mice: protective role of bleomycin hydrolase. Amino Acids. doi:10.1007/s00726-011-1207-5
Bouman HJ, Schömig E et al (2011) Paraoxonase-1 is a major determinant of clopidogrel efficacy. Nat Med 17(1):110–116
Brophy VH, Jarvik GP et al (2000) Analysis of paraoxonase (PON1) L55M status requires both genotype and phenotype. Pharmacogenetics 10(5):453–460
Camps J, Joven J et al (2011) Paraoxonase-1 and clopidogrel efficacy. Nat Med 17(9):1041–1042
Can Demirdöğen B, Türkanoğlu A et al (2008) Paraoxonase/arylesterase ratio, PON1 192Q/R polymorphism and PON1 status are associated with increased risk of ischemic stroke. Clin Biochem 41(1–2):1–9
Cao H, Girard-Globa A et al (1999) Paraoxonase protection of LDL against peroxidation is independent of its esterase activity towards paraoxon and is unaffected by the Q– > R genetic polymorphism. J Lipid Res 40(1):133–139
Chang HH, Lin DP et al (2011) Intravitreal homocysteine-thiolactone injection leads to the degeneration of multiple retinal cells, including photoreceptors. Mol Vis 17:1946–1956
Chwatko G, Jakubowski H (2005a) The determination of homocysteine-thiolactone in human plasma. Anal Biochem 337(2):271–277
Chwatko G, Jakubowski H (2005b) Urinary excretion of homocysteine-thiolactone in humans. Clin Chem 51(2):408–415
Chwatko G, Boers GHJ et al (2007) Mutations in methylenetetrahydrofolate reductase or cystathionine beta-syntase gene, or a high-methionine diet, increase homocysteine thiolactone levels in humans and mice. Faseb J 21(8):1707–1713
Coombes RH, Crow JA et al (2011) Relationship of human paraoxonase-1 serum activity and genotype with atherosclerosis in individuals from the Deep South. Pharmacogenet Genomics 21(12):867–875
Costa LG, Vitalone A et al (2005) Modulation of paraoxonase (PON1) activity. Biochem Pharmacol 69(4):541–550
Costa LG, Giordano G et al (2011) Pharmacological and dietary modulators of paraoxonase 1 (PON1) activity and expression: the hunt goes on. Biochem Pharmacol 81(3):337–344
Dansette PM, Rosi J et al (2011) Paraoxonase-1 and clopidogrel efficacy. Nat Med 17(9):1040–1041
Dansette PM, Rosi J et al (2012) Cytochromes P450 catalyze both steps of the major pathway of clopidogrel bioactivation, whereas paraoxonase catalyzes the formation of a minor thiol metabolite isomer. Chem Res Toxicol 25(2):348–356
Davies HG, Richter RJ et al (1996) The effect of the human serum paraoxonase polymorphism is reversed with diazoxon, soman and sarin. Nat Genet 14(3):334–336
Demirdöğen BC, Demirkaya S et al (2009) Analysis of paraoxonase 1 (PON1) genetic polymorphisms and activities as risk factors for ischemic stroke in Turkish population. Cell Biochem Funct 27(8):558–567
Domagala TB, Lacinski M et al (2006) The correlation of homocysteine-thiolactonase activity of the paraoxonase (PON1) protein with coronary heart disease status. Cell Mol Biol 52(5):4–10
Draganov DI, Teiber JF et al (2005) Human paraoxonases (PON1, PON2, and PON3) are lactonases with overlapping and distinct substrate specificities. J Lipid Res 46(6):1239–1247
Dudman NP, Hicks C et al (1991) Homocysteine thiolactone disposal by human arterial endothelial cells and serum in vitro. Arterioscler Thromb 11(3):663–670
Dunet V, Ruiz J et al (2011) Effects of paraoxonase activity and gene polymorphism on coronary vasomotion. EJNMMI Res 1(1):27
Fan AZ, Yesupriya A et al (2010) Gene polymorphisms in association with emerging cardiovascular risk markers in adult women. BMC Med Genet 11:6
Ferretti G, Bacchetti T et al (2003) Effect of homocysteinylation on human high-density lipoproteins: a correlation with paraoxonase activity. Metabolism 52(2):146–151
Ferretti G, Bacchetti T et al (2010) Effect of homocysteinylation on high density lipoprotein physico-chemical properties. Chem Phys Lipids 163(2):228–235
Furlong CE, Holland N et al (2006) PON1 status of farm worker mothers and children as a predictor of organophosphate sensitivity. Pharmacogenet Genomics 16(3):183–190
Gan KN, Smolen A et al (1991) Purification of human serum paraoxonase/arylesterase. Evidence for one esterase catalyzing both activities. Drug Metab Dispos 19(1):100–106
Garin MC, James RW et al (1997) Paraoxonase polymorphism Met-Leu54 is associated with modified serum concentrations of the enzyme. A possible link between the paraoxonase gene and increased risk of cardiovascular disease in diabetes. J Clin Invest 99(1):62–66
Ghorbanihaghjo A, Javadzadeh A et al (2008) Lipoprotein(a), homocysteine, and retinal arteriosclerosis. Mol Vis 14:1692–1697
Giusti B, Saracini C et al (2008) Genetic analysis of 56 polymorphisms in 17 genes involved in methionine metabolism in patients with abdominal aortic aneurysm. J Med Genet 45(11):721–730
Giusti B, Saracini C et al (2010) Early-onset ischaemic stroke: analysis of 58 polymorphisms in 17 genes involved in methionine metabolism. Thromb Haemost 104(2):231–242
Glowacki R, Jakubowski H (2004) Cross-talk between Cys(34) and lysine residues in human serum albumin revealed by N-homocysteinylation. J Biol Chem 279(12):10864–10871
Gong M, Garige M et al (2009) Quercetin up-regulates paraoxonase 1 gene expression with concomitant protection against LDL oxidation. Biochem Biophys Res Commun 379(4):1001–1004
Guéant-Rodriguez RM, Spada R et al (2011) Homocysteine is a determinant of ApoA-I and both are associated with ankle brachial index, in an ambulatory elderly population. Atherosclerosis 214(2):480–485
Gugliucci A, Kinugasa E et al (2011) Serum paraoxonase 1 (PON1) lactonase activity is lower in end-stage renal disease patients than in healthy control subjects and increases after hemodialysis. Clin Chem Lab Med 49(1):61–67
Gupta N, Binukumar BK et al (2011a) Serum paraoxonase-1 (PON1) activities (PONase/AREase) and polymorphisms in patients with type 2 diabetes mellitus in a North-West Indian population. Gene 487(1):88–95
Gupta N, Singh S et al (2011b) Paraoxonase 1 (PON1) polymorphisms, haplotypes and activity in predicting cad risk in North-West Indian Punjabis. PLoS ONE 6(5):e17805
Harel M, Aharoni A et al (2004) Structure and evolution of the serum paraoxonase family of detoxifying and anti-atherosclerotic enzymes. Nat Struct Mol Biol 11(5):412–419
Hassett C, Richter RJ et al (1991) Characterization of cDNA clones encoding rabbit and human serum paraoxonase: the mature protein retains its signal sequence. Biochemistry 30(42):10141–10149
Holven KB, Aukrust P et al (2008) The antiatherogenic function of HDL is impaired in hyperhomocysteinemic subjects. J Nutr 138(11):2070–2075
Humbert R, Adler DA et al (1993) The molecular basis of the human serum paraoxonase activity polymorphism. Nat Genet 3(1):73–76
Ikeda Y, Suehiro T et al (2007) Human serum paraoxonase concentration predicts cardiovascular mortality in hemodialysis patients. Clin Nephrol 67(6):358–365
Isbilen E, Yilmaz H et al (2009) Association of paraoxonase 55 and 192 gene polymorphisms on serum homocysteine concentrations in preeclampsia. Folia Biol (Praha) 55(2):35–40
Ishimine N, Usami Y et al (2010) Identification of N-homocysteinylated apolipoprotein AI in normal human serum. Ann Clin Biochem 47(Pt 5):453–459
Itahara T, Suehiro T et al (2000) Serum paraoxonase and arylesterase activities in hemodialysis patients. J Atheroscler Thromb 7(3):152–158
Jakubowski H (1997) Metabolism of homocysteine thiolactone in human cell cultures—possible mechanism for pathological consequences of elevated homocysteine levels. J Biol Chem 272(3):1935–1942
Jakubowski H (1999) Protein homocysteinylation: possible mechanism underlying pathological consequences of elevated homocysteine levels. Faseb J 13(15):2277–2283
Jakubowski H (2000a) Calcium-dependent human serum homocysteine thiolactone hydrolase—a protective mechanism against protein s-homocysteinylation. J Biol Chem 275(6):3957–3962
Jakubowski H (2000b) Homocysteine thiolactone: metabolic origin and protein homocysteinylation in humans. J Nutr 130(2):377S–381S
Jakubowski, H. (2008). Paraoxonase 1 (PON1), a junction between the metabolism of homocysteine and lipids. Paraoxonases Their Role Dis Dev Xenobiotic Metabol 6:87–102
Jakubowski H (2010) The Role of Paraoxonase 1 in the Detoxification of Homocysteine Thiolactone. Paraoxonases Inflamm Infection Toxicol 660:113–127
Jakubowski H (2011) Quality control in tRNA charging—editing of homocysteine. Acta Biochim Pol 58(2):149–163
Jakubowski H (2012) Quality control in tRNA charging. Wiley Interdiscip Rev RNA 3(3):295–310
Jakubowski H, Zhang L et al (2000) Homocysteine thiolactone and protein homocysteinylation in human endothelial cells—implications for atherosclerosis. Circ Res 87(1):45–51
Jakubowski H, Ambrosius WT et al (2001) Genetic determinants of homocysteine thiolactonase activity in humans: implications for atherosclerosis. FEBS Lett 491(1–2):35–39
Jakubowski H, Boers GHJ et al (2008) Mutations in cystathionine beta-synthase or methylenetetrahydrofolate reductase gene increase N-homocysteinylated protein levels in humans. Faseb J 22(12):4071–4076
Jakubowski H, Perla-Kajan J et al (2009) Genetic or nutritional disorders in homocysteine or folate metabolism increase protein N-homocysteinylation in mice. Faseb J 23(6):1721–1727
Javadzadeh A, Ghorbanihaghjo A et al (2010) Plasma oxidized LDL and thiol-containing molecules in patients with exudative age-related macular degeneration. Mol Vis 16:2578–2584
Javadzadeh A, Ghorbanihaghjo A et al (2012) Serum paraoxonase phenotype distribution in exudative age-related macular degeneration and its relationship to homocysteine and oxidized low-density lipoprotein. Retina 32(4):658–666
Kelso GJ, Stuart WD et al (1994) Apolipoprotein J is associated with paraoxonase in human plasma. Biochemistry 33(3):832–839
Khersonsky O, Tawfik DS (2005) Structure-reactivity studies of serum paraoxonase PON1 suggest that its native activity is lactonase. Biochemistry 44(16):6371–6382
Kimak E, Hałabiś M et al (2011) Association between moderately oxidized low-density lipoprotein and high-density lipoprotein particle subclass distribution in hemodialyzed and post-renal transplant patients. J Zhejiang Univ Sci B 12(5):365–371
Kosaka T, Yamaguchi M et al (2005) Investigation of the relationship between atherosclerosis and paraoxonase or homocysteine thiolactonase activity in patients with type 2 diabetes mellitus using a commercially available assay. Clin Chim Acta 359(1–2):156–162
Koubaa N, Nakbi A et al (2009) Association of homocysteine thiolactonase activity and PON1 polymorphisms with the severity of acute coronary syndrome. Clin Biochem 42(9):771–776
Kreutz RP, Nystrom P et al (2012) Influence of paraoxonase-1 Q192R and cytochrome P450 2C19 polymorphisms on clopidogrel response. Clin Pharmacol 4:13–20
Lacinski M, Skorupski W et al (2004) Determinants of homocysteine-thiolactonase activity of the paraoxonase-1 (PON1) protein in humans. Cell Mol Biol (Noisy-le-grand) 50(8):885–893
Lacinski M, Skorupski W et al (2004) Determinants of homocysteine-thiolactonase activity of the paraoxonase-1 (PON1) protein in humans. Cell Mol Biol 50(8):885–893
Lakshman R, Garige M et al (2009) Is alcohol beneficial or harmful for cardioprotection? Genes Nutr 5(2):111–120
Leviev I, Negro F et al (1997) Two alleles of the human paraoxonase gene produce different amounts of mRNA. An explanation for differences in serum concentrations of paraoxonase associated with the (Leu-Met54) polymorphism. Arterioscler Thromb Vasc Biol 17(11):2935–2939
Leviev I, Deakin S et al (2001) Decreased stability of the M54 isoform of paraoxonase as a contributory factor to variations in human serum paraoxonase concentrations. J Lipid Res 42(4):528–535
Lewis JP, Shuldiner AR (2012) Paraoxonase 1 Q192R variant and clopidogrel efficacy: fact or fiction? Circ Cardiovasc Genet 5(2):153–155
Liao D, Tan H et al (2006) Hyperhomocysteinemia decreases circulating high-density lipoprotein by inhibiting apolipoprotein A-I Protein synthesis and enhancing HDL cholesterol clearance. Circ Res 99(6):598–606
Liu G, Nellaiappan K et al (1997) Irreversible inhibition of lysyl oxidase by homocysteine thiolactone and its selenium and oxygen analogues. Implications for homocystinuria. J Biol Chem 272(51):32370–32377
Mackness MI, Arrol S et al (1993) Protection of low-density lipoprotein against oxidative modification by high-density lipoprotein associated paraoxonase. Atherosclerosis 104(1–2):129–135
Mackness B, Mackness MI et al (1998) Serum paraoxonase (PON1) 55 and 192 polymorphism and paraoxonase activity and concentration in non-insulin dependent diabetes mellitus. Atherosclerosis 139(2):341–349
Mackness B, Davies GK et al (2001) Paraoxonase status in coronary heart disease: are activity and concentration more important than genotype? Arterioscler Thromb Vasc Biol 21(9):1451–1457
Man BL, Baum L et al (2010) Genetic polymorphisms of Chinese patients with ischemic stroke and concurrent stenoses of extracranial and intracranial vessels. J Clin Neurosci 17(10):1244–1247
Marathe GK, Zimmerman GA et al (2003) Platelet-activating factor acetylhydrolase, and not paraoxonase-1, is the oxidized phospholipid hydrolase of high density lipoprotein particles. J Biol Chem 278(6):3937–3947
Marsillach J, Mackness B et al (2008) Immunohistochemical analysis of paraoxonases-1, 2, and 3 expression in normal mouse tissues. Free Radic Biol Med 45(2):146–157
Mikael LG, Genest J et al (2006) Elevated homocysteine reduces apolipoprotein A-I expression in hyperhomocysteinemic mice and in males with coronary artery disease. Circ Res 98(4):564–571
Mu X, Yu N et al (2012) Evaluation of a new substrate for measurement of serum PON arylesterase activity. Talanta 88:711–716
Oda MN, Bielicki JK et al (2002) Paraoxonase 1 overexpression in mice and its effect on high-density lipoproteins. Biochem Biophys Res Commun 290(3):921–927
Ohmori T, Yano Y et al (2012) Lack of association between serum paraoxonase-1 activity and residual platelet aggregation during dual anti-platelet therapy. Thromb Res 129(4):e36–e40
Perla J, Undas A et al (2004) Purification of antibodies against N-homocysteinylated proteins by affinity chromatography on N omega-homocysteinyl-aminohexyl-Agarose. J Chromatograph B-Anal Technol Biomed Life Sci 807(2):257–261
Perla-Kaján J, Jakubowski H (2010) Paraoxonase 1 protects against protein N-homocysteinylation in humans. FASEB J 24(3):931–936
Perla-Kajan J, Stanger O et al (2008) Immunohistochernical detection of N-homocysteinylated proteins in humans and mice. Biomed Pharmacother 62(7):473–479
Perna AF, Ingrosso D et al (2004) Homocysteine metabolism in renal failure. Curr Opin Clin Nutr Metab Care 7(1):53–57
Perna AF, Satta E et al (2006) Increased plasma protein homocysteinylation in hemodialysis patients. Kidney Int 69(5):869–876
Qujeq D, Omran TS et al (2001) Correlation between total homocysteine, low-density lipoprotein cholesterol and high-density lipoprotein cholesterol in the serum of patients with myocardial infarction. Clin Biochem 34(2):97–101
Rajković MG, Rumora L et al (2010) Effect of non-genetic factors on paraoxonase 1 activity in patients undergoing hemodialysis. Clin Biochem 43(18):1375–1380
Reddy ST, Wadleigh DJ et al (2001) Human paraoxonase-3 is an HDL-associated enzyme with biological activity similar to paraoxonase-1 protein but is not regulated by oxidized lipids. Arterioscler Thromb Vasc Biol 21(4):542–547
Richter RJ, Jarvik GP et al (2010) Paraoxonase 1 status as a risk factor for disease or exposure. Adv Exp Med Biol 660:29–35
Rozenberg O, Shih DM et al (2005) Paraoxonase 1 (PON1) attenuates macrophage oxidative status: studies in PON1 transfected cells and in PON1 transgenic mice. Atherosclerosis 181(1):9–18
Rozenberg O, Shiner M et al (2008) Paraoxonase 1 (PON1) attenuates diabetes development in mice through its antioxidative properties. Free Radic Biol Med 44(11):1951–1959
Sauls DL, Lockhart E et al (2006) Modification of fibrinogen by homocysteine thiolactone increases resistance to fibrinolysis: a potential mechanism of the thrombotic tendency in hyperhomocysteinemia. Biochemistry 45(8):2480–2487
She ZG, Zheng W et al (2009) Human paraoxonase gene cluster transgenic overexpression represses atherogenesis and promotes atherosclerotic plaque stability in ApoE-null mice. Circ Res 104(10):1160–1168
Shih DM, Gu L et al (1998) Mice lacking serum paraoxonase are susceptible to organophosphate toxicity and atherosclerosis. Nature 394(6690):284–287
Shih DM, Xia YR et al (2000) Combined serum paraoxonase knockout/apolipoprotein E knockout mice exhibit increased lipoprotein oxidation and atherosclerosis. J Biol Chem 275(23):17527–17535
Shih DM, Xia YR et al (2007) Decreased obesity and atherosclerosis in human paraoxonase 3 transgenic mice. Circ Res 100(8):1200–1207
Shin BS, Oh SY et al (2008) The paraoxonase gene polymorphism in stroke patients and lipid profile. Acta Neurol Scand 117(4):237–243
Sodi A, Giambene B et al (2008) Atherosclerotic and thrombophilic risk factors in patients with recurrent central retinal vein occlusion. Eur J Ophthalmol 18(2):233–238
Suehiro T, Ikeda Y et al (2002) Serum paraoxonase (PON1) concentration in patients undergoing hemodialysis. J Atheroscler Thromb 9(3):133–138
Suszynska J, Tisonczyk J et al (2010) Reduced homocysteine-thiolactonase activity in Alzheimer’s disease. J Alzheimers Dis 19(4):1177–1183
Sztanek F, Seres I et al (2012) Decreased paraoxonase 1 (PON1) lactonase activity in hemodialyzed and renal transplanted patients. A novel cardiovascular biomarker in end-stage renal disease. Nephrol Dial Transplant. doi:10.1093/ndt/gfr753
Teiber JF, Draganov DI et al (2004) Purified human serum PON1 does not protect LDL against oxidation in the in vitro assays initiated with copper or AAPH. J Lipid Res 45(12):2260–2268
Türkeli H, Caycı T et al (2010) Paraoxonase-1 activity determination via paraoxon substrate yields no significant difference in mild hyperhomocysteinemia. Int J Cardiol 145(1):42–43
Tward A, Xia YR et al (2002) Decreased atherosclerotic lesion formation in human serum paraoxonase transgenic mice. Circulation 106(4):484–490
Undas A, Perla J et al (2004) Autoantibodies against N-homocysteinylated proteins in humans—Implications for atherosclerosis. Stroke 35(6):1299–1304
Undas A, Stepien E et al (2006) Folic acid administration and antibodies against homocysteinylated proteins in subjects with hyperhomocysteinemia. Thromb Haemost 96(3):342–347
Varga E, Seres I et al (2009) Serum cystatin C is a determinant of paraoxonase activity in hemodialyzed and renal transplanted patients. Dis Markers 26(3):141–148
Vos E (2008) Homocysteine levels, paraoxonase 1 (PON1) activity, and cardiovascular risk. JAMA 300(2):168–169 (author reply 169)
Wehr H, Bednarska-Makaruk M et al (2009) Paraoxonase activity and dementia. J Neurol Sci 283(1–2):107–108
Weijun G, Juming L et al (2008) Effects of plasma homocysteine levels on serum HTase/PON activity in patients with type 2 diabetes. Adv Ther 25(9):884–893
Zafiropoulos A, Linardakis M et al (2010) Paraoxonase 1 R/Q alleles are associated with differential accumulation of saturated versus 20:5n3 fatty acid in human adipose tissue. J Lipid Res 51(7):1991–2000
Zengi O, Karakas A et al (2011) Urinary 8-hydroxy-2′-deoxyguanosine level and plasma paraoxonase 1 activity with Alzheimer’s disease. Clin Chem Lab Med 50(3):529–534
Zhao Y, Ma Y et al (2012) Association between PON1 activity and coronary heart disease risk: a meta-analysis based on 43 studies. Mol Genet Metab 105(1):141–148
Acknowledgments
Supported in part by a grant from the National Science Center, Poland (MAY-2011/02/1/NZ1/00010).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Perła-Kaján, J., Jakubowski, H. Paraoxonase 1 and homocysteine metabolism. Amino Acids 43, 1405–1417 (2012). https://doi.org/10.1007/s00726-012-1321-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-012-1321-z