Abstract
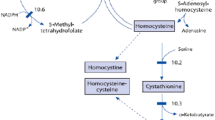
Genetic or nutritional disorders in homocysteine (Hcy) metabolism elevate Hcy-thiolactone and cause heart and brain diseases. Hcy-thiolactone has been implicated in these diseases because it has the ability to modify protein lysine residues and generate toxic N-Hcy-proteins with auto-immunogenic, pro-thrombotic, and amyloidogenic properties. Bleomycin hydrolase (Blmh) has the ability to hydrolyze l-Hcy-thiolactone (but not d-Hcy-thiolactone) to Hcy in vitro, but whether this reflects a physiological function has been unknown. Here, we show that Blmh −/− mice excreted in urine 1.8-fold more Hcy-thiolactone than wild-type Blmh +/+ animals (P = 0.02). Hcy-thiolactone was elevated 2.3-fold in brains (P = 0.004) and 2.0-fold in kidneys (P = 0.047) of Blmh −/− mice relative to Blmh +/+ animals. Plasma N-Hcy-protein was elevated in Blmh −/− mice fed a normal (2.3-fold, P < 0.001) or hyperhomocysteinemic diet (1.5-fold, P < 0.001), compared with Blmh +/+ animals. More intraperitoneally injected l-Hcy-thiolactone was recovered in plasma in Blmh −/− mice than in wild-type Blmh +/+ animals (83.1 vs. 39.3 μM, P < 0.0001). In Blmh +/+ mice injected intraperitoneally with d-Hcy-thiolactone, d,l-Hcy-thiolactone, or l-Hcy-thiolactone, 88, 47, or 6.3%, respectively, of the injected dose was recovered in plasma. The incidence of seizures induced by l-Hcy-thiolactone injections (3,700 nmol/g body weight) was higher in Blmh −/− than in Blmh +/+ mice (93.8 vs. 29.5%, P < 0.001). Using the Blmh null mice, we provide the first direct evidence that a specific Hcy metabolite, Hcy-thiolactone, rather than Hcy itself, is neurotoxic in vivo. Taken together, our findings indicate that Blmh protects mice against l-Hcy-thiolactone toxicity by metabolizing it to Hcy and suggest a mechanism by which Blmh might protect against neurodegeneration associated with hyperhomocysteinemia and Alzheimer’s disease.


Similar content being viewed by others
Abbreviations
- Blmh:
-
Bleomycin hydrolase
- Cbs:
-
Cystathionine β-synthase
- Hcy:
-
Homocysteine
- tHcy:
-
Total Hcy
- i.p.:
-
Intraperitoneal
- Mthfr:
-
Methylenetetrahydrofolate reductase
References
Bromme D, Rossi AB, Smeekens SP, Anderson DC, Payan DG (1996) Human bleomycin hydrolase: molecular cloning, sequencing, functional expression, and enzymatic characterization. Biochemistry 35:6706–6714
Chwatko G, Jakubowski H (2005a) The determination of homocysteine-thiolactone in human plasma. Anal Biochem 337:271–277
Chwatko G, Jakubowski H (2005b) Urinary excretion of homocysteine-thiolactone in humans. Clin Chem 51:408–415
Chwatko G, Boers GH, Strauss KA, Shih DM, Jakubowski H (2007) Mutations in methylenetetrahydrofolate reductase or cystathionine beta-synthase gene, or a high-methionine diet, increase homocysteine thiolactone levels in humans and mice. FASEB J 21:1707–1713
Endo N, Nishiyama K, Otsuka A, Kanouchi H, Taga M, Oka T (2006) Antioxidant activity of vitamin B6 delays homocysteine-induced atherosclerosis in rats. Br J Nutr 95:1088–1093
Folbergrova J (1997) Anticonvulsant action of both NMDA and non-NMDA receptor antagonists against seizures induced by homocysteine in immature rats. Exp Neurol 145:442–450
Glowacki R, Jakubowski H (2004) Cross-talk between Cys34 and lysine residues in human serum albumin revealed by N-homocysteinylation. J Biol Chem 279:10864–10871
Glowacki R, Bald E, Jakubowski H (2010) Identification and origin of Nε-homocysteinyl-lysine isopeptide in humans and mice. Amino Acids 39:1563–1569
Gu W, Lu J, Yang G, Dou J, Mu Y, Meng J, Pan C (2008) Plasma homocysteine thiolactone associated with risk of macrovasculopathy in Chinese patients with type 2 diabetes mellitus. Adv Ther 25:914–924
Harker LA, Slichter SJ, Scott CR, Ross R (1974) Homocystinemia. Vascular injury and arterial thrombosis. N Engl J Med 291:537–543
Jakubowski H (1990) Proofreading in vivo: editing of homocysteine by methionyl-tRNA synthetase in Escherichia coli. Proc Natl Acad Sci USA 87:4504–4508
Jakubowski H (1997) Metabolism of homocysteine thiolactone in human cell cultures. Possible mechanism for pathological consequences of elevated homocysteine levels. J Biol Chem 272:1935–1942
Jakubowski H (1999) Protein homocysteinylation: possible mechanism underlying pathological consequences of elevated homocysteine levels. FASEB J 13:2277–2283
Jakubowski H (2000a) Homocysteine thiolactone: metabolic origin and protein homocysteinylation in humans. J Nutr 130:377S–381S
Jakubowski H (2000b) Calcium-dependent human serum homocysteine thiolactone hydrolase. A protective mechanism against protein N-homocysteinylation. J Biol Chem 275:3957–3962
Jakubowski H (2002) Homocysteine is a protein amino acid in humans. Implications for homocysteine-linked disease. J Biol Chem 277:30425–30428
Jakubowski H (2006) Mechanism of the condensation of homocysteine thiolactone with aldehydes. Chemistry 12:8039–8043
Jakubowski H (2007) Facile syntheses of [35S]homocysteine-thiolactone, [35S]homocystine, [35S]homocysteine, and [S-nitroso-35S]homocysteine. Anal Biochem 370:124–126
Jakubowski H (2008a) The pathophysiological hypothesis of homocysteine thiolactone-mediated vascular disease. J Physiol Pharmacol 59(Suppl 9):155–167
Jakubowski H (2008b) New method for the determination of protein N-linked homocysteine. Anal Biochem 380:257–261
Jakubowski H (2011) Quality control in tRNA charging—editing of homocysteine. Acta Biochim Pol 58:149–163
Jakubowski H, Goldman E (1993) Synthesis of homocysteine thiolactone by methionyl-tRNA synthetase in cultured mammalian cells. FEBS Lett 317:237–240
Jakubowski H, Zhang L, Bardeguez A, Aviv A (2000) Homocysteine thiolactone and protein homocysteinylation in human endothelial cells: implications for atherosclerosis. Circ Res 87:45–51
Jakubowski H, Boers GH, Strauss KA (2008) Mutations in cystathionine beta-synthase or methylenetetrahydrofolate reductase gene increase N-homocysteinylated protein levels in humans. FASEB J 22:4071–4076
Jakubowski H, Perla-Kajan J, Finnell RH, Cabrera RM, Wang H, Gupta S, Kruger WD, Kraus JP, Shih DM (2009) Genetic or nutritional disorders in homocysteine or folate metabolism increase protein N-homocysteinylation in mice. FASEB J 23:1721–1727
Kajiya A, Kaji H, Isobe T, Takeda A (2006) Processing of amyloid beta-peptides by neutral cysteine protease bleomycin hydrolase. Protein Pept Lett 13:119–123
Kamata Y, Itoh Y, Kajiya A, Karasawa S, Sakatani C, Takekoshi S, Osamura RY, Takeda A (2007) Quantification of neutral cysteine protease bleomycin hydrolase and its localization in rat tissues. J Biochem 141:69–76
Kamata Y, Taniguchi A, Yamamoto M, Nomura J, Ishihara K, Takahara H, Hibino T, Takeda A (2009) Neutral cysteine protease bleomycin hydrolase is essential for the breakdown of deiminated filaggrin into amino acids. J Biol Chem 284:12829–12836
Lefterov IM, Koldamova RP, Lefterova MI, Schwartz DR, Lazo JS (2001) Cysteine 73 in bleomycin hydrolase is critical for amyloid precursor protein processing. Biochem Biophys Res Commun 283:994–999
Mudd SH, Levy HL, Krauss JP (2001) Disorders of transsulfuration. In: Scriver CR, Beaudet AL, Sly WS, Valle D, Childs B, Kinzler KW, Vogelstein B (eds) The metabolic and molecular bases of inherited disease, vol 2. Mc Graw-Hill, New York, pp 2007–2056
Paoli P, Sbrana F, Tiribilli B, Caselli A, Pantera B, Cirri P, De Donatis A, Formigli L, Nosi D, Manao G, Camici G, Ramponi G (2010) Protein N-homocysteinylation induces the formation of toxic amyloid-like protofibrils. J Mol Biol 400:889–907
Papassotiropoulos A, Bagli M, Jessen F, Frahnert C, Rao ML, Maier W, Heun R (2000) Confirmation of the association between bleomycin hydrolase genotype and Alzheimer’s disease. Mol Psychiatry 5:213–215
Perla-Kajan J, Jakubowski H (2010) Paraoxonase 1 protects against protein N-homocysteinylation in humans. FASEB J 24:931–936
Perla-Kajan J, Marczak L, Kajan L, Skowronek P, Twardowski T, Jakubowski H (2007) Modification by homocysteine thiolactone affects redox status of cytochrome C. Biochemistry 46:6225–6231
Perla-Kajan J, Stanger O, Luczak M, Ziolkowska A, Malendowicz LK, Twardowski T, Lhotak S, Austin RC, Jakubowski H (2008) Immunohistochemical detection of N-homocysteinylated proteins in humans and mice. Biomed Pharmacother 62:473–479
Rasic-Markovic A, Stanojlovic O, Hrncic D, Krstic D, Colovic M, Susic V, Radosavljevic T, Djuric D (2009) The activity of erythrocyte and brain Na+/K+ and Mg2+-ATPases in rats subjected to acute homocysteine and homocysteine thiolactone administration. Mol Cell Biochem 327:39–45
Sauls DL, Lockhart E, Warren ME, Lenkowski A, Wilhelm SE, Hoffman M (2006) Modification of fibrinogen by homocysteine thiolactone increases resistance to fibrinolysis: a potential mechanism of the thrombotic tendency in hyperhomocysteinemia. Biochemistry 45:2480–2487
Schwartz DR, Homanics GE, Hoyt DG, Klein E, Abernethy J, Lazo JS (1999) The neutral cysteine protease bleomycin hydrolase is essential for epidermal integrity and bleomycin resistance. Proc Natl Acad Sci USA 96:4680–4685
Selhub J, Troen A, Rosenberg IH (2010) B vitamins and the aging brain. Nutr Rev 68(Suppl 2):S112–S118
Sibrian-Vazquez M, Escobedo JO, Lim S, Samoei GK, Strongin RM (2010) Homocystamides promote free-radical and oxidative damage to proteins. Proc Natl Acad Sci USA 107:551–554
Smith AD (2008) The worldwide challenge of the dementias: a role for B vitamins and homocysteine? Food Nutr Bull 29:S143–S172
Smith AD, Smith SM, de Jager CA, Whitbread P, Johnston C, Agacinski G, Oulhaj A, Bradley KM, Jacoby R, Refsum H (2010) Homocysteine-lowering by B vitamins slows the rate of accelerated brain atrophy in mild cognitive impairment: a randomized controlled trial. PLoS One 5:e12244
Spence AM, Rasey JS, Dwyer-Hansen L, Grunbaum Z, Livesey J, Chin L, Nelson N, Stein D, Krohn KA, Ali-Osman F (1995) Toxicity, biodistribution and radioprotective capacity of l-homocysteine thiolactone in CNS tissues and tumors in rodents: comparison with prior results with phosphorothioates. Radiother Oncol 35:216–226
Sprince H, Parker CM, Josephs JA Jr, Magazino J (1969) Convulsant activity of homocysteine and other short-chain mercaptoacids: protection therefrom. Ann N Y Acad Sci 166:323–325
Suszynska J, Tisonczyk J, Lee HG, Smith MA, Jakubowski H (2010) Reduced homocysteine-thiolactonase activity in Alzheimer’s disease. J Alzheimers Dis 19:1177–1183
Undas A, Perla J, Lacinski M, Trzeciak W, Kazmierski R, Jakubowski H (2004) Autoantibodies against N-homocysteinylated proteins in humans: implications for atherosclerosis. Stroke 35:1299–1304
Undas A, Jankowski M, Twardowska M, Padjas A, Jakubowski H, Szczeklik A (2005) Antibodies to N-homocysteinylated albumin as a marker for early-onset coronary artery disease in men. Thromb Haemost 93:346–350
Velez-Carrasco W, Merkel M, Twiss CO, Smith JD (2008) Dietary methionine effects on plasma homocysteine and HDL metabolism in mice. J Nutr Biochem 19:362–370
Yang X, Gao Y, Zhou J, Zhen Y, Yang Y, Wang J, Song L, Liu Y, Xu H, Chen Z, Hui R (2006) Plasma homocysteine thiolactone adducts associated with risk of coronary heart disease. Clin Chim Acta 364:230–234
Zabczyk M, Glowacki R, Machnik A, Herod P, Kazek G, Jakubowski H, Undas A (2011) Elevated concentrations of Nε-homocysteinyl-lysine isopeptide in acute myocardial infarction: links with ADMA formation. Clin Chem Lab Med 49:729–735
Zhou J, Moller J, Danielsen CC, Bentzon J, Ravn HB, Austin RC, Falk E (2001) Dietary supplementation with methionine and homocysteine promotes early atherosclerosis but not plaque rupture in ApoE-deficient mice. Arterioscler Thromb Vasc Biol 21:1470–1476
Zimny J, Sikora M, Guranowski A, Jakubowski H (2006) Protective mechanisms against homocysteine toxicity: the role of bleomycin hydrolase. J Biol Chem 281:22485–22492
Acknowledgments
We thank John Lazo for kindly providing Blmh null mice and for his comments on the manuscript. This work was supported in part by grants from the American Heart Association (0855919D) and the National Science Center, Poland (DEC-2011/01/B/NZ1/03417).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Borowczyk, K., Tisończyk, J. & Jakubowski, H. Metabolism and neurotoxicity of homocysteine thiolactone in mice: protective role of bleomycin hydrolase. Amino Acids 43, 1339–1348 (2012). https://doi.org/10.1007/s00726-011-1207-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-011-1207-5