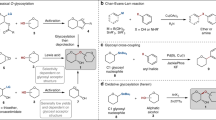
Abstract
The synthesis of N-protected glycosyl amino acids from amines has been investigated and it was found that, under microwave conditions, glycosylamines could be hydrolyzed leading to new products containing a glycosyl ester linkage. The efficiency of the microwave-induced glycosylation of aspartic acid was studied comparing the microwave activity between amide and ester bond formation. Different sugar moieties have been employed to demonstrate the simple and reproducible coupling methodology. New glycosyl ester compounds were further characterized by NMR spectroscopy.



Similar content being viewed by others
Abbreviations
- DCM:
-
Dichloromethane
- FCC:
-
Flash column chromatography
- Fmoc:
-
9H-Fluoren-9-ylmethoxycarbonyl
- H2BC:
-
Heteronuclear two bond correlation
- HMBC:
-
Heteronuclear multiple bond correlation
- HSQC:
-
Heteronuclear single quantum coherence
- MS:
-
Mass spectrometry
- MW:
-
Microwave
- NMM:
-
N-Methylmorpholine
- NMR:
-
Nuclear magnetic resonance
- RP-HPLC:
-
Reverse phase-high performance liquid chromatography
- SPGPS:
-
Solid phase glycopeptide synthesis
- TBCR:
-
Triazine-based coupling reagent
- t-Bu:
-
tert-Butyl
- TFA:
-
Trifluoroacetic acid
- TLC:
-
Thin layer chromatography
References
Christiansen-Brams I, Meldal M, Bock K (1993) Protected-mode synthesis of N-linked glycopeptides: single-step preparation of building blocks as peracetyl glycosylated N αFmoc asparagine OPfp esters. J Chem Soc Perkin Trans 1 13:1461–1471
Gelens E, Smeets L, Sliedregt L, Van Steen BJ, Kruse CG, Leurs R, Orru RVA (2005) An atom efficient and solvent-free synthesis of structurally diverse amides using microwaves. Tetrahedron Lett 46:3751–3754
Kaminski ZJ (1994) The concept of superactive esters. Could peptide synthesis be improved by inventing superactive esters? Int J Pept Protein Res 43:312–319
Kaminski ZJ, Papini AM, Kolesinska B, Kolesinska J, Jastrzabek K, Sabatino G, Bianchini R (2005a) Process for the preparation of N-triazinylammonium salts. Applicant: Italvelluti S.p.a. PCT/EP2005/055793
Kaminski ZJ, Kolesinska B, Kolesinska J, Sabatino G, Chelli M, Rovero P, Blaszczyk M, Glowka ML, Papini AM (2005b) N-Triazinylammonium tetrafluoroborates. A new generation of efficient coupling reagents useful for peptide synthesis. J Am Chem Soc 127:16912–16920
Kappe CO (2004) Controlled microwave heating in modern organic synthesis. Angew Chem Int Ed 43:6250–6284
Kappe CO, Dallinger D (2006) The impact of microwave synthesis on drug discovery. Nat Rev Drug Disc 5:51–63
Keglevic D, Valentekovic S (1976) Syntheses and rearrangements of d-glucosyl esters of aspartic acid linked through the l- or 4-carboxyl group. Carbohydr Res 47:35–48
Keglevic D, Valentekovic S, Roglic G, Goles D, Plavsic F (1973) Glycosyl esters of amino acids. Carbohydr Res 29:25–39
Keglevic D, Horvat J, Plavsic F (1976) Synthesis and chemical behaviour of d-glycosyl esters of glutamic acid having the side chain carboxyl group involved in the glycosidic linkage. Carbohydr Res 47:49–61
Klein J (1958) Preparation of cis-2-hydroxycyclohexaneacetic acid lactone. J Org Chem 23:1209–1210
Liautard V, Pillard C, Desvergnes V, Martin OR (2008) One-step synthesis of N-protected glycosylamines from sugar hemiacetals. Carbohydr Res 343:2111–2117
Meldal M (1994a) Glycopeptide synthesis. In: Lee YC, Lee RT (eds) Neoglycoconjugates: preparation and application. Academic Press, Orlando, pp 145–198
Meldal M (1994b) Recent developments in glycopeptide and oligosaccharide synthesis. Curr Opin Struct Biol 4:710–718
Meldal M, Bock K (1994) A general approach to the synthesis of O- and N-linked glycopeptides. Glycoconjugate J 11:59–63
Paolini I, Nuti F, Pozo-Carrero MC, Barbetti F, Kolesinska B, Kaminski ZJ, Chelli M, Papini AM (2007) A convenient microwave-assisted synthesis of N-glycosyl amino acids. Tetrahedron Lett 48:2901–2904
Rademacher TW, Pareckh RB, Dwek RA (1988) Glycobiology. Ann Rev Biochem 57:785–838
Seijas JA, Vazquez-Tato MP, Crecente-Campo J (2007) Microwave-assisted solvent-free synthesis of enol carbamates. Syn Lett 15:2420–2424
Soli ED, Manoso MC, Patterson MC, DeShong J (1999) Azide and cyanide displacements via hypervalent silicate intermediates. J Org Chem 64:3171–3177
Toshuki I (1994) patent JP 93-77583 19930311
Van Ameijde J, Albada HB, Liskamp RJ (2002) A convenient preparation of several N-linked glycoamino acid building blocks for efficient solid-phase synthesis of glycopeptides. J Chem Soc Perkin Trans 1 8:1042–1049
Varki A (1999) Essentials of glycobiology, ed. Cold Spring Harbor Laboratory Press, New York
Vetter D, Gallop MA (1995) Strategies for the synthesis and screening of glycoconjugates: a library of glycosylamines. Bioconjugate Chem 6:316–318
Ying L, Liu R, Zhang J, Lam K, Lebrilla CB, Gervay-Hague J (2005) A topologically segregated one-bead-one-compound combinatorial glycopeptide library for identification of lectin ligands. J Comb Chem 7:372–384
Acknowledgments
We acknowledge Ente Cassa Risparmio di Firenze for supporting PeptLab. We thank Dr. Duccio Lambardi for his contribution in performing the high resolution mass spectra.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Real-Fernández, F., Nuti, F., Bonache, M.A. et al. Microwave-assisted reaction of glycosylamine with aspartic acid. Amino Acids 39, 599–604 (2010). https://doi.org/10.1007/s00726-010-0484-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-010-0484-8