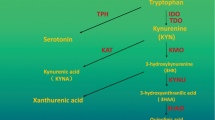
Abstract
Indoleamine 2,3-dioxygenase (IDO) catalyzes the first and rate-limiting step of Kynurenine pathway along the major route of Tryptophan catabolism. The scientific interest in the enzyme has been growing since the observations of the involvement of IDO in the mechanisms of immune tolerance and in the mechanisms of tumor immuno-editing process. In view of this latter observation, in particular, preclinical studies of small molecule inhibitors of the enzyme have indicated the feasibility to thwart the immuno-editing process and to enhance the efficacy of current chemotherapeutic agents, supporting the notion that IDO is a novel target in cancer disease.
This review covers the structural and conformational aspects of substrate recognition by IDO, including the catalytic mechanism and the so-far puzzling mechanisms of enzyme activation. Furthermore, we discuss the recent advances of medicinal chemistry in the field of IDO inhibitors.











Similar content being viewed by others
References
Austin CJ, Astelbauer F, Kosim-Satyaputra P, Ball HJ, Willows RD, Jamie JF, Hunt NH (2008) Mouse and human indoleamine 2,3-dioxygenase display some distinct biochemical and structural properties. Amino Acids (in press)
Ball HJ, Sanchez-Perez A, Weiser S, Austin CJ, Astelbauer F, Miu J, McQuillan JA, Stocker R, Jermiin LS, Hunt NH (2007) Characterization of an indoleamine 2,3-dioxygenase-like protein found in humans and mice. Gene 396:203–213
Ball HJ, Yuasa HJ, Austin CJ, Weiser S, Hunt NH (2008) Indoleamine 2,3-dioxygenase-2; a new enzyme in the kynurenine pathway. Int J Biochem Cell Biol (in press)
Batabyal D, Yeh SR (2007) Human tryptophan dioxygenase: a comparison to indoleamine 2,3-dioxygenase. J Am Chem Soc 129:15690–15701
Bendall DS (1996) Protein electron transfer. BIOS Scientific Publishers, Oxford
Brastianos HC, Vottero E, Patrick BO, Soest RV, Matainaho T, Mauk AG, Andersen RJ (2006) Exiguamine A, an indoleamine-2,3-dioxygenase (IDO) inhibitor isolated from the marine sponge Neopetrosia exigua. J Am Chem Soc 128:16046–16047
Cady SG, Sono M (1991) 1-Methyl-dl-tryptophan, beta-(3-benzofuranyl)-dl-alanine (the oxygen analog of tryptophan), and beta-[3-benzo(b)thienyl]-dl-alanine (the sulfur analog of tryptophan) are competitive inhibitors for indoleamine 2,3-dioxygenase. Arch Biochem Biophys 291:326–333
Carr G, Chung MK, Mauk AG, Andersen RJ (2008) Synthesis of indoleamine 2,3-dioxygenase inhibitory analogues of the sponge alkaloid exiguamine A. J Med Chem 51:2634–2637
Chauhan N, Basran J, Efimov I, Svistunenko DA, Seward HE, Moody PC, Raven EL (2008) The role of serine 167 in human indoleamine 2,3-dioxygenase: a comparison with tryptophan 2,3-dioxygenase. Biochemistry 47:4761–4769
Eguchi N, Watanabe Y, Kawanishi K, Hashimoto Y, Hayaishi O (1984) Inhibition of indoleamine 2,3-dioxygenase and tryptophan 2,3-dioxygenase by beta-carboline and indole derivatives. Arch Biochem Biophys 232:602–609
Fallarino F, Grohmann U, Hwang KW, Orabona C, Vacca C, Bianchi R, Belladonna ML, Fioretti MC, Alegre ML, Puccetti P (2003) Modulation of tryptophan catabolism by regulatory T cells. Nat Immunol 4:1206–1212
Gaspari P, Banerjee T, Malachowski WP, Muller AJ, Prendergast GC, DuHadaway J, Bennett S, Donovan AM (2006) Structure-activity study of brassinin derivatives as indoleamine 2,3-dioxygenase inhibitors. J Med Chem 49:684–692
Grohmann U, Fallarino F, Puccetti P (2003) Tolerance, DCs and tryptophan: much ado about IDO. Trends Immunol 24:242–248
Hayaishi O (1993) My life with tryptophan-never a dull moment. Protein Sci 2:472–475
Hayaishi O, Hirata F, Ohnishi T, Henry JP, Rosenthal I, Katoh A (1977) Indoleamine 2,3-dioxygenase: incorporation of 18O2 and 18O2 into the reaction products. J Biol Chem 252:3548–3550
Higuchi K, Kuno S, Hayaishi O (1963) Enzymatic formation of d-kynurenin. Federation Proc 22:243 (abstr.)
Hirata F, Hayaishi O (1975) Studies on indoleamine 2,3-dioxygenase. I. Superoxide anion as substrate. J Biol Chem 250:5960–5966
Hirata F, Ohnishi T, Hayaishi O (1977) Indoleamine 2,3-dioxygenase. Characterization and properties of enzyme. O2-complex. J Biol Chem 252:4637–4642
Hou DY, Muller AJ, Sharma MD, DuHadaway J, Banerjee T, Johnson M, Mellor AL, Prendergast GC, Munn DH (2007) Inhibition of indoleamine 2,3-dioxygenase in dendritic cells by stereoisomers of 1-methyl-tryptophan correlates with antitumor responses. Cancer Res 67:792–801
Katz JB, Muller AJ, Prendergast GC (2008) Indoleamine 2,3-dioxygenase in T-cell tolerance and tumoral immune escape. Immunol Rev 222:206–221
Kumar S, Malachowski WP, DuHadaway JB, LaLonde JM, Carroll PJ, Jaller D, Metz R, Prendergast GC, Muller AJ (2008) Indoleamine 2,3-dioxygenase is the anticancer target for a novel series of potent naphthoquinone-based inhibitors. J Med Chem 51:1706–1718
Lob S, Konigsrainer A, Schafer R, Rammensee HG, Opelz G, Terness P (2008a) Levo- but not dextro-1-methyl tryptophan abrogates the IDO activity of human dendritic cells. Blood 111:2152–2154
Lob S, Konigsrainer A, Zieker D, Brucher BL, Rammensee HG, Opelz G, Terness P (2008b) IDO1 and IDO2 are expressed in human tumors: levo- but not dextro-1-methyl tryptophan inhibits tryptophan catabolism. Cancer Immunol Immunother. In press
Macchiarulo A, Nuti R, Bellocchi D, Camaioni E, Pellicciari R (2007) Molecular docking and spatial coarse graining simulations as tools to investigate substrate recognition, enhancer binding and conformational transitions in indoleamine-2,3-dioxygenase (IDO). Biochim Biophys Acta 1774:1058–1068
Maghzal GJ, Thomas SR, Hunt NH, Stocker R (2008) Cytochrome b5, not superoxide anion radical, is a major reductant of indoleamine 2,3-dioxygenase in human cells. J Biol Chem 283:12014–12025
Malachowski WP, Metz R, Prendergast GC, Muller AJ (2005) A new cancer immunosuppression target: indoleamine 2,3-dioxygenase (IDO). A review of the IDO mechanism, inhibition and therapeutic applications. Drugs Future 30:897–909
Mellor AL, Munn DH (2004) IDO expression by dendritic cells: tolerance and tryptophan catabolism. Nat Rev Immunol 4:762–774
Metz R, Duhadaway JB, Kamasani U, Laury-Kleintop L, Muller AJ, Prendergast GC (2007) Novel tryptophan catabolic enzyme IDO2 is the preferred biochemical target of the antitumor indoleamine 2,3-dioxygenase inhibitory compound d-1-methyl-tryptophan. Cancer Res 67:7082–7087
Muller AJ, DuHadaway JB, Donover PS, Sutanto-Ward E, Prendergast GC (2005a) Inhibition of indoleamine 2,3-dioxygenase, an immunoregulatory target of the cancer suppression gene Bin1, potentiates cancer chemotherapy. Nat Med 11:312–319
Muller AJ, Malachowski WP, Prendergast GC (2005b) Indoleamine 2,3-dioxygenase in cancer: targeting pathological immune tolerance with small-molecule inhibitors. Expert Opin Ther Targets 9:831–849
Muller AJ, Prendergast GC (2007) Indoleamine 2,3-dioxygenase in immune suppression and cancer. Curr Cancer Drug Targets 7:31–40
Muller AJ, Scherle PA (2006) Targeting the mechanisms of tumoral immune tolerance with small-molecule inhibitors. Nat Rev Cancer 6:613–625
Munn DH, Zhou M, Attwood JT, Bondarev I, Conway SJ, Marshall B, Brown C, Mellor AL (1998) Prevention of allogeneic fetal rejection by tryptophan catabolism. Science 281:1191–1193
Ozaki Y, Nichol CA, Duch DS (1987) Utilization of dihydroflavin mononucleotide and superoxide anion for the decyclization of l-tryptophan by murine epididymal indoleamine 2,3-dioxygenase. Arch Biochem Biophys 257:207–216
Ozaki Y, Reinhard JF Jr, Nichol CA (1986) Cofactor activity of dihydroflavin mononucleotide and tetrahydrobiopterin for murine epididymal indoleamine 2,3-dioxygenase. Biochem Biophys Res Commun 137:1106–1111
Papadopoulou ND, Mewies M, McLean KJ, Seward HE, Svistunenko DA, Munro AW, Raven EL (2005) Redox and spectroscopic properties of human indoleamine 2,3-dioxygenase and a His303Ala variant: implications for catalysis. Biochemistry 44:14318–14328
Pereira A, Vottero E, Roberge M, Mauk AG, Andersen RJ (2006) Indoleamine 2,3-dioxygenase inhibitors from the Northeastern Pacific Marine Hydroid Garveia annulata. J Nat Prod 69:1496–1499
Peters JC (1991) Tryptophan nutrition and metabolism: an overview. Adv Exp Med Biol 294:345–358
Peterson AC, La Loggia AJ, Hamaker LK et al (1993) Evaluation of substituted beta-carbolines as noncompetitive indoleamine 2,3-dioxygenase inhibitors. Med Chem Res 4:473–482
Peterson AC, Migawa MT, Martin MM et al (1994) Evaluation of functionalized tryptophan derivatives and related compounds as competitive inhibitors of Indoleamine 2,3-dioxygenase. Med Chem Res 3:531–544
Prendergast GC (2008) Immune escape as a fundamental trait of cancer: focus on IDO. Oncogene.
Puccetti P, Grohmann U (2007) IDO and regulatory T cells: a role for reverse signalling and non-canonical NF-kappaB activation. Nat Rev Immunol 7:817–823
Romani L, Fallarino F, De Luca A, Montagnoli C, D’Angelo C, Zelante T, Vacca C, Bistoni F, Fioretti MC, Grohmann U, Segal BH, Puccetti P (2008) Defective tryptophan catabolism underlies inflammation in mouse chronic granulomatous disease. Nature 451:211–215
Ruddick JP, Evans AK, Nutt DJ, Lightman SL, Rook GA, Lowry CA (2006) Tryptophan metabolism in the central nervous system: medical implications. Expert Rev Mol Med 8:1–27
Samelson-Jones BJ, Yeh SR (2006) Interactions between nitric oxide and indoleamine 2,3-dioxygenase. Biochemistry 45:8527–8538
Sono M (1989) Enzyme kinetic and spectroscopic studies of inhibitor and effector interactions with indoleamine 2,3-dioxygenase. 2. Evidence for the existence of another binding site in the enzyme for indole derivative effectors. Biochemistry 28:5400–5407
Sono M, Cady SG (1989) Enzyme kinetic and spectroscopic studies of inhibitor and effector interactions with indoleamine 2,3-dioxygenase. 1. Norharman and 4-phenylimidazole binding to the enzyme as inhibitors and heme ligands. Biochemistry 28:5392–5399
Sono M, Roach MP, Coulter ED, Dawson JH (1996) Heme-containing oxygenases. Chem Rev 96:2841–2888
Sono M, Taniguchi T, Watanabe Y, Hayaishi O (1980) Indoleamine 2,3-dioxygenase. Equilibrium studies of the tryptophan binding to the ferric, ferrous, and CO-bound enzymes. J Biol Chem 255:1339–1345
Southan MD, Truscott RJW, Jamie JF, Pelosi L, Walker MJ, Maeda H, Iwamoto Y, Tone S (1996) Structural requirements of the competitive binding site of recombinant human indoleamine 2,3-dioxygenase. Med Chem Res 6:343–352
Stone TW, Darlington LG (2002) Endogenous kynurenines as targets for drug discovery and development. Nat Rev Drug Discov 1:609–620
Sugimoto H, Oda S, Otsuki T, Hino T, Yoshida T, Shiro Y (2006) Crystal structure of human indoleamine 2,3-dioxygenase: catalytic mechanism of O2 incorporation by a heme-containing dioxygenase. Proc Natl Acad Sci USA 103:2611–2616
Taniguchi T, Hirata F, Hayaishi O (1977) Intracellular utilization of superoxide anion by indoleamine 2,3-dioxygenase of rabbit enterocytes. J Biol Chem 252:2774–2776
Taylor MW, Feng GS (1991) Relationship between interferon-gamma, indoleamine 2,3-dioxygenase, and tryptophan catabolism. FASEB J 5:2516–2522
Uchida K, Bandow H, Makino R, Sakaguchi K, Iizuka T, Ishimura Y (1985) Infrared spectra of carbon monoxide complexes of indoleamine 2,3-dioxygenase and l-tryptophan 2,3-dioxygenases. Effects of substrates on the CO-stretching frequencies. J Biol Chem 260:1400–1406
Uyttenhove C, Pilotte L, Theate I, Stroobant V, Colau D, Parmentier N, Boon T, Van den Eynde BJ (2003) Evidence for a tumoral immune resistance mechanism based on tryptophan degradation by indoleamine 2,3-dioxygenase. Nat Med 9:1269–1274
Vottero E, Balgi A, Woods K, Tugendreich S, Melese T, Andersen RJ, Mauk AG, Roberge M (2006a) Inhibitors of human indoleamine 2,3-dioxygenase identified with a target-based screen in yeast. Biotechnol J 1:282–288
Vottero E, Mitchell DA, Page MJ, MacGillivray RT, Sadowski IJ, Roberge M, Mauk AG (2006b) Cytochrome b(5) is a major reductant in vivo of human indoleamine 2,3-dioxygenase expressed in yeast. FEBS Lett 580:2265–2268
Watanabe Y, Fujiwara M, O H, Takeuchi T, Hamao U (1978) 2,5-dihydro-l-phemilalanine: a competitive inhibitor of indoleamine 2,3-dioxygenase and tryptophan 2,3-dioxygenase. Biochem Biophys Res Commun 85:273–279
Wheeler KE, Nocek JM, Cull DA, Yatsunyk LA, Rosenzweig AC, Hoffman BM (2007) Dynamic docking of cytochrome b5 with myoglobin and alpha-hemoglobin: heme-neutralization “squares” and the binding of electron-transfer-reactive configurations. J Am Chem Soc 129:3906–3917
Zhang Y, Kang SA, Mukherjee T, Bale S, Crane BR, Begley TP, Ealick SE (2007) Crystal structure and mechanism of tryptophan 2,3-dioxygenase, a heme enzyme involved in tryptophan catabolism and in quinolinate biosynthesis. Biochemistry 46:145–155
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Macchiarulo, A., Camaioni, E., Nuti, R. et al. Highlights at the gate of tryptophan catabolism: a review on the mechanisms of activation and regulation of indoleamine 2,3-dioxygenase (IDO), a novel target in cancer disease. Amino Acids 37, 219–229 (2009). https://doi.org/10.1007/s00726-008-0137-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-008-0137-3