Abstract
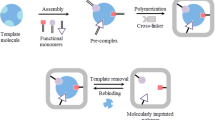
Molecularly imprinted polymers that could recognize the tripeptide Arg-Gly-Asp have been produced with the use of two functional monomers and three different cross-linkers, respectively. Methacrylic acid and acrylamide were used as functional monomers and the role of the ethylene glycol dimethacrylate, trimethylpropane trimethacrylate and N,N′-methylene-bisacrylamide as crosslinking monomers, was investigated on their recognition capability. The % net rebinding and the imprinting factor values were obtained, giving for the methacrylic acid–trimethylpropane trimethacrylate polymer the highest values 12.3% and 2.44, respectively. In addition, this polymer presented lower dissociation constant (K D) value and the higher B max% of theoretical total binding sites than all the other polymers. Rebinding experiments with Lys-Gly-Asp, an analogue of Arg-Gly-Asp, and other different peptides, such as cholecystokinin C-terminal tri- and pentapeptide and gramicidin, further indicated the selectivity of methacrylic acid-trimethylpropane trimethacrylate copolymer for Arg-Gly-Asp giving specific selectivity factor values 1.27, 1.98, 1.31 and 1.67, respectively.






Similar content being viewed by others
Abbreviations
- ABCN:
-
Azo-bis-cyclohexane-carbonitrile
- RGD:
-
Arg-Gly-Asp tripeptide
- KGD:
-
Lys-Gly-Asp tripeptide
- CCK-3, CCK-5:
-
Cholecystokinin C-terminal tripeptide and pentapeptide, respectively
- EGDMA:
-
Ethylene glycol dimethacrylate
- TRIM:
-
Trimethylpropane trimethacrylate
- MAA:
-
Methacrylic acid
- MIPs:
-
Molecularly imprinted polymers
- NIPs:
-
Non imprinted polymers
- SEM:
-
Scanning electron microscopy
References
Allender CJ, Brain KR, Heard CM (1997) Binding cross-reactivity of Boc-phenylalanine enantiomers on molecularly imprinted polymers. Chirality 9:233–237
Andersson LI, Müller R, Vlatakis G, Mosbach K (1995) Mimics of the binding sites of opioid receptors obtained by molecular imprinting of enkephalin and morphine. Proc Natl Acad Sci USA 92:4788–4792
Andersson LI, Müller R, Mosbach K (1996) Molecular imprinting of the endogenous neuropeptide Leu5-enkephalin and some derivatives thereof. Macromol Rapid Commun 17:65–71
Asanuma H, Akiyama T, Kajiya K, Hishiya T, Komiyama M (2001) Molecular imprinting of cyclodextrin in water for the recognition of nanometer-scaled guests. Anal Chim Acta 435:25–33
Basani RB, D’Andrea G, Mitra N, Vilaire G, Richbergs M, Kowalska MA, Bennet JS, Poncz M (2001) RGD-containing Peptides Inhibit Fibrinogen Binding to Platelet αIIbβ3 by Inducing an Allosteric Change in the Amino-terminal Portion of αIib. J Biol Chem 276:13975–13981
Cheong SH, McNiven S, Rachkov A, Levi R, Yano K, Karube I (1997) Testosterone receptor binding mimic constructed using molecular imprinting. Macromolecules 30:1317–1322
Glad M, Norrlow O, Sellergren B, Siegbahn N, Mosbach K (1985) Use of silane monomers for molecular imprinting and enzyme entrapment in polysiloxane-coated porous silica. J Chromatogr 347:11–23
Kempe M (1996) Antibody-mimicking polymers as chiral stationary phases in HPLC. Anal Chem 68:1948–1953
Kempe M, Mosbach K (1995) Separation of amino acids, peptides and proteins on molecularly imprinted stationary phases. J Chromatogr A 691:317–323
Krlz D, Berggren Krlz C, Andersson LI, Mosbach K (1994) Thin-layer chromatography based on the molecular imprinting technique. Anal Chem 66:2636–2639
Komiyama M, Takeuchi T, Mukawa T, Asanuma H (2003) Molecular imprinting for fundamentals to applications. Wiley, VCH
Lanza F, Sellergren B (2004) Molecularly imprinted polymers via high-throughput and combinatorial techniques. Macromol Rapid Commun 25:59–68
Liao JL, Wang Y, Hjertèn S (1996) A novel support with artificially created recognition for the selective removal of proteins and for affinity chromatography. Chromatographia 42:259–262
Mosbach K (1994) Molecular imprinting. Trends Biochem Sci 19:9–14
Mosbach K, Ramström O (1996) The emerging technique of molecular imprinting and its future impact on biotechnology. Biotechnology 14:163–170
Mosbach K, Haupt K (1998) Some new developments and challenges in noncovalent molecular imprinting technology. J Mol Recogn 2:62–68
Nishino H, Huang CS, Shea KJ (2006) Selective protein capture by epitope imprinting. Angew Chem Int Ed 45:2392–2396
Ojima I, Chakravarty S, Dong Q (1995) Antithrombotic agents: from RGD to peptide mimetics. Bioorg Med Chem 3:337–360
Papaioannou EH, Liakopoulou-Kyriakides M, Papi RM, Kyriakidis DA (2007) Molecularly imprinted polymers for cholecystokinin C-terminal pentapeptide. Macromol Chem Phys 208:2621–2627
Ramström O (2005) Synthesis and selection of functional and structural monomers. In: Ramström O, Yan M (eds) Molecularly imprinted materials: science and technology. Marcel Dekker, New York
Ramström O, Nicholls IA, Mösbach K (1994) Synthetic peptide receptor mimics: highly stereoselective recognition in non-covalent molecularly imprinted polymers. Tetrah Asym 5:649–656
Sellergren B (2001) The non-covalent approach to molecular imprinting. In: Sellergren B (ed) Molecularly imprinted polymers: man-made mimics of antibodies and their applications in analytical chemistry. Elsevier Science BV, London
Sellergren B, Lepisto M, Mosbach K (1988) Highly enantioselective and substrate-selective polymers obtained by molecular imprinting utilizing noncovalent interactions. NMR and chromatographic studies on the nature of recognition. J Am Chem Soc 110:5853–5860
Shea KJ, Spivak DA, Sellergren B (1993) Polymer complements to nucleotide bases. Selective binding of adenine derivatives to imprinted polymers. J Am Chem Soc 115:3368–3369
Wulff G (1995) Molecular imprinting in cross-linked materials with the aid of molecular templates-a way towards artificial antibodies. Angew Chem Int Ed Engl 34:1812–1832
Wulff G, Sarhan A, Zabrocki K (1973) Enzyme-analogue built polymers and their use for the resolution of racemates. Tetrahedron Lett 44:4329–4332
Yilmaz E, Schmidt RH, Mosbach K (2005) The noncovalent approach. In: Ramström O, Yan M (eds) Molecularly imprinted materials: science and technology. Marcel Dekker, New York
Ye L, Ramström O, Mosbach K (1998) Molecularly imprinted polymeric adsorbents for byproduct removal. Anal Chem 70:2789–2795
Ye L, Weiss R, Mosbach K (2000) Synthesis and characterization of molecularly imprinted microspheres. Macromolecules 33:8239–8245
Yu C, Mosbach K (1997) Molecular imprinting utilizing an amide functional group for hydrogen bonding leading to highly efficient polymers. J Org Chem 62:4057–4064
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Papaioannou, E., Koutsas, C. & Liakopoulou-Kyriakides, M. Molecularly imprinted polymers for RGD selective recognition and separation. Amino Acids 36, 563–569 (2009). https://doi.org/10.1007/s00726-008-0118-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-008-0118-6