Abstract
Nuclear Magnetic Resonance (NMR) and Electron Paramagnetic Resonance (EPR) discovered around the mid-twentieth century became two of the fastest evolving spectroscopic techniques with applications starting in physics and slowly and steadily developing to play very important roles in structural organic chemistry, bio-molecular structure, solid state, transition metal compounds, diagnostic radiology, medicine and almost any field. NMR has been developing relatively faster than EPR due to reasons of large differences in their dynamics in the respective time scales. With the development of Fourier Transform (FT)-NMR and diagnostic imaging with MRI, NMR grew very fast to be an invaluable technique for research in structural biology and medicine. This mind-boggling speed of the development in the field of NMR kindled the efforts in the development of FT-EPR imaging attempts as well. With relentless efforts supported by the developments in electronics, signal processing, fast switches and, especially, synthetic narrow-line free electron spin probes, we are now in a position to routinely generate fast in vivo EPR images of small animals using time-domain EPR. EPR imaging holds a unique promise of quantitatively mapping the in vivo tissue oxygen distribution non-invasively. Many tumors are characterized by a hypoxic core that is highly resistant to radiation and chemotherapeutic treatment. EPRI enables fast quantitative non-invasive assessment and monitoring of tumor hypoxia based on the relaxivity, both T1 and T2, by the intrinsic concentration of molecular oxygen (pO2). The present article describes the development of radiofrequency Time-domain EPR imaging at the National Cancer Institute, National Institutes of Health, Department of Health and Human Services, USA. Some representative examples are also included. The International Zavoisky Award (by the Zavoisky Physical-Technical Institute in Kazan, the Kazan Federal University, the Government of the Republic of Tatarstan, and the Springer-Verlag) was bestowed on the author in October, 2022 distinguished in recognition of his pioneering contributions to FT-EPR imaging for non-invasive quantitative tumor oximetry in cancer research.


















Similar content being viewed by others
Availability of Data and Materials
Not applicable.
References
P.C. Lauterbur, Image formation by induced local interactions: examples employing nuclear magnetic resonance. Nature 242, 190–191 (1973)
P. Mansfield, Multi-planar image formation using NMR spin echoes. J. Phys. C10, L55–L58 (1977)
J. Radon, P.C. Parks, On the determination of functions from their integral values along certain manifolds IEEE transactions on medical imaging. IEEE Trans. Med. Imaging 5, 170–176 (1986)
D. Kumar, R.R. Welti, Ernst NMR fourier zeugmatography. J. Magn. Reson. 213, 495–509 (2011)
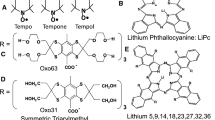
K.J. Liu, P. Gast, M. Moussavi, S.W. Norby, N. Vahidi, T. Walzak, M. Wu, H.M. Swartz, Lithium phthalocyanine: a probe for electron paramagnetic resonance oximetry in viable biologic systems. Proc. Natl. Acad. Sci. U. S. A. 90, 5438–5442 (1993)
A. Manivannan, H. Yanagi, G. Ilangovan, P. Kuppusamy, Lithium naphthalocyanine as a new molecular radical probe for electron paramagnetic resonance oximetry. J. Magn. Magn. Mater. 233(3), L131–L135 (2001)
S. Pfenninger, W. Froncisz, J. Forrer, J. Luglio, J.S. Hyde, General-method for adjusting the quality factor of EPR resonators. Rev. Sci. Instrum. 66(10), 4857–4865 (1995)
R. Murugesan, M. Afeworki, J.A. Cook, N. Devasahayam, R. Tschudin, J.B. Mitchell, S. Subramanian, M.C. Krishna, A broadband pulsed radio frequency electron paramagnetic resonance spectrometer for biological applications. Rev. Sci. Instrum. 69(4), 1869–1876 (1998)
Rinard, G. A., Eaton, S. S., Eaton, G. R., Pool, Jr., C. P. and Farach, H. A. (1999) Handbook of Electron Spin resonance, Poole, Jr., C. P. and Farach, H. A., eds. vol 2, API Press, 1–23.
G.A. Rinard, R.W. Quine, S.S. Eaton, G.R. Eaton, W. Froncisz, Relative benefits of overcoupled resobators vs inherently low-Q resonators for pulsed magnetic resonance. J. Magn. Reson. Series A. 108, 71–81 (1994)
J.H. Ardenkjaer-Larsen, I. Laursen, I. Leunbach, G.J. Ehnholm, EPR and DNP properties of certain novel single electroncontrast agents intended for oximetric imaging. J. Magn. Reson. 133, 1–12 (1998)
K. Golman, I. Leunbach, J.H. Ardenkjaer-Larsen, G.J. Ehnholm, L.G. Wistrand, J.S. Petersson, A. Jarvi, S. Vahasalo, Overhauser-enhanced MR imaging (OMRI). Acta Radiol. 39(1), 10–17 (1998)
K.I. Matsumoto, S. Subramanian, R. Murugesan, J.B. Mitchell, M.C. Krishna, Spatially resolved biologic information from in vivo EPRI, OMRI, and MRI. Antioxid. Redox Signal. 9(8), 1125–1141 (2007)
N. Devasahayam, S. Subramanian, R. Murugesan, J.A. Cook, M. Afeworki, R.G. Tschudin, J.B. Mitchell, M.C. Krishna, Parallel coil resonators for time-domain radiofrequency electron paramagnetic resonance imaging of biological objects. J. Magn. Reson. 142(1), 168–176 (2000)
S. Subramanian, J.W. Koscielniak, N. Devasahayam, R.H. Pursley, T.J. Pohida, M.C. Krishna, A new strategy for fast radiofrequency CW EPR imaging: direct detection with rapid scan and rotating gradients. J. Magn. Reson. 186(2), 212–219 (2007)
S. Subramanian, N. Devasahayam, R. Murugesan, K. Yamada, J. Cook, A. Taube, J.B. Mitchell, J.A.B. Lohman, M.C. Krishna, Single-point (constant-time) imaging in radiofrequency Fourier transform electron paramagnetic resonance. Magn. Reason. Med. 48, 370–379 (2002)
A. Matsumoto, S. Matsumoto, A. Sowers, P. Kuppusomy, J. Mitchell, S. Subramanian, M. Krishna, K.I. Matsumoto, Assessment of absolute oxygen concentration in normal mouse tissues using low field magnetic resonance spectroscopy. Free Radic. Biol. Med. 37, S184–S184 (2004)
A. Matsumoto, S. Matsumoto, A.L. Sowers, J.W. Koscielniak, N.J. Trigg, P. Kuppusamy, J.B. Mitchell, S. Subramanian, M.C. Krishna, K. Matsumoto, Absolute oxygen tension (pO(2)) muscle tissue as determined by in murine fatty and EPR. Magn. Reson. Med. 54, 1530–1535 (2005)
R.H. Pursley, G. Salem, N. Devasahayam, S. Subramanian, J. Koscielniak, M.C. Krishna, T.J. Pohida, Integration of digital signal processing technologies with pulsed electron paramagnetic resonance imaging. J. Magn. Reson. 178, 220–227 (2006)
S Subramanian and M C Krishna, Reference module in chemistry,molecular sciences and chemical engineering, CMSE:05870 In Vivo Electron Paramagnetic Resonance Imaging, and references therein.
R. Czoch, A. Francik, J. Indyka, J. Koscielniak, EPR spectrometer with rapid scan. Meas. Automatic Contr. 29, 41–43 (1983)
Hornak, J.P. The Basics of MRI; J.P. Hornak, 1996–2011; http://www.cis.rit.edu/htbooks/mri/.
G.A. Rinard, R.W. Quine, B.T. Ghim, S.S. Eaton, G.R. Eaton, Easily tunable crossed-loop (bimodal) EPR resonator. J. Magn. Reson. Ser. A. 122(1), 50–57 (1996)
G.A. Rinard, R.W. Quine, L.A. Buchanan et al., Resonators for in vivo imaging: practical experience. Appl Magn Reson 48, 1227–1247 (2017)
V.S. Subramanian, E. Boris, H.J. Halpern, Orthogonal resonators for pulse in vivo electron paramagnetic imaging at 250 MHz. J. Magn. Reson. 240, 45–51 (2014)
D.I. Hoult, R.E. Richards, Critical factors in the design of sensitive high resolution nuclear magnetic resonance spectrometers. Proc. R. Soc. Lond. A 344, 311–340 (1975)
S. Emid, J.H.N. Creyghton, High-resolution NMR imaging in solids. Physica 128B, 81–83 (1985)
G.G. Maresch, M. Mehring, S. Emid, High resolution ESR imaging. Physica 138B, 261–263 (1986)
M. Lustig, D. Donoho, J.M. Pauly, Sparse MRI: the application of compressed sensing for rapid MR imaging. Magn. Reson. Med. 58, 1182–1195 (2007)
B. Epel, M.K. Bowman, C. Mailer, H.J. Halpern, Absolute oxygen R1e imaging in vivo with pulse electron paramagnetic resonance. Magn. Reson. Med. 72, 362–368 (2014)
E.L. Hahn, Spin echoes. Phys. Rev. 80(4), 580–594 (1950)
G.R. Eaton, S.S. Eaton, M.M. Maltempo, 3 approaches to spectral spatial EPR imaging. Appl. Radiat. Isot. 40(10–12), 1227–1231 (1989)
S.S. Eaton, M.M. Maltempo, E.D.A. Stemp, G.R. Eaton, Three-dimensional EPR imaging with one spectral and two spatial dimensions. Chem. Phys. Lett. 142, 567–569 (1987)
M.M. Maltempo, S.S. Eaton, G.R. Eaton, Reconstruction of spectral-spatial two dimensional images from incomplete sets of projections without prior knowledge of the component spectra. J. Magn. Reson. 77, 75–83 (1988)
K. Ohno, M. Watanabe, Electron paramagnetic resonance imaging using magnetic-field-gradient spinning. J. Magn. Reson. 2000(143), 274–279 (2000)
Y.M. Deng, G.L. He, S. Petryakov, P. Kuppusamy, J.L. Zweier, Fast EPR imaging at 300 MHz using spinning magnetic field gradients. J. Magn. Reson. Imaging 168, 220–227 (2004)
Acknowledgements
This research was supported by the Intramural Research Program of the National Institute of Health at the Center for Cancer Research, where the work described herein was carried out over a period of 15 years. Several visiting fellows and post-doctoral fellows were involved, too numerous to thank individually, and we thank all of them. The instrumentation help throughout the course of development from Mr. Nallathambi Devasahayam, Rolf Tschudin, Tom Pohida, Randal Pursley, Salem Ghadi, and Janusz Koscielniak is gratefully acknowledged. Special thanks go to Gadisetti Chandramouli for help with image processing and computational software help. The very idea of initiating and developing RF FT-EPR imaging at NIH was due to the insatiable interest and encouragement initiated by Angelo Russo and James Mitchell of the Radiation Biology Branch, NCI, NIH, and the continuous support and encouragement from the Biophysical Spectroscopy and Imaging Section chief Murali Krishna Cherukuri, whom, incidentally, I had the pleasure of mentoring for his Ph. D program at the Indian Institute of Technology. Madras, India some 40 years ago. Last but not the least, I place on record the unparalleled guidance of my Ph. D. mentor Late Professor Martyn Christian Raymond Symons, University of Leicester, UK, whose dream was to develop an EPR imager for small animals leading to applications to biology and medicine. I wish he was alive when I was selected for the 2022 International Zavoisky Award. I place on record my sincere appreciation and thanks to Professor Kev Salikhov and Dr. Laila Mosina of the Zavoisky Physical-Technical Institute, FRC Kazan Scientific Center of Russian Academy of Sciences and the International Zavoisky Award Committee for help and guidance throughout during the preparation of this manuscript. And above all, I place on record my hearty gratitude to my better-half Rajalakshmi Subramanian, who has been such an angel for more than half a century bestowing me with love, care gourmet food, and timely counsel, keeping me always happy and relaxed.
Funding
This research was supported by the Intramural Research Program of the National Institutes of Health, Bethesda, MD, USA at the Center for Cancer Research, where part of the work covering CW and pulsed EPR imaging that has been described in this chapter was developed.
Author information
Authors and Affiliations
Contributions
Not applicable (single author).
Corresponding author
Ethics declarations
Conflict of interest
Nil.
Ethical Approval
Female C3H mice produced by the National Cancer Institute Animal Production Area (Frederick, MD) were used for the study. The mice were 6–8 weeks of age at the time of experiment and weighed between 20 and 30 g. EPR imaging experiments were carried out in compliance with the Guide for the Care and Use of Laboratory Animal Resources, (1996) National Research Council, and approved by the National Cancer Institute, NIH, Bethesda, MD, USA.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Subramanian, S. The Development of In Vivo FT-EPR Imaging at 300 MHz: Applications in Cancer Research. Appl Magn Reson 54, 807–832 (2023). https://doi.org/10.1007/s00723-023-01563-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00723-023-01563-6