Abstract
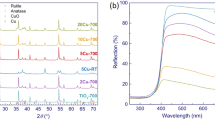
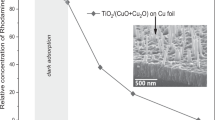
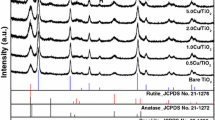
High surface area TiO2 was synthetized by means of a solvothermal method and it was then used to prepare two photocatalysts based on copper-modified TiO2. Two different preparation approaches have been adopted based on the use of the same amount of Cu2O (0.5% w/w) as a reactant. The spectroscopic characterization shows that the two preparation procedures lead to distinct distributions of Cu2O and other Cu-based species in the final composite materials and that the photoactivity of the solids is strictly related to the nature of the copper species. The joint application of CW-EPR and optical spectroscopy, both employed in catalytic conditions as a sort of “operando” mode, allowed monitoring the evolution of the photocatalytic systems occurring during the H2 evolution reaction (HER). The effect of water coordination on the Cu(II) species at the surface of titania has been revealed by EPR. The successive photoreduction of the system in the early steps of the photocatalytic reaction involves the reduction of Cu2+, the formation of Ti3+ centres in the oxide matrix and that of metallic copper particles that partially inactivate the system.











Similar content being viewed by others
References
Z. Wang, C. Li, K. Domen, Chem. Soc. Rev. 48, 2109–2125 (2019)
G.L. Chiarello, M.V. Dozzi, E. Selli, J. Energy Chem. 26, 250–258 (2017)
D. Barreca, G. Carraro, V. Gombac, A. Gasparotto, C. Maccato, P. Fornasiero, E. Tondello, Adv. Funct. Mater. 21, 2611–2623 (2011)
V. Polliotto, S. Livraghi, A. Krukowska, M.V. Dozzi, A. Zaleska-Medynska, E. Selli, E. Giamello, A.C.S. Appl, Mater. Interfaces 10, 27745–27756 (2018)
C.Y. Toe, Z. Zheng, H. Wu, J. Scott, R. Amal, Y.H. Ng, Angew. Chem. Int. Ed. 57, 13613–13617 (2018)
X. Chang, T. Wang, P. Zhang, Y. Wei, J. Zhao, J. Gong, Angew. Chem. Int. Ed. 55, 8840–8845 (2016)
Y.-H. Zhang, Y.-L. Li, B.-B. Jiu, F.-L. Gong, J. Chen, S. Fang, H. Zhang, Nanotechnology 30, 145401 (2019)
Z. Zhang, R. Dua, L. Zhang, H. Zhu, H. Zhang, P. Wang, ACS Nano 7, 1709–1717 (2013)
M.E. Aguirre, R. Zhou, A.J. Eugene, M.I. Guzman, M.A. Grela, Appl. Catal B: Environ. 217, 485–493 (2017)
K.-H. Kim, S.-K. Ihm, J. Hazard. Mater. 146, 610–616 (2007)
V. Gombac, L. Sordelli, T. Montini, J.J. Delgado, A. Adamski, G. Adami, M. Cargnello, S. Bernal, P. Fornasiero, J. Phys. Chem. A 114, 3916–3925 (2010)
I. Tamiolakis, I.T. Papadas, K.C. Spyridopoulos, G. S. Armatas RSC Adv. 6, 54848 (2016)
G. Li, J. Huang, Z. Deng, J. Chen, Q. Huang, Z. Liu, W. Guo, R. Cao, Cryst. Growth Des. 19, 5784–5790 (2019)
S. Hejazi, S. Mohajernia, Y. Wu, P. Andryskova, G. Zoppellaro, I. Hwang, O. Tomanec, R. Zboril, P. Schmuki, Electrochem. Commun. 98, 82–86 (2019)
S. Banerjee, D. Chakravorty, Europhys. Lett. 52(4), 468–473 (2000)
A. Kellersohn, E. Kniizinger, W. Langel, M. Gievsig, Adv. Mater. 7, 652–655 (1995)
W. Cui, W. An, L. Liu, J. Hu, Y. Lian, J. Hazard. Mater. 280, 417–427 (2014)
X. Zou, H. Fan, Y. Tian, M. Zhang, X. Yan, RSC Adv. 5, 23401–23409 (2015)
I.L. Soroka, A. Shchukarev, M. Jonsson, N.V. Tarakina, P.A. Korzhavyi, Dalton Trans. 42, 9585–9594 (2013)
M. Bennati, D.M. Murphy, in Electron paramagnetic resonance: a practitioner toolkit, ed. by M. Brustolon, E. Giamello (Wiley, USA, 2009)
G. Li, N.M. Dimitrijevic, L. Chen, T. Rajh, K.A. Gray, J. Phys. Chem. C 112, 19040–19044 (2008)
Y. Nosaka, S. Takahashi, H. Sakamoto, A.Y. Nosaka, J. Phys. Chem. C 115, 21283–21290 (2011)
M.V. Dozzi, G.L. Chiarello, M. Pedroni, S. Livraghi, E. Giamello, E. Selli, Appl. Catal. B: Environ. 209, 417–428 (2017)
W. Burton Lewis, M. Alei Jr., L.O. Morgan, J. Chem. Phys. 45, 4003 (1966)
L. Calabrese, J. Rotilio, in Structure and Function of Haemocyanin, ed. by J.V. Bannister (Springer, Berlin, Heidelberg, New York, 1977) pp. 180–184
G.T. Palomino, P. Fisicaro, S. Bordiga, A. Zecchina, E. Giamello, C. Lamberti, J. Phys. Chem. B 104, 4064–4073 (2000)
J. Biedrzycki, S. Livraghi, E. Giamello, S. Agnoli, G. Granozzi, J. Phys. Chem. C 118, 8462–8473 (2014)
F. Parveen, B. Sannakki, M.V. Mandke, H.M. Pathan, Sol. Energy Mater. Sol. Cells 144, 371–382 (2016)
M. Singh, I. Sinha, M. Premkumar, A.K. Singh, R.K. Mandal, Colloids Surf. A 359(1), 88–94 (2010)
Acknowledgements
The FEG-SEM S9000 by Tescan was purchased with funds from Regione Piemonte (project POR FESR 2014-20 INFRA-P SAX).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zollo, A., Polliotto, V., Livraghi, S. et al. Self-Organisation of Copper Species at the Surface of Cu–TiO2 Systems During H2 Evolution Reaction: A Combined Investigation by EPR and Optical Spectroscopy. Appl Magn Reson 51, 1497–1513 (2020). https://doi.org/10.1007/s00723-020-01226-w
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00723-020-01226-w