Abstract
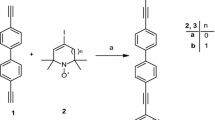
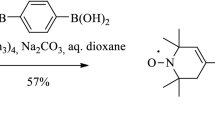
New nitroxide biradicals 15NR6–C≡C–(p-C6H4)2–C≡C–14NR6 (B2) and 15NR6–C≡C–(p-C6H4)2–C≡C–15NR6 (B3), where R6 denotes 1-oxyl-2,2,6,6-tetramethyl–1,2,3,6-tetrahydropyridine ring, are synthesized and studied in liquid solutions by continuous-wave electron paramagnetic resonance (EPR) spectroscopy. Hyperfine interaction constants and exchange integrals are determined from the simulation and fitting of experimental spectra and calculated ones. The best fitting of the EPR spectra detected in experiments and their temperature dependence was achieved under the assumption that the biradicals perform transitions between conformations with different exchange integrals. The conformation of about 75% of biradicals has the exchange integral |\(J\)| = 4.4 G. Thus, the ground electronic state of these biradicals has the conformation with |\(J\)| = 4.4 G. About 20% of biradicals have the conformation with the zero exchange integral. A minor fraction of biradicals have conformations with |\(J\)| = 11.2 G and |\(J\)| = 57 G. The activation energy for the transition from the conformation with |\(J\)| = 4.4 G to the conformation with \(J\) = 0 is 8.5 kcal/mol, while that for the reverse reaction is 1.7 kcal/mol. It is shown that the isotope substitution provides a valuable resource in the EPR studies of the exchange interaction in biradicals.





Similar content being viewed by others
References
V.N. Parmon, A.I. Kokorin, G.M. Zhidomirov, Russ. J. Struct. Chem. 18, 104 (1977)
E.G. Rozantsev, Free Nitroxyl Radicals (Plenum Press, New York, 1970)
L.B. Volodarsky (ed.), Nitroxide. Synthesis, Properties, Applications, 1st–2 edn. (CRC Press, Boca Raton, 1988)
R.I. Zhdanov (ed.), Bioactive Spin Labels (Springer, Berlin, 1992)
A. Rassat, Pure Appl. Chem. 62, 223 (1990)
M.T. Lemaire, Pure Appl. Chem. 76, 277 (2004)
M. Abe, Chem. Rev. 113, 7011 (2013)
Yu.N. Molin, K.M. Salikhov, K.I. Zamaraev, Spin Exchange (Springer, Berlin, 1980)
B.D. Armstrong, S. Han, J. Chem. Phys. 127, 1045081 (2007)
M.J. Prandolini, V.P. Denysenko, M. Gafurov, S. Lyubenova, B. Endeward, M. Bennati, T.E. Prisner, Appl. Magn. Reson. 34, 399 (2008)
K. Sato, S. Nakazawa, R. Rahimi, T. Ise, S. Nishida, T. Yoshino, N. Mori, K. Toyota, D. Shiomi, Y. Yakiyama, Y. Morita, M. Kitagawa, K. Nakasuji, M. Nakahara, H. Hara, P. Carl, P. Höfer, T. Takui, J. Mater. Chem. 19, 3739 (2009)
A.B. Shapiro, M.G. Goldfield, E.G. Rozantsev, Tetrahedron Lett. 14, 2183 (1973)
V.V. Pavlikov, V.V. Muraviev, A.B. Shapiro, Izv. AN SSSR Ser. Khim 5, 1200 (1980)
S. Torii, T. Hase, M. Kuroboshi, C. Amatore, A. Jutand, H. Kawafuchi, Tetrahedron Lett. 38, 7391 (1997)
A.I. Kokorin, Appl. Magn. Reson. 26, 253 (2004)
A.I. Kokorin, V.V. Pavlikov, A.B. Shapiro, Proc. Acad. Sci. Phys. Chem. 253, 147 (1980)
A.B. Shapiro, V.N. Parmon, V.V. Pavlikov, V.I. Rubtsov, E.G. Rozantsev, Izv. AN SSSR Ser. Khim. 2, 449 (1980)
A.I. Kokorin, V.A. Tran, K. Rasmussen, G. Grampp, Appl. Magn. Reson. 30, 35 (2006)
V.A. Tran, A.I. Kokorin, G. Grampp, K. Rasmussen, Appl. Magn. Reson. 35, 389 (2009)
A.I. Kokorin, E.N. Golubeva, B. Mladenova, V.A. Tran, T. Kálai, K. Hideg, G. Grampp, Appl. Magn. Reson. 44, 1041 (2013)
S.Y. Umanskiy, E.N. Golubeva, B.N. Plakhutin, Russ. Chem. Bull. 62, 1511 (2013)
A.I. Kokorin, O.I. Gromov, T. Kálai, K. Hideg, Appl. Magn. Reson. 47, 1283 (2016)
A.I. Kokorin, R.B. Zaripov, O.I. Gromov, A.A. Sukhanov, T. Kálai, E. Lamperth, K. Hideg, Appl. Magn. Reson. 47, 1057 (2016)
A.I. Kokorin, R.B. Zaripov, O.I. Gromov, K. Hideg, T. Kálai, Appl. Magn. Reson. 49, 137 (2018)
L.A. Shundrin, I.A. Kirilyuk, I.A. Grigor’ev, Mendeleev Commun. 24, 298 (2014)
T. Kálai, J. Jeko, Z. Berente, K. Hideg, Synthesis 38, 439 (2006)
J.A. Riddick, W.B. Bunger, K.T. Sakano, Techniques of Chemistry: Organic Solvents, Physical Chemistry and Methods of Purification, 2nd edn. (Wiley, New York, 1986), p. 785
Ya.S. Lebedev, O.Ya. Grinberg, A.A. Dubinsky, O.G. Poluektov, in Bioactive Spin Labels, ed. by R.I. Zhdanov (Springer, Berlin, 1992), p. 227
A.M. Portis, Phys. Rev. 97, 1071 (1953)
Acknowledgements
Authors T. Kálai and K. Kish (Pécs University) gratefully acknowledge the support of the grant GINOP-2.3.2-15-2016-00049.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zaripov, R.B., Khairutdinov, I.T., Kálai, T. et al. Potential of Isotope Substitution in EPR Studies of Nitroxide Biradicals. Appl Magn Reson 51, 523–543 (2020). https://doi.org/10.1007/s00723-020-01199-w
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00723-020-01199-w