Abstract
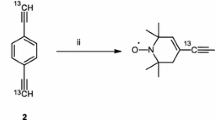
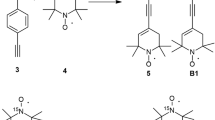
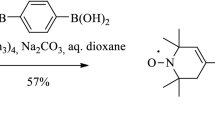
A specially synthesized nitroxide biradical R5-C≡13C-(p-C6H4)2-13C≡C-R5 (B4) and two radicals, R5-C≡13CH (RCC) and R5-C≡13C-C6H5 (RCCPh), where R5 is 1-oxyl-2,2,5,5-tetramethyl-pyrroline group, have been studied by X- and W-band electron paramagnetic resonance (EPR) spectroscopy, and by W-band electron-nuclear double resonance (ENDOR). Spin density distribution and hyperfine splitting (hfs) constants on 13C atoms were experimentally determined and also calculated using ORCA 3.0.3 program package. The biradical and radicals geometries were optimized on UKS/B3LYP/cc-pVDZ level. Hfs constants were calculated using density functional theory (DFT) with PBE0 functional and N07D, and were compared with the experimental value of the hfs constant on 13C atoms, measured from ENDOR spectra. It is concluded that at small values of the exchange integral as J ≤ a/2 ≈ 7–8 G, the current quantum chemical approaches do not allow determining precise values of the hfs constants on the 13C atoms in the bridge connecting two paramagnetic nitroxide rings of the biradical.






Similar content being viewed by others
References
L.J. Berliner (ed.), Spin Labeling. Theory and Applications (Academic Press, New-York, 1976)
G.I. Likhtenshtein, Spin Labeling Methods in Molecular Biology (Wiley, New York, 1976)
A.M. Wasserman, A.L. Kovarsky, Spin Labels and Pprobes in Physical Chemistry of Polymers (Nauka, Moscow, 1986)
Imidazoline Nitroxides. Synthesis, Properties, Applications, vol. 1, 2, ed. by L.B. Volodarsky (CRC Press, Boca Raton, 1988)
S.S. Eaton, G.R. Eaton, Electron Paramagnetic Resonance, vol. 19, p. 318 (2004)
G. Likhtenshtein, J. Yamauchi, S. Nakatsuji, A.I. Smirnov, R. Tamura, Nitroxides: Applications in Chemistry, Biomedicine, and Materials Science (Wiley, New York, 2008)
G.I. Likhtenshtein, Electron Spin Interactions in Chemistry and Biology (Springer, Wien, 2016)
H.P. Nguyen, A.M. Popova, K. Hideg, P.Z. Qin, BMC Biophys. 8, 6 (2015)
C. Altenbach, C.J. López, K. Hideg, W.L. Hubbell, Methods Enzymol. 564, 59 (2015)
S.M. Winter, S. Hill, R.T. Oakley, J. Am. Chem. Soc. 137, 3720 (2015)
A. Rajca, Y. Wang, M. Boska, J.T. Paletta, A. Olankitwanit, M.A. Swanson, D.G. Mitchell, S.S. Eaton, G.R. Eaton, S. Rajca, J. Am. Chem. Soc. 134, 15724 (2012)
T. Janoschka, N. Martin, U. Martin, C. Friebe, S. Morgenstern, H. Hiller, M.D. Hager, U.S. Schubert, Nature 527, 78 (2015)
M. Rafiee, K.C. Miles, S.S. Stahl, J. Am. Chem. Soc. 137, 14751 (2015)
M.Y. Zaremski, Polymer Sci., Ser. C 57, 65 (2015)
F. Mentink-Vigier, U. Akbey, H. Oschkinat, S. Vega, A.J. Feintuch, J. Magn. Reson. 258, 102 (2015)
C. Sauvée, M. Rosai, G. Casano, F. Aussennc, T.R. Weber, R. Ouari, P. Tordo, Angew. Chemie. Int. Ed. 52, 10858 (2013)
E.G. Rozantsev, Free Nitroxyl Radicals (Plenum Press, New York, 1970)
V.N. Parmon, A.I. Kokorin, G.M. Zhidomirov, Stable Biradicals (Nauka, Moscow, 1980)
V.V. Pavlikov, A.B. Shapiro, E.G. Rozantsev, Izv. AN SSSR Ser. Khim. 1, 128 (1980)
A.I. Kokorin, V.V. Pavlikov, A.B. Shapiro, Proc. Acad. Sci. USSR Doklady Phys. Chem. 253, 525 (1980)
S. Torii, T. Hase, M. Kuroboshi, C. Amatore, A. Jutand, H. Kawafuchi, Tetrahedron Lett. 38(42), 7391 (1997)
A.I. Kokorin, Appl. Magn. Reson. 26, 253 (2004)
A.I. Kokorin, V.A. Tran, K. Rasmussen, G. Grampp, Appl. Magn. Reson. 30, 35 (2006)
A.I. Kokorin, E.N. Golubeva, B.Y. Mladenova, V.A. Tran, T. Kálai, K. Hideg, G. Grampp, Appl. Magn. Reson. 44, 1041 (2013)
O.I. Gromov, E.N. Golubeva, V.N. Khrustalev, T. Kálai, K. Hideg, A.I. Kokorin, Appl. Magn. Reson. 45, 981 (2014)
A.I. Kokorin, O.I. Gromov, T. Kálai, K. Hideg, Appl. Magn. Reson. 47, 1283 (2016)
A.I. Kokorin, R.B. Zaripov, O.I. Gromov, A.A. Sukhanov, T. Kálai, É. Lamperth, K. Hideg, Appl. Magn. Reson. 47, 1057 (2016)
R.P. Shibaeva, Zh. Strukt. Khimii 16, 330 (1975)
V.N. Parmon, G.M. Zhidomirov, Mol. Phys. 27, 367 (1974)
K. Hideg, H.O. Hankovszky, L. Lex, Gy. Kulcsár, Synthesis, 911 (1980)
K. Hideg, J. Csekő, H.O. Hankovszky, P. Sohár, Can. J. Chem. 64, 1482 (1986)
T. Kálai, M. Balog, J.K. Hideg, Synthesis, 973 (1999)
T. Kálai, M. Balog, J. Jekő, W.L. Hubbell, K. Hideg, Synthesis, 2365 (2002)
J.A. Riddick, W.B. Bunger, K.T. Sakano, in Organic Solvents: Physical Properties and Methods of Purification, vol. 2, 4th ed. (Wiley, New York, 1986) Chap IV, p. 785
YuN Molin, K.M. Salikhov, K.I. Zamaraev, Spin Exchange (Springer, Berlin, 1980)
A.I. Kokorin, V.N. Parmon, A.A. Shubin, Atlas of Anisotropic EPR Spectra of Nitroxide Biradicals (Nauka, Moscow, 1984)
S.H. Glarum, J.H. Marshall, J. Chem. Phys. 47, 1374 (1967)
H. Lemaire, J. Chim. Phys. 64, 559 (1967)
Ya.S. Lebedev, O.Ya. Grinberg, A.A. Dubinsky, O.G. Poluektov, in Bioactive Spin Labels, ed. by R.I. Zhdanov (Springer, Berlin, 1992), pp. 228–254
E.L. Hahn, Phys. Rev. 80, 580 (1950)
A. Schweiger, G. Jeschke, Principles of Pulse Electron Paramagnetic Resonance (Clarendon Press, Oxford, 2001)
W.B. Mims, Proc. R. Soc. Lond. 283, 452 (1965)
F. Neese, Wiley Interdiscip. Rev. Comput. Mol. Sci. 2, 73 (2012)
V. Barone, P. Cimino, E. Stendardo, J. Chem. Theory Comput. 4, 751 (2008)
S. Sinnecker, A. Rajendran, A. Klamt, M. Diedenhofen, F. Neese, J. Phys. Chem. A 110, 2235 (2006)
F. Mentink-Vigier, A. Collauto, A. Feintuch, I. Kaminker, V. Tarle, D. Goldfarb, J. Magn. Reson. 236, 117 (2013)
A. Nalepa, K. Möbius, W. Lubitz, A. Savitsky, J. Magn. Reson. 242, 203 (2014)
S. Stoll, A. Schweiger, J. Magn. Reson. 78, 42 (2006)
V. Sadovnichy, A. Tikhonravov, Vl. Voevodin, V. Opanasenko, in Contemporary High Performance Computing: From Petascale toward Exascale (Chapman and Hall/CRC Computational Science, CRC Press, Boca Raton, 2013), pp. 283–307
Acknowledgements
This work was supported by the Grants GINOP 2.3.2-15-2016-00049 and GINOP-2.3.2-15-2016-00025. The present scientific contribution is dedicated to the 650th anniversary of the foundation of the University of Pécs, Hungary. The study was also supported by the Supercomputing Center of M. V. Lomonosov Moscow State University [49], and through the Program of Fundamental Research of the Presidium of the Russian Academy of Sciences (no. 1.26П). AIK also thanks Dr. A. A. Shubin (Boreskov Institute of Catalysis, Siberian Branch, Russian Academy of Sciences, Novosibirsk) who provided us with his computer program package of EPR spectra simulation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kokorin, A.I., Zaripov, R.B., Gromov, O.I. et al. Tailored Nitroxide Radicals and Biradical Containing 13C Enriched Acetylene Groups: ENDOR and DFT Investigation. Appl Magn Reson 49, 137–149 (2018). https://doi.org/10.1007/s00723-017-0942-5
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00723-017-0942-5