Summary
Background
To assess changes in corneal biomechanical parameters measured by the Ocular Response Analyzer device (ORA, Reichert Inc., Depew, NY, USA) before and on the first day and the first week following intravitreal aflibercept (IVA) injection.
Methods
In this retrospective cross-sectional study, intraocular pressure (Goldmann applanation tonometry, IOPgat), ORA measurements (Goldmann-correlated intraocular pressure [IOPg], corneal-compensated IOP [IOPcc], corneal resistance factor [CRF], and corneal hysteresis) were made. Also, pachymetry and ocular biometric examination (anterior chamber depth, axial length, and lens thickness) were performed. Data before and on the first day and the first week after IVA injection were compared.
Results
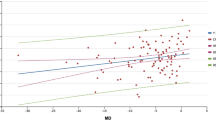
In total, 51 patients (20 female [39.2%] and 31 male [60.8%]) were enrolled in the study. The mean age was 69.51 ± 8.43 years. Indication for IVA treatment was age-related macular degeneration in 22 patients (43.1%) and retinal vein occlusion in 29 patients (56.9%). No significant differences were observed following IVA in terms of central corneal thickness (p = 0.40), axial length (p = 0.80), anterior chamber depth (p = 0.69), and lens thickness (p = 0.49). IOPgat significantly reduced on the first day after IVA (13.3 mm Hg to 11.8 mm Hg; p < 0.001). A decrease in CRF was observed on the first day following IVA (9.94 to 9.38; p = 0.03). Furthermore, CRF and IOPgat were positively correlated at baseline and on the first day and the first week after IVA.
Conclusion
Intravitreal aflibercept treatment induces corneal biomechanical alterations that might be associated with IOP change following IVA injection.
Zusammenfassung
Hintergrund
Ziel war, die Veränderungen der biomechanischen Parameter der Hornhaut zu beurteilen, die mit dem Ocular-Response-Analyzer-Gerät (ORA, Fa. Reichert Inc., Depew/NY, USA) vor und am ersten Tag und in der ersten Woche nach der intravitrealen Injektion von Aflibercept (IVA) gemessen wurden.
Methodik
In dieser retrospektiven Querschnittstudie wurden Augeninnendruck (Goldmann-Applanationstonometrie, IOPgat), ORA-Messungen (Goldmann-korrelierter Augeninnendruck [IOPg], Hornhaut-kompensierter Augeninnendruck [IOPcc], Hornhautwiderstandsfaktor und Hornhauthysterese) durchgeführt. Außerdem wurden eine Pachymetrie und eine okuläre biometrische Untersuchung (Vorderkammertiefe, axiale Länge und Linsendicke) durchgeführt. Daten vor und an dem ersten Tag und in der ersten Woche nach der IVA-Injektion wurden verglichen.
Ergebnisse
Insgesamt wurden 51 Patient*innen (20 Frauen [39,2 %] und 31 Männer [60,8 %]) in die Studie aufgenommen. Das Durchschnittsalter betrug 69,51 ± 8,43 Jahre. Indikation zur IVA-Behandlung war die altersbedingte Makuladegeneration bei 22 Patient*innen (43,1 %) und der retinale Venenverschluss bei 29 Patient*innen (56,9 %). Nach der IVA wurden keine signifikanten Unterschiede in Bezug auf die zentrale Hornhautdicke (p = 0,40), die axiale Länge (p = 0,80), die Vorderkammertiefe (p = 0,69) und die Linsendicke (p = 0,49) beobachtet. Am ersten Tag nach IVA war der IOPgat signifikant vermindert (von 13,3 mm Hg auf 11,8 mm Hg; p < 0,001). Auch der Hornhautwiderstandsfaktor zeigte am ersten Tag nach IVA eine Abnahme (von 9,94 auf 9,38; p = 0,03). CRF und IOPgat waren zu Beginn der Studie sowie am ersten Tag und in der ersten Woche nach IVA positiv korreliert.
Schlussfolgerung
Die intravitreale Aflibercept-Behandlung induziert biomechanische Veränderungen der Hornhaut, die mit einer IOP-Änderung nach einer IVA-Injektion verbunden sein könnten.




Similar content being viewed by others
References
Falavarjani KG, Nguyen QD. Adverse events and complications associated with intravitreal injection of anti-VEGF agents: a review of literature. Eye (Lond). 2013;27:787–94.
Dedania VS, Bakri SJ. Systemic safety of intravitreal anti-vascular endothelial growth factor agents in age-related macular degeneration. Curr Opin Ophthalmol. 2016;27:224–43.
Torres-Costa S, Ramos D, Brandão E, Carneiro Â, Rosas V, Rocha-Sousa A, et al. Incidence of endophthalmitis after intravitreal injection with and without topical antibiotic prophylaxis. Eur J Ophthalmol. 2021;31(2):600–6.
Tolentino M. Systemic and ocular safety of intravitreal anti-VEGF therapies for ocular neovascular disease. Surv Ophthalmol. 2011;56:95–113.
Meyer CH, Michels S, Rodrigues EB, Hager A, Mennel S, Schmidt JC, et al. Incidence of rhegmatogenous retinal detachments after intravitreal antivascular endothelial factor injections. Acta Ophthalmol. 2011;89:70–5.
Baek SU, Park IW, Suh W. Long-term intraocular pressure changes after intravitreal injection of bevacizumab. Cutan Ocul Toxicol. 2016;35:310–4.
Kling S, Hafezi F. Corneal biomechanics—a review. Ophthalmic Physiol Opt. 2017;37:240–52.
Piñero DP, Alcón N. Corneal biomechanics: a review. Clin Exp Optom. 2015;98:107–16.
Shoeibi N, Ansari-Astaneh MR, Sedaghat MR, Shokoohi Rad S. Effect of intravitreal bevacizumab injection on corneal in vivo biomechanics: a pilot study. J Ophthalmic Vis Res. 2019;14:151–6.
Bekmez S, Cakmak H, Kocaturk T, Cantas F, Dundar S. Biomechanical properties of the cornea following intravitreal ranibizumab injection. Graefes Arch Clin Exp Ophthalmol. 2021;259:691–6.
Goktas A, Goktas S, Atas M, Demircan S, Yurtsever Y. Short-term impact of intravitreal ranibizumab injection on axial ocular dimension and intraocular pressure. Cutan Ocul Toxicol. 2013;32:23–6.
Kymionis GD, Giarmoukakis A, Apostolidi IK, Blazaki SV, Tsoulnaras KI, Moschandrea J, et al. Optical biometry derived axial length measurements following Intravitreal anti-vascular endothelial growth factor treatment for macular edema. Semin Ophthalmol. 2018;33:488–91.
Bracha P, Moore NA, Ciulla TA, WuDunn D, Cantor LB. The acute and chronic effects of intravitreal anti-vascular endothelial growth factor injections on intraocular pressure: a review. Surv Ophthalmol. 2018;63:281–95.
Cui QN, Gray IN, Yu Y, VanderBeek BL. Repeated intravitreal injections of antivascular endothelial growth factors and risk of intraocular pressure medication use. Graefes Arch Clin Exp Ophthalmol. 2019;257:1931–9.
Atchison EA, Wood KM, Mattox CG, Barry CN, Lum F, MacCumber MW. The real-world effect of intravitreous anti-vascular endothelial growth factor drugs on intraocular pressure: an analysis using the IRIS registry. Ophthalmology. 2018;125:676–82.
Kahook MY, Ammar DA. In vitro effects of antivascular endothelial growth factors on cultured human trabecular meshwork cells. J Glaucoma. 2010;19:437–41.
Ford KM, Saint-Geniez M, Walshe TE, D’Amore PA. Expression and role of VEGF—a in the ciliary body. Invest Ophthalmol Vis Sci. 2012;53:7520–7.
Hashemi H, Jafarzadehpur E, Mehravaran S, Yekta A, Ostadimoghaddam H, Norouzirad R, et al. Corneal resistance factor and corneal hysteresis in a 6- to 18-year-old population. J Cataract Refract Surg. 2014;40:1446–53.
Broman AT, Congdon NG, Bandeen-Roche K, Quigley HA. Influence of corneal structure, corneal responsiveness, and other ocular parameters on tonometric measurement of intraocular pressure. J Glaucoma. 2007;16:581–8.
Dackowski EK, Moon JY, Wang J, Shrivastava A, Schultz JS. The relationship between corneal biomechanics and Intraocular pressure dynamics in patients undergoing intravitreal injection. J Glaucoma. 2021;30:451–8.
Caruso A, Füth M, Alvarez-Sánchez R, Belli S, Diack C, Maass KF, et al. Ocular half-life of intravitreal biologics in humans and other species: meta-analysis and model-based prediction. Mol Pharm. 2020;17:695–709.
Foster PJ, Broadway DC, Garway-Heath DF, Yip JL, Luben R, Hayat S, et al. Intraocular pressure and corneal biomechanics in an adult British population: the EPIC-Norfolk eye study. Invest Ophthalmol Vis Sci. 2011;52:8179–85.
Terai N, Raiskup F, Haustein M, Pillunat LE, Spoerl E. Identification of biomechanical properties of the cornea: the ocular response analyzer. Curr Eye Res. 2012;37:553–62.
Funding
There is no financial support or grant to disclose.
Funding
No funding is available for this study
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
B.O. Gunay and C.M. Esenulku declare that they have no competing interests.
Ethical standards
Ethical approval was obtained from the same institute review board number: 2019/06. Consent to participate and for publication: Due to the retrospective nature of the study, informed consent was not obtained from the patients.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Availability of data and material
Available on request from Betul Onal Gunay
Rights and permissions
About this article
Cite this article
Gunay, B.O., Esenulku, C.M. Corneal biomechanical assessment via ocular response analyzer following intravitreal aflibercept therapy. Spektrum Augenheilkd. 37, 22–28 (2023). https://doi.org/10.1007/s00717-022-00534-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00717-022-00534-9