Summary
Background
A leading cause for permanent vision loss in diabetic patients with diabetic retinopathy is the occurrence of diabetic macular edema. New treatments, antivascular endothelial growth factors, for diabetic macular edema (DME) have been under trial and found to improve visual acuity more effective than the standard care treatment, central photocoagulation. The purpose of the following study was to estimate the future number of visiting DME patients and their treatment demands at the University Eye-Clinic Graz.
Material and methods
A retrospective 2-year-analysis was performed on all eyes of patients with initial occurrence of diabetic macular edema, visiting the University Eye Clinic Graz between 1 July 2009 and 31 June 2011. Main outcome was to establish a 2-year-incidence for DME at the University Eye Clinic Graz. Further assessments were the distribution of treatments received and the number of intravitreal medication applied.
Results
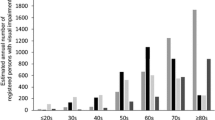
The incidence rate for the first year period was 105 persons and for the second year period 101 persons. Of the 226 treated eyes, 40.1 % received “laser treatment plus bevacizumab injections”, 25.2 % “laser treatment”, 21.1 % “bevacizumab injections” and 13.2 % “additional triamcinolone acetonide injections”. During the 2 years a total number of 343 intravitreal injections were administered, of which 293 were bevacizumab injections with the majority of bevacizumab treated eyes receiving 1–2 injections.
Conclusions
There seems to be a seasonal influence increasing the development of diabetic macular edema. The high treatment demands of DME with foveal involvement have made antivascular endothelial growth factors essential in the treatment of diabetic macular edema.
Zusammenfassung
Hintergrund
Eine Hauptursache für den Sehverlust von Diabetikern mit diabetischer Retinopathie ist das Auftreten eines diabetischen Makulaödems. Neue Therapeutika, antivaskuläre endotheliale Wachstumsfaktoren, wurden in Studien überprüft und haben gezeigt, dass sie die Sehleistung mit größerem Erfolg verbessern können, als der Goldstandard, die Laser Therapie. Die Absicht der folgenden Studie war es, die zukünftige Anzahl der diabetischen Makulaödem Patienten und deren Behandlungsbedarf an der Universitäts-Augenklinik Graz abschätzen zu können.
Material und Methode
Es handelt sich um eine retrospektive 2-Jahres-Analyse an den Augen von Patienten, die mit einem diabetischen Erstödem im Zeitraum von 01.07.2009 bis 31.06.2011 an der Universitäts-Augenklinik Graz vorstellig wurden. Hauptziel war es, eine 2-Jahres-Inzidenz für das diabetische Makulaödem an der Universitäts-Augenklinik Graz zu ermitteln. Weiters galt es, die Verteilung der angewendeten Therapieformen und die Anzahl der intravitrealen Medikamentenapplikation zu erfassen.
Resultate
Die Jahresinzidenz pro Auge beträgt für das erste Jahr 105 Personen und für das zweite Jahr 101 Personen. Von 226 behandelten Augen erhielten 40,1 % „Laser Therapie plus Bevacizumab Injektionen“, 25,2 % „Laser Therapie“, 21,1 % „Bevacizumab Injektionen“ und 13,2 % zu einer der genannten Therapieformen „zusätzliche Triamcinolone Acetonide Injektionen“. Die Gesamtanzahl der intravitrealen Medikamentenapplikation über die zwei Jahre betrug 343 Injektionen, von welchen 293 Bevacizumab Injektionen waren. Die Mehrzahl der mit Bevacizumab behandelten Augen erhielten ein bis zwei Injektionen.
Schlussfolgerung
Es hat den Anschein, dass gewisse Jahreszeiten Einfluss auf die Entstehung des diabetischen Makulaödems haben. Der hohe Behandlungsbedarf des diabetischen Makulaödems mit fovealer Betroffenheit hat die antivaskulären endothelialen Wachstumsfaktoren unerlässlich in der Therapie des diabetischen Makulaödems gemacht.

Similar content being viewed by others
References
Resnikoff S, Pascolini D, Etya’ale D, et al. Global data on visual impairment in the year 2002. Bull World Health Organ. 2004;82:844–51.
Moss SE, Klein R, Klein BE. The 14-year incidence of visual loss in a diabetic population. Ophthalmology. 1998;105:998–1003.
Moss SE, Klein R, Klein BE. The 10-year incidence of visual loss in a diabetic population. Ophthalmology. 1994;101:1061–70.
Wild S, Roglic G, Green A, et al. Global prevalence of diabetes: estimates for the year 2000 and projections for 2030. Diabetes Care. 2004;27:1047.
Early Treatment Diabetic Retinopathy Study Research Group. Photocoagulation for diabetic macular edema. Early Treatment Diabetic Retinopathy Study Report number 1. Arch Opthalmol. 1985;103:1796–806.
The Diabetic Retinopathy Clinical Research Network, Elan MJ, Aiello LP, Beck RW, et al. Randomized trail evaluating ranibizumab plus prompt or deferred laser or triamcinolone plus prompt laser for diabetic macular edema. Ophthalmology. 2010;117:1064–77.
The Diabetic Retinopathy Clinical Research Network, Elan MJ, Bressler NM, Qin H, et al. Expanded 2-year follow-up of ranibizumab plus prompt or deferred laser or triamcinolone plus prompt laser for diabetic macular edema. Ophthalmology. 2011;118:609–14.
Nguyen QD, Shah SM, Khwaja AA, et al., READ-2 Study Group. Two-year outcome of ranibizumab for edema of the macula in diabetes (READ-2) study. Ophthalmology. 2010;117:2146–51.
Nguyen QD, Shah SM, Khwaja AA, et al., READ-2 Study Group. Primary end point (six months) results of the ranibizumab for edema of the macula in diabetes (READ-2) study. Ophthalmology. 2009;116:2175–81.
Mitchell P, Bandello F, Schmidt-Erfurth U, et al., RESTORE Study Group. The RESTORE study: ranibizumab monotherapy or combined with laser versus laser monotherapy for diabetic macular edema. Ophthalmology. 2011;118:615–25.
Massin P, Bandello F, Justus GG, et al. Safety and efficacy of ranibizumab in diabetic macular edema (RESOLVE study). Diabetes Care. 2010;33:2399–405.
Nguyen QD, Brown DM, Marcus DM, et al. Ranibizumab for diabetic macular edema. Results from 2 phase III randomized trials: rise and ride. Ophthalmology. 2012;119:789–801.
Michaelides M, Kaines A, Hamilton RD, et al. A prospective randomized trail of intravitreal bevacizumab or laser therapy in the management of diabetic macular edema (BOLT study) 12-month data: report 2. Ophthalmology. 2010;117:1078–86.
Rajendram R, Fraser-Bell S, Kaines A, et al. A 2-year prospective randomized controlled trial of intravitreal bevacizumab or laser therapy (BOLT) in the management of diabetic macular edema 24-month data: report 3. Arch Opthalmol. 2012;130:927–79.
Comparison of Age-related Macular Degeneration Treatments Trials (CATT) Research Group, Martin DF, Maguire MG, Ying GS, et al. Ranibizumab and bevacizumab for neovascular age-related macular degeneration. N Engl J Med. 2011;364:1897–908.
Comparison of Age-related Macular Degeneration Treatments Trials (CATT) Research Group, Martin DF, Maguire MG, Fine SL, et al. Ranibizumab and bevacizumab for treatment of neovascular age-related macular degeneration: two-year results. Ophthalmology. 2012;119:1388–98.
Conflict of interest
Monja Michelitsch, Vanessa Gasser-Steiner, and Peter Gasser-Steiner declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Michelitsch, M., Gasser-Steiner, V. & Gasser-Steiner, P. The incidence and treatment of diabetic macular edema at the University Eye-Clinic Graz for a period of 2 years. Spektrum Augenheilkd. 27, 234–238 (2013). https://doi.org/10.1007/s00717-013-0185-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00717-013-0185-2
Keywords
- Diabetic macular edema
- Intravitreal anti-VEGF treatment
- Bevacizumab
- Triamcinolone acetonide
- Central photocoagulation