Abstract
Natural occurrence of chrysotile, a rock-forming serpentine mineral, is largely associated with fluid-rich environments, thus making the fluid chemistry an important factor governing chrysotile growth and stability. Paper examines the effect of alkali chlorides, lowering the H2O activity in fluid, on chrysotile stability at high pressure. The behavior of natural chrysotile (Thetford) in the presence of concentrated NaCl-H2O fluids was studied up to 5 GPa and 400 °C using in-situ Raman spectroscopy and X-ray diffraction combined with resistively heated diamond anvil cell. Reference experiments were also performed on salt-free samples. In salt-free H2O-saturated conditions, chrysotile is stable within the whole temperature range at 1.5–5 GPa. The presence of NaCl in the fluid at XNaCl = mole NaCl/(NaCl+H2O) ≈ 0.1–0.15 strongly suppresses the temperature of chrysotile dehydration at 2–4 GPa to 380 °C, which is about 170 °C lower compared to the equilibrium data for H2O-saturated salt-free system. Talc/talc-like phase only crystallizes at HP-HT conditions as product of the reaction “chrysotile → forsterite + talc”, whereas forsterite appears after the P,T release. This indicates an enhanced solubility of forsterite in supercritical NaCl-H2O fluid at HP-HT conditions. Chrysotile shows disordering/amorphization at unusually low temperature of 150 °C. The shift of the chrysotile dehydration to lower temperatures in saline environment makes this process more operable in natural metamorphic processes in serpentinites. This effect can be extended to other serpentine minerals, including antigorite, a common participant of HP-HT metamorphic reactions in subduction zones.






Similar content being viewed by others
References
Aranovich LY, Newton RC (1996) H2O activity in concentrated NaCl solutions at high pressures and temperatures measured by the brucite-periclase equilibrium. Contrib Mineral Petrol 125:200–212
Aranovich LY, Newton RC (1997) H2O activity in concentrated KCl and KCl-NaCl solutions at high temperatures and pressures measured by the brucite-periclase equilibrium. Contrib Mineral Petrol 127:261–271
Auzende A-L, Daniel I, Reynard B, Lemaire C, Guyot F (2004) High-pressure behavior of serpentine minerals: a Raman spectroscopic study. Am Mineral 31:269–277
Chernosky JV Jr, Berman RG, Bryndzia LT (1988) Serpentine and chlorite equilibria. In: Bailey SW (ed) Hydrous phyllosilicates other than micas, vol 19. Mineralogical Society of America, reviews in mineralogy, V, pp 295–346
Chligui M, Guimbretiere G, Canizares A, Matzen G, Vaills Y (2010) New features in the Raman spectrum of silica: key-points in the improvement on structure knowledge. <hal-00520823> 2010. https://hal.archives-ouvertes.fr/hal-00520823/
Evans B (2004) The serpentinite multisystem revisited: chrysotile is metastable. Int Geol Rev 46:479–506
Fumagalli P, Stixrude L, Poli S, Snyder D (2001) The 10A phase: a high-pressure expandable sheet silicate stable during subduction of hydrated lithosphere. Earth Planet Sci Lett 186:125–141
Gualtieri AF, Giacobbe C, Viti C (2012) The dehydroxylation of serpentine group minerals. Am Mineral 97:666–680
Hammersley AP, Svensson SO, Hanfland M, Fitch AN, Hausermann D (1996) Two-dimensional detector software: from real detector to idealized image or two-theta scan. High Pressure Res 14:235–248
Hilairet N, Daniel I, Reynard B (2006) P–V equations of state and the relative stabilities of serpentine varieties. Am Mineral 33:629–637
Iishi K, Saito M (1973) Synthesis of antigorite. Am Mineral 58:915–919
Kendrick MA, Hémond C, Kamenetsky VS, Danyushevsky L, Devey CW, Rodemann T, Perfit M (2017) Seawater cycled throughout earth/'s mantle in partially serpentinized lithosphere. Nat Geosci 10:222–228
Kitahara S, Takenouchi S, Kennedy GC (1966) Phase relations in the system MgO–SiO2–H2O at high temperatures and pressures. Am J Sci 264:223–233
Kloprogge JT, Frost RL, Rintoul L (1999) Single crystal Raman microscopic study of the asbestos mineral chrysotile. Phys Chem Chem Phys 1:2559–2564
Koster Van Groos AF (1991) Differential thermal analysis of the liquidus relations in the system NaCl2H2O to 6 kbar. Geochim Cosmochim Acta 55:2811–2817
Likhacheva AY, Rashchenko SV (2016) Formation of the high-pressure modification of hydrated talc at 450°C and 4 GPa: in situ diffraction study. J Struct Chem 57:1392–1397
Luсе RW, Ваrtlеtt RW, Раrсs GА (1972) Dissolution kinetics of magnesium silicates. Geochim Cosmochim Acta 36:35–50
MacKenzie KJD, Meinhold RH (1994) Thermal reactions of chrysotile revised: a 29Si and 25Mg MAS NMR study. Am Mineral 79:43–50
Macris CA, Newton RC, Wykes J, Pan R, Manning CE (2020) Diopside, enstatite and forsterite solubilities in H2O and H2O-NaCl solutions at lower crustal and upper mantle conditions. Geochim Cosmochim Acta 279:119–142
Mantegazzi D, Sanchez-Valle C, Driesner T (2013) Thermodynamic properties of aqueous NaCl solutions to 1073 K and 4.5 GPa, and implications for dehydration reactions in subducting slabs. Geochim Cosmochim Acta 121:263–290
Mellini M (1982) The crystal-structure of lizardite-1T: hydrogen bonds and polytypism. Am Mineral 67:587–598
Mellini M, Trommsdorff V, Compagnoni R (1987) Antigorite polysomatism: behaviour during progressive metamorphism. Contrib Mineral Petrol 97:147–155
Moore DE, Summers R, Shengli M, Byerlee JD (1996) Strength of chrysotile-serpentinite gouge under hydrothermal conditions: can it explain a weak San Andreas fault? Geology 24:1041–1044
O’Hanley DS (1996) Serpentinites: records of tectonic and petrologic history. Oxford University Press, Oxford, p 277
O’Hanley DS, Chernosky JV Jr, Wicks JF (1989) The stability of lizardite and chrysotile. Can Mineral 27:483–494
Okada T, Utsumi W, Kaneko H, Yamakata M, Shimomura O (2002) In situ X-ray observations of the decomposition of brucite and the graphite–diamond conversion in aqueous fluid at high pressure and temperature. Phys Chem Mineral 29:439–445
Perillat JP, Daniel I, Koga KT, Reynard B, Cardon H, Crichton WA (2005) Kinetics of antigorite dehydration: a real time X-ray study. Earth Planet Sci Lett 236:899–913
Philippot P, Agrinier P, Scambelluri M (1998) Chlorine cycling during subduction of altered oceanic crust. Earth Planet Sci Lett 161:33–44
Piminov PA, Baranov GN, Bogomyagkov AV, Berkaev DE, Borin VM, Dorokhov VL, Karnaev SE, Kiselev VA, Levichev EB, Meshkov OI, Mishnev SI, Nikitin SA, Nikolaev IB, Sinyatkin SV, Vobly PD, Zolotarev KV, Zhuravlev AN (2016) Synchrotron radiation research and application at VEPP-4. Phys Procedia 84:19–26
Pistorius CW (1963) Some phase relations in the system MgO–SiO2–H2O to high pressures and temperatures. Neues Jahrb Mineral Monatshefte 11:283–293
Rashchenko SV, Kurnosov A, Dubrovinsky L, Litasov KD (2015) Revised calibration of the Sm:SrB4O7 pressure sensor using the Sm-doped yttrium-aluminum garnet primary pressure scale. J Appl Phys 117:145902
Rashchenko SV, Likhacheva AY, Goryainov SV, Krylov AS, Litasov KD (2016) In situ spectroscopic study of water intercalation into talc: new features of ‘ten-angstrom phase’ formation. Am Mineral 101:431–436
Romanenko AV, Rashchenko SV, Kurnosov A, Dubrovinsky L, Goryainov SV, Likhacheva AY, Litasov KD (2018) Single-standard method for simultaneous pressure and temperature estimation using Sm2+:SrB4O7 fluorescence. J Appl Phys 124:165902
Schmidt MW, Poli S (1998) Experimentally based water budgets for dehydrating slabs and consequences for arc magma generation. Earth Planet Sci Lett 163:361–379
Shurupov YV, Kalinin DV (1979) On the magnesian silicates solubility in view of their monocrystals growth. Geol Geophys 9:71–74 (in Russian)
Silantyev SA, Mironenko MV, Novoselov AA (2009) Hydrothermal systems in peridotites of slow-spreading mid-oceanic ridges. Modeling phase transitions and material balance: downwelling limb of a hydrothermal circulation cell. Petrology 17:138–157
Trittschack R, Grobéty B (2013) The dehydroxylation of chrysotile: a combined in situ micro-Raman and micro-FTIR study. Am Mineral 98:1133–1145
Ulmer P, Trommsdorff V (1999) Phase relations of hydrous mantle subducting to 300 km. In: Fei, Bertka CM, Mysen BO (eds) Mantle petrology: field observations and high pressure experimentation: a tribute to Francis R. Boyd, Geochemical, vol 6. Society, Special Publication, pp 259–281
Whitney DL, Evans BW (2010) Abbreviations for names of rock-forming minerals. Am Mineral 95:185–187
Wicks FJ, Whittaker EJW (1975) A reappraisal of the structures of the serpentine minerals. Can Mineral 13:227–243
Yada K (1967) Study of chrysotile asbestos by a high resolution electron microscope. Acta Cryst 23:704–707
Zussman J (1954) Investigation of the crystal structure of antigorite. Mineral Mag 30:498–512
Acknowledgements
We thank anonymous reviewers for their profound and constructive criticisms, which helped to improve the manuscript, as well as Prof. L.K. Kazantseva for discussion of the results and Dr. A. Pan for providing us with the chrysotile sample. Diffraction experiments were carried out at the shared research center SSTRC on the basis of the Novosibirsk VEPP-4 - VEPP-2000 complex at BINP SB RAS, using equipment supported by project RFMEFI62119X0022.
Funding
This work is performed on state assignment of IGM SB RAS and is supported by the RFBR project # 18–05-00312.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editorial handling: D. Paktunc
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Suppl Fig. 1
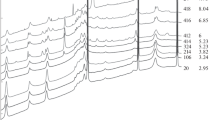
HP-HT Raman spectra of Ctl, added with 5 wt.% of Tlc + Fo, in the presence of saturated NaCl-H2O fluid (run #2s) collected on P,T increase (a) and on P,T release (b). Ctl bands are numerated with smaller letters, the minor bands of seed Tlc (Tlc-s) and Fo (Fo-s), as well as the bands of new-formed Tlc are numerated with larger letters. Circles mark the bands of a tungstate phase produced by the oxidation of W gasket (the most similar spectrum in RRUFF database #R050139.3). Long vertical arrow in Fig.(a) indicates the run time. (PNG 1289 kb)
Suppl Fig. 1b
(PNG 545 kb)
Suppl Fig. 2
HP-HT Raman spectra of Ctl, added with 5 wt.% of Tlc + Fo, in the presence of saturated NaCl-H2O fluid (run #3s). Ctl bands are numerated with smaller letters, the minor bands of seed Tlc (Tlc-s) and Fo (Fo-s) are numerated with larger letters. (PNG 771 kb)
Rights and permissions
About this article
Cite this article
Likhacheva, A.Y., Goryainov, S.V., Rashchenko, S.V. et al. In situ observation of chrysotile decomposition in the presence of NaCl-bearing aqueous fluid up to 5 GPa and 400 °C. Miner Petrol 115, 213–222 (2021). https://doi.org/10.1007/s00710-020-00731-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00710-020-00731-x