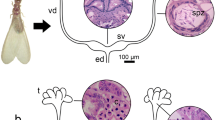
Abstract
The spermatheca and colleterial glands of female insects are organs associated with the reproductive system, responsible for sperm storage and secretion of egg coverings, respectively. Here we compared the development, secretory activity, and chemical nature of the secretion in the spermatheca and colleterial glands of different-aged females of the drywood termite Cryptotermes brevis. We also provide the ultrastructure of these organs in alate females. These structures have been poorly investigated in termites when compared to other eusocial insects (Hymenoptera) and termite-related dictyopterans (mantises and cockroaches). The spermatheca of C. brevis comprises a cone-shaped structure, connected to the genital chamber by a short duct. The colleterial glands, in turn, are divided into anterior and posterior tubules, each showing a basal trunk, and join into a common duct. Histological and histochemical analyses showed that the secretion of proteins and polysaccharides by the spermatheca takes place before pairing, but increases as females mate and store sperm. Colleterial glands of alates showed non-synchronous secretory activity, but the synthesis of products increased in egg-laying queens, together with the epithelium height. Ultrastructure of the spermatheca and colleterial glands revealed epithelia composed of class III secretory cells. Richness of mitochondria and electron-dense secretion in the spermatheca indicates synthesis and transport of content. Presence and absence of colleterial gland secretion in different individuals may reflect variable maturation stages of the females and secretory cells. Assuming that termites are iteroparous, the development and secretion of the spermatheca and colleterial glands play a crucial role for C. brevis queens.










Similar content being viewed by others
Data Availability
The authors confirm that the data supporting the findings of this study are available within the article and its supplementary material.
References
Ahmed I, Gillott C (1981) The spermatheca of Melanoplus sanguinipes (Fabr.) II. Ultrastructure. Int J Invertebr Reprod 4:281–295. https://doi.org/10.1080/01651269.1981.10553438
Baccetti B, Dallai R, Callaini G (1981) The spermatozoon of Arthropoda: Zootermopsis nevadensis and isoptera sperm phylogeny. Int J Invertebr Reprod 3:87–99. https://doi.org/10.1080/01651269.1981.10553385
Baer B (2005) Sexual selection in Apis bees. Apidologie 36:187–200. https://doi.org/10.1051/apido:2005013
Baer B, Eubel H, Taylor NL et al (2009) Insights into female sperm storage from the spermathecal fluid proteome of the honeybee Apis mellifera. Genome Biol 10:R67. https://doi.org/10.1186/gb-2009-10-6-r67
Bednarska AJ, Laskowski R, Pyza E et al (2016) Metal toxicokinetics and metal-driven damage to the gut of the ground beetle Pterostichus oblongopunctatus. Environ Sci Pollut Res 23:22047–22058. https://doi.org/10.1007/s11356-016-7412-8
Bhatnagar RD, Musgrave AJ (1970) Cytochemistry, morphogenesis, and tentative identification of mycetomal microorganisms of Sitophilus granarius L. (Coleoptera). Can J Microbiol 16:1357–1362. https://doi.org/10.1139/M70-223
Boomsma JJ, Baer B, Heinzen J (2005) The evolution of male traits in social insects. Annu Rev Entomol 50:395–420. https://doi.org/10.1146/annurev.ento.50.071803.130416
Brannoch SK, Wieland F, Rivera J et al (2017) Manual of praying mantis morphology, nomenclature, and practices (Insecta, Mantodea). ZooKeys 696:1–100. https://doi.org/10.3897/zookeys.696.12542
Chapman RF (1998) The insects: structure and function. Cambridge University Press, Cambridge
Cheeseman MT, Gillott C (1988) Identification and partial characterization of the major secretory protein of the long hyaline gland in the male grasshopper, Melanoplus sanguinipes. Insect Biochem 18:135–144. https://doi.org/10.1016/0020-1790(88)90017-0
Cheeseman MT, Gillott C (1989) Long hyaline gland discharge and multiple spermatophore formation by the male grasshopper, Melanoplus sanguinipes. Physiol Entomol 14:257–264. https://doi.org/10.1111/j.1365-3032.1989.tb01091
Costa-Leonardo AM, Patrício GB (2005) Structure of the spermatheca in five families of isoptera. Sociobiology 45:659–670
Costa-Leonardo AM, Janei V, da Silva IB (2022) Comparative reproductive biology of pre-, imaginal, and neotenic castes of the Asian termite Coptotermes gestroi (Blattaria, Isoptera, Rhinotermitidae). Bull Entomol Res 112:827–836. https://doi.org/10.1017/S0007485322000232
Costa-Leonardo AM, da Silva IB, Laranjo LT (2023) Termite exocrine systems: a review of current knowledge. Entomol Exp Appl 171:325–342. https://doi.org/10.1111/eea.13292
Costa-Leonardo AM (2002) Cupins-Praga: morfologia, biologia e controle. Editora Divisa, Rio Claro
Courrent A, Quennedey A, Nalepa CA et al (2008) The fine structure of colleterial glands in two cockroaches and three termites, including a detailed study of Cryptocercus punctulatus (Blattaria, Cryptocercidae) and Mastotermes darwiniensis (Isoptera, Mastotermitidae). Arthropod Struct Dev 37:55–66. https://doi.org/10.1016/j.asd.2007.03.004
da Silva IB, Costa-Leonardo AM (2022) Mating mediates morphophysiological changes in the spermathecae of Coptotermes gestroi queens. Entomol Exp Appl 171:361–373. https://doi.org/10.1111/eea.13253
da Silva IB, Costa-Leonardo AM (2023) Functional morphology and development of the colleterial glands in non- and egg-laying females of the pest termite Coptotermes gestroi (Blattaria, Isoptera, Rhinotermitidae). Microsc Microanal 29:1277–1288. https://doi.org/10.1093/micmic/ozad040
da Silva IB, Haifig I, Vargo EL et al (2019) Ergatoid reproductives in the Neotropical termite Nasutitermes aquilinus (Holmgren) (Blattaria: Isoptera: Termitidae): developmental origin, fecundity, and genetics. Insect Sci 27:1322–1333. https://doi.org/10.1111/1744-7917.12727
Du E, Wang S, Luan YX et al (2022) Convergent adaptation of ootheca formation as a reproductive strategy in Polyneoptera. Mol Biol Evol 39:msac042. https://doi.org/10.1093/MOLBEV/MSAC042
Engelmann F (1970) The Physiology of Insect Reproduction. Pergamon, Oxford
Farder-Gomes CF, Santos HCP, Oliveira MA et al (2019) Morphology of ovary and spermathecae of the parasitoid Eibesfeldtphora tonhascai Brown (Diptera: Phoridae). Protoplasma 256:3–11. https://doi.org/10.1007/s00709-018-1276-3
Feng KC, Roelofs WL (1977) Sex pheromone gland development in redbanded leafroller moth, Argyrotaenia velutinana, pupae and adults. Ann Entomol Soc Am 70:721–732. https://doi.org/10.1093/aesa/70.5.721
Gillott C (2002) Accessory reproductive glands: key players in production and protection of eggs. In: Hilker M, Meiners T (eds) Chemoecology of insect eggs and egg deposition. Blackwell Verlag, Oxford, pp 37-59
Gonzalez AN, Ing N, Rangel J (2018) Upregulation of antioxidant genes in the spermathecae of honey bee (Apis mellifera) queens after mating. Apidologie 49:224–234. https://doi.org/10.1007/s13592-017-0546-y
Gotoh A, Shigenobu S, Yamaguchi K et al (2017) Transcriptome profiling of the spermatheca identifies genes potentially involved in the long-term sperm storage of ant queens. Sci Reports 7:1–14. https://doi.org/10.1038/s41598-017-05818-8
Grandi G (1992) Ultrastructural study of testis development and spermatogenesis in Kalotermes flavicollis (Fabr.)(Isoptera, Kalotermitidae). Ital J Zool 59:225–238. https://doi.org/10.1080/11250009209386675
Grassé PP (1982) Termitologia, Anatomie; Physiologie; Reproduction des Termites. Masson, Paris
Greenberg SLW, Stuart AM (1979) The influence of group size on ovarian development in adult and neotenic reproductives of the termite Zootermopsis angusticollis (Hagen)(Hodotermitidae). Int J Invertebr Reprod 1:99–108. https://doi.org/10.1080/01651269.1979.10553304
Happ GM, Happ CM (1982) Cytodifferentiation in the accessory glands of Tenebrio molitor. X. Ultrastructure of the tubular gland in the male pupa. J Morphol 172:97–112. https://doi.org/10.1002/JMOR.1051720109
Happ GM, Yuncker C, Dailey PJ (1982) Cytodifferentiation in the accessory glands of Tenebrio molitor. VII. Patterns of leucine incorporation by the bean-shaped glands of males. J Exp Zool 220:81–92. https://doi.org/10.1002/JEZ.1402200111
Heming BS (2003) The female reproductive system and oogenesis. In: Heming BS (ed) Insect Development and Evolution. Cornell University Press, Ithaca, pp 29–63
Hosken DJ, Uhía E, Ward PI (2002) The function of female accessory reproductive gland secretion and a cost to polyandry in the yellow dung fly. Physiol Entomol 27:87–91. https://doi.org/10.1046/j.1365-3032.2002.00271.x
Hou X-Y, Hua B-Z (2008) Structures of the female reproductive systems in Panorpidae (Mecoptera) with remarks on their taxonomic significance. Acta Zootaxonomica Sin 33:427–434
Kramer KJ, Ong J, Law LH (1973) Onthecal proteins of the oriental praying mantid, Tenodera sinensis. Insect Biochem 3:297–302. https://doi.org/10.1016/0020-1790(73)90060-7
Laranjo LT, da Silva IB, Costa-Leonardo AM (2020) Development and comparative morphology of the reproductive system in different aged males of the drywood termite Cryptotermes brevis (Blattaria, Isoptera, Kalotermitidae). Protoplasma 257:31–42. https://doi.org/10.1007/s00709-019-01417-8
Lawson FA, Thomas JC (1970) Ultrastructural comparison of the spermathecae in Periplaneta americana (Blattaria: Blattidae). J Kansas Entomol Soc 43:418–434
Lay M, Zissler D, Hartman ED (1999) Ultrastructural and functional aspects of the spermatheca of the african migratory locust Locusta migratoria migratorioides (Reiche and Fairmaire) (Orthoptera: Acrididae). Int J Insect Morphol Embryol 28:349–361. https://doi.org/10.1016/S0020-7322(99)00036-7
Ma N, Wang M, Hua B (2013) Ultrastructure of female accessory glands in the scorpionfly Panorpa sexspinosa Cheng (Mecoptera: Panorpidae). Tissue Cell 45:107–114. https://doi.org/10.1016/j.tice.2012.09.010
Martins GF, Serrão JE (2004) Changes in the reproductive tract of Melipona quadrifasciata anthidioides (Hymenoptera: Apidae, Meliponini) queen after mating. Sociobiology 44:241–254
Matsuda R (1976) Morphology and evolution of the insect abdomen. Pergamon Press, Oxford
McMahan EA (1962) Laboratory studies of colony establishment and development in Cryptotermes brevis (Walker)(Isoptera: Kalotermitidae). Proc Hawaii Entomol Soc 18:145–153
Nalepa CA (2015) Origin of termite eusociality: trophallaxis integrates the social, nutritional, and microbial environments. Ecol Entomol 40:323–335. https://doi.org/10.1111/een.12197
Nalepa CA, Lenz M (2000) The ootheca of Mastotermes darwiniensis Froggatt (Isoptera: Mastotermitidae): homology with cockroach oothecae. Proc R Soc London Ser B Biol Sci 267:1809–1813. https://doi.org/10.1098/rspb.2000.1214
Noirot C (1990) Sexual Castes and Reproductive Strategies in Termites. In: Engels W (ed) Social Insects: An Evolutionary Approach to Castes and Reproduction. Springer, Dordrecht, pp 5–35
Noirot C, Quennedey A (1974) Fine structure of insect epidermal glands. Annu Rev Entomol 19:61–80
Noirot C, Quennedey A (1991) Glands, gland cells, glandular units: some comments on terminology and classification. Ann La Société Entomol Fr 27:123–128
Nutting WL (1969) Flight and colony foundation. In: Krishna K, Weesner FM (eds) Biology of termites. Academic Press, New York, pp 233–282
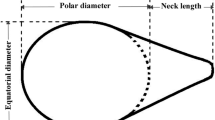
Pascini TV, Martins GF (2017) The insect spermatheca: an overview. Zoology 121:56–71. https://doi.org/10.1016/j.zool.2016.12.001
Pascini TV, Ramalho-Ortigão M, Martins GF (2012) Morphological and morphometrical assessment of spermathecae of Aedes aegypti females. Mem Inst Oswaldo Cruz 107:705–712. https://doi.org/10.1590/S0074-02762012000600001
Raina A, Park YI, Florane C (2003) Behavior and reproductive biology of the primary reproductives of the Formosan subterranean termite (Isoptera: Rhinotermitidae). Sociobiology 41:37–48
Raina A, Murphy C, Florane C et al (2007) Structure of spermatheca, sperm dynamics, and associated bacteria in Formosan subterranean termite (Isoptera: Rhinotermitidae). Ann Entomol Soc Am 100:418–424. https://doi.org/10.1603/0013-8746(2007)100[418:SOSSDA]2.0.CO;2
Rangel J, Shepherd TF, Gonzalez AN et al (2021) Transcriptomic analysis of the honey bee (Apis mellifera) queen spermathecae reveals genes that may be involved in sperm storage after mating. PLoS One 16:e0244648. https://doi.org/10.1371/journal.pone.0244648
RStudio Team (2020) RStudio: Integrated Development for R. RStudio, PBC, Boston, MA. http://www.rstudio.com/. Accessed 25 Apr 2023
Scheffrahn RH, Křeček J, Ripa R, Luppichini P (2009) Endemic origin and vast anthropogenic dispersal of the West Indian drywood termite. Biol Invasions 11:787–799. https://doi.org/10.1007/s10530-008-9293-3
Schoeters E, Billen J (2000) The importance of the spermathecal duct in bumblebees. J Insect Physiol 46:1303–1312. https://doi.org/10.1016/S0022-1910(00)00052-4
Simmons LW (2013) Reproductive system: female. In: Chapman R, Simpson S, Douglas A (eds) The Insects: Structure and Function, 5th edn. Cambridge University Press, Cambridge, p 929
Soltani-Mazouni N, Bordereau C (1987) Changes in the cuticle, ovaries and colleterial. glands during the pseudergate and neotenic molt in Kalotermes flavicollis (FABR.) (Isoptera : Kalotermitidae). Int J Insect Morphol Embryol 16:221–235. https://doi.org/10.1016/0020-7322(87)90022-5
Souza EA, Campos LAO, Neves CA, Zanuncio JC, Serrão JE (2008) Effect of delayed mating on spermathecal activation in Melipona quadrifasciata anthidioides (Hymenoptera, Apidae) queens. Apidologie 39:293–301. https://doi.org/10.1051/apido:2008008
Stay B, Coop AC (1974) ‘Milk’ secretion for embryogenesis in a viviparous cockroach. Tissue Cell 6:669–693. https://doi.org/10.1016/0040-8166(74)90009-3
Stay B, Roth LM (1962) The Colleterial Glands of Cockroaches. Ann Entomol Soc Am 55:124–130. https://doi.org/10.1093/AESA/55.1.124
Sturm R (2002) Development of the accessory glands in the genital tract of female Teleogryllus commodus Walker (Insecta, Orthoptera). Arthropod Struct Dev 31:231–241. https://doi.org/10.1016/S1467-8039(02)00023-3
Sturm R (2016) Morphology and development of the accessory glands in various female cricket species. Arthropod Struct Dev 45:585–593. https://doi.org/10.1016/j.asd.2016.10.001
Szopa TM (1982) Development of the accessory reproductive glands and genital ducts in female Schistocerca gregaria. J Insect Physiol 28:475–483. https://doi.org/10.1016/0022-1910(82)90026-9
Thorne BL (1997) Evolution of eusociality in termites. Annu Rev Ecol Syst 28:27–54
Tirone G, Avancini RMP (1997) Development of female accessory glands of Chrysomya putoria (Wiedemann) (Diptera: Calliphoridae) during oogenesis. Int J Insect Morphol Embryol 26:1–7. https://doi.org/10.1016/S0020-7322(96)00011-6
Wheeler DE, Krutzsch PH (1994) Ultrastructure of the spermatheca and its associated gland in the ant Crematogaster opuntiae (Hymenoptera, Formicidae). Zoomorphology 114:203–212. https://doi.org/10.1007/BF00416859
Wigglesworth VB (1983) The Physiology of Insect Tracheoles. In: Berridge MJ, Treherne JE, Wigglesworth VB (eds) Advances in Insect Physiology. Elsevier, Berlim, pp 85–148
Winnick CG, Holwell GI, Herberstein ME (2009) Internal reproductive anatomy of the praying mantid Ciulfina klassi (Mantodea: Liturgusidae). Arthropod Struct Dev 38:60–69. https://doi.org/10.1016/j.asd.2008.07.002
Yang LY, Hua BZ (2021) Spermatheca of the scorpionfly Sinopanorpa tincta (Navás, 1931) (Mecoptera: Panorpidae). Protoplasma 258:507–515. https://doi.org/10.1007/s00709-020-01572-3
Ye Y, Jones SC, Ammar ED (2009) Reproductive characteristics of imagos of Reticulitermes flavipes (Isoptera: Rhinotermitidae). Ann Entomol Soc Am 102:889–894. https://doi.org/10.1603/008.102.0515
Acknowledgements
This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior-Brasil (CAPES)—Finance Code 001, and Conselho Nacional de Desenvolvimento Tecnológico – CNPq (grant number 305539/2014-0). We thank the CMI (Centro de Microscopia e Imagem, Faculdade de Odontologia de Piracicaba, FOP – UNICAMP), Dr. Flávia S. M. Rodrigues, and Antonio Teruyoshi Yabuki for the technical assistance with Transmission Electron Microscopy; and Dr. Adna Dorigo, Dr. Osmar Malaspina, and LECA (Laboratório de Ecotoxicologia e Conservação de Abelhas – UNESP Rio Claro) for their support with Histology.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
This article does not contain any studies with human participants performed by any of the authors.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Handling Editor: Georg Krohne.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
da Silva, I.B., Costa-Leonardo, A.M. Mating- and oviposition-dependent changes of the spermatheca and colleterial glands in the pest termite Cryptotermes brevis (Blattaria, Isoptera, Kalotermitidae). Protoplasma 261, 213–225 (2024). https://doi.org/10.1007/s00709-023-01891-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-023-01891-1