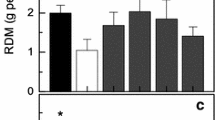
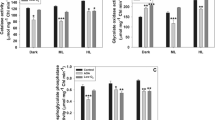
Abstract
When plants are exposed to water stress, photosynthesis is downregulated due to enhanced reactive oxygen species (ROS) and nitric oxide (NO). In contrast, photorespiratory metabolism protected photosynthesis and sustained yield. Modulation of photorespiration by ROS was established, but the effect of NO on photorespiratory metabolism was unclear. We, therefore, examined the impact of externally added NO by using S-nitrosoglutathione (GSNO), a natural NO donor, in leaf discs of pea (Pisum sativum) under dark or light: moderate or high light (HL). Maximum NO accumulation with GSNO was under high light. The presence of 2-4-carboxyphenyl-4,4,5,5-tetramethylimidazoline-1-oxyl-3-oxide (cPTIO), a NO scavenger, prevented the increase in NO, confirming the release of NO in leaves. The increase in S-nitrosothiols and tyrosine-nitrated proteins on exposure to GSNO confirmed the nitrosative stress in leaves. However, the changes by GSNO in the activities and transcripts of five photorespiratory enzymes: glycolate oxidase, hydroxypyruvate reductase, catalase, glycerate kinase, and phosphoglycolate phosphatase activities were marginal. The changes in photorespiratory enzymes caused by GSNO were much less than those with HL. Since GSNO caused only mild oxidative stress, we felt that the key modulator of photorespiration might be ROS, but not NO.







Similar content being viewed by others
Abbreviations
- APX:
-
Ascorbate peroxidase
- CAT:
-
Catalase
- Chl:
-
Chlorophyll
- cPTIO:
-
2-4-Carboxyphenyl-4,4,5,5-tetramethylimidazoline-1-oxyl-3-oxide
- FBPase:
-
Fructose 1,6-bisphosphatase
- GK:
-
Glycerate kinase
- GO:
-
Glycolate oxidase
- GSNO:
-
S-Nitrosoglutathione
- HL:
-
High light
- HPR:
-
Hydroxypyruvate reductase
- MD:
-
Menadione
- ML:
-
Moderate light
- NO:
-
Nitric oxide
- PGLP:
-
Phosphoglycolate phosphatase
- PTM:
-
Post-translational modification
- SOD:
-
Superoxide dismutase
References
Arnon DI (1949) Copper enzymes in isolated chloroplasts. Polyphenoloxidase in Beta vulgaris. Plant Physiol 24:1–15. https://doi.org/10.1104/pp.24.1.1
Aswani V, Rajsheel P, Bapatla RB, Sunil B, Raghavendra AS (2019) Oxidative stress induced in chloroplasts or mitochondria promotes proline accumulation in leaves of pea (Pisum sativum): another example of chloroplast-mitochondria interactions. Protoplasma 256:449–457. https://doi.org/10.1007/s00709-018-1306-1
Bapatla RB, Saini D, Aswani V, Rajsheel P, Sunil B, Timm S, Raghavendra AS (2021) Modulation of photorespiratory enzymes by oxidative and photo-oxidative stress induced by menadione in leaves of Pea (Pisum sativum). Plants 10:987. https://doi.org/10.3390/plants10050987
Bauwe H, Hagemann M, Fernie AR (2010) Photorespiration: players, partners and origin. Trends Plant Sci 15:330–336. https://doi.org/10.1016/j.tplants.2010.03.006
Beyer WF Jr, Fridovich I (1987) Assaying for superoxide dismutase activity: some large consequences of minor changes in conditions. Anal Biochem 161:559–566. https://doi.org/10.1016/0003-2697(87)90489-1
Bhardwaj S, Kapoor D, Singh S, Gautam V, Dhanjal DS, Jan S, Ramamurthy PC, Prasad R, Singh J (2021) Nitric oxide: a ubiquitous signal molecule for enhancing plant tolerance to salinity stress and their molecular mechanisms. J Plant Growth Regul 40:2329–2341. https://doi.org/10.1007/s00344-021-10394-3
Blackwell RD, Murray AJS, Lea PJ (1987a) The isolation and characterisation of photorespiratory mutants of barley and pea. In: In Progress in Photosynthesis Research. Springer, Dordrecht, The Netherlands, pp 625–628
Blackwell RD, Murray AJS, Lea PJ (1987b) Inhibition of photosynthesis in barley with decreased levels of chloroplastic glutamine synthetase activity. J Exp Bot 38:1799–1809. https://doi.org/10.1093/jxb/38.11.1799
Blackwell RD, Murray AJ, Lea PJ, Kendall AC, Hall NP, Turner JC, Wallsgrove RM (1988) The value of mutants unable to carry out photorespiration. Photosynth Res 16:155–176. https://doi.org/10.1007/BF00039491
Chaki M, Carreras A, López-Jaramillo J, Begara-Morales JC, Sánchez-Calvo B, Valderrama R, Corpas FJ, Barroso JB (2013) Tyrosine nitration provokes inhibition of sunflower carbonic anhydrase (β-CA) activity under high temperature stress. Nitric Oxide 29:30–33. https://doi.org/10.1016/j.niox.2012.12.003
Chen P, Li X, Huo K, Wei X, Dai C, Lv C (2014) Promotion of photosynthesis in transgenic rice over-expressing of maize C4 phosphoenolpyruvate carboxylase gene by nitric oxide donors. J Plant Physiol 171:458–466. https://doi.org/10.1016/j.jplph.2013.11.006
Clark D, Durner J, Navarre DA, Klessig DF (2000) Nitric oxide inhibition of tobacco catalase and ascorbate peroxidase. Mol Plant Microbe Interac 13:1380–1384. https://doi.org/10.1094/MPMI.2000.13.12.1380
Corpas FJ, Chaki M, Fernandez-Ocana A, Valderrama R, Palma JM, Carreras A, Begara-Morales JC, Airaki M, Del Rio LA, Barroso JB (2008) Metabolism of reactive nitrogen species in pea plants under abiotic stress conditions. Plant Cell Physiol 49:1711–1722. https://doi.org/10.1093/pcp/pcn144
Corpas FJ, Del Río LA, Palma JM (2019) Impact of nitric oxide (NO) on the ROS metabolism of peroxisomes. Plants 8:37. https://doi.org/10.3390/plants8020037
Corpas FJ, González-Gordo S, Palma JM (2021) Protein nitration: a connecting bridge between nitric oxide (NO) and plant stress. Plant Stress 2:100026. https://doi.org/10.1016/j.stress.2021.100026
Cui LL, Lu YS, Li Y, Yang C, Peng XX (2016) Overexpression of glycolate oxidase confers improved photosynthesis under high light and high temperature in rice. Front Plant Sci 7:1165. https://doi.org/10.3389/fpls.2016.01165
Dellero Y, Jossier M, Schmitz J, Maurino VG, Hodges M (2016) Photorespiratory glycolate–glyoxylate metabolism. J Exp Bot 67:3041–3052. https://doi.org/10.1093/jxb/erw090
Ederli L, Reale L, Madeo L, Ferranti F, Gehring C, Fornaciari M, Romano B, Pasqualini S (2009) NO release by nitric oxide donors in vitro and in planta. Plant Physiol Biochem 47:42–48. https://doi.org/10.1016/j.plaphy.2008.09.008
Eisenhut M, Roell MS, Weber AP (2019) Mechanistic understanding of photorespiration paves the way to a new green revolution. New Phytol 223:1762–1769. https://doi.org/10.1111/nph.15872
Guidi L, Lo Piccolo E, Landi M (2019) Chlorophyll fluorescence, photoinhibition and abiotic stress: does it make any difference the fact to be a C3 or C4 species? Front Plant Sci 10:174. https://doi.org/10.3389/fpls.2019.00174
Gupta KJ, Shah JK, Brotman Y, Jahnke K, Willmitzer L, Kaiser WM, Bauwe H, Igamberdiev AU (2012) Inhibition of aconitase by nitric oxide leads to induction of the alternative oxidase and to a shift of metabolism towards biosynthesis of amino acids. J Exp Bot 63:1773–1784. https://doi.org/10.1093/jxb/ers053
Gupta KJ, Kolbert Z, Durner J, Lindermayr C, Corpas FJ, Brouquisse R, Barroso JB, Umbreen S, Palma JM, Hancock JT, Petrivalsky M, Wendehenne D, Loake GJ (2020) Regulating the regulator: nitric oxide control of post-translational modifications. New Phytol 227:1319–1325. https://doi.org/10.1111/nph.16622
Gururani MA, Venkatesh J, Tran LSP (2015) Regulation of photosynthesis during abiotic stress-induced photoinhibition. Mol Plant 8:1304–1320. https://doi.org/10.1016/j.molp.2015.05.005
Imran M, Shazad R, Bilal S, Imran QM, Khan M, Kang SM, Khan AL, Yun BW, Lee IJ (2021) Exogenous melatonin mediates the regulation of endogenous nitric oxide in Glycine max L. to reduce effects of drought stress. Environ Exp Bot 188:104511. https://doi.org/10.1016/j.envexpbot.2021.104511
Jedelská T, Šmotková Kraiczová V, Berčíková L, Činčalová L, Luhová L, Petřivalský M (2019) Tomato root growth inhibition by salinity and cadmium is mediated by S-nitrosative modifications of ROS metabolic enzymes controlled by S-nitrosoglutathione reductase. Biomolecules 9:393. https://doi.org/10.3390/biom9090393
Kleczkowski LA, Randall DD (1985) Light and thiol activation of maize leaf glycerate kinase: the stimulating effect of reduced thioredoxins and ATP. Plant Physiol 79:274–277. https://doi.org/10.1104/pp.79.1.274
Lee SW, Hahn TR (2003) Light-regulated differential expression of pea chloroplast and cytosolic fructose-1, 6-bisphosphatases. Plant Cell Rep 21:611–618. https://doi.org/10.1007/s00299-002-0563-8
Lehmann M, Schwarzländer M, Obata T, Sirikantaramas S, Burow M, Olsen CE, Tohge T, Ficker MD, Moller BL, Fernie AR, Sweetlove LJ, Laxa M (2009) The metabolic response of Arabidopsis roots to oxidative stress is distinct from that of heterotrophic cells in culture and highlights a complex relationship between the levels of transcripts, metabolites, and flux. Mol Plant 2:390–406. https://doi.org/10.1093/mp/ssn080
Manai J, Kalai T, Gouia H, Corpas FJ (2014) Exogenous nitric oxide (NO) ameliorates salinity-induced oxidative stress in tomato (Solanum lycopersicum) plants. J Soil Sci Plant Nutr 14:433–446. https://doi.org/10.4067/S0718-95162014005000034
Mandal M, Sarkar M, Khan A, Biswas M, Masi A, Rakwal R, Agrawal GK, Srivastava A, Sarkar A (2022) Reactive oxygen species (ROS) and reactive nitrogen species (RNS) in plants–maintenance of structural individuality and functional blend. Adv Redox Res 5:100039. https://doi.org/10.1016/j.arres.2022.100039
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol 22:867–880. https://doi.org/10.1093/oxfordjournals.pcp.a076232
Ortega-Galisteo AP, Rodríguez-Serrano M, Pazmiño DM, Gupta DK, Sandalio LM, Romero-Puertas MC (2012) S-Nitrosylated proteins in pea (Pisum sativum L.) leaf peroxisomes: changes under abiotic stress. J Exp Bot 63:2089–2103. https://doi.org/10.1093/jxb/err414
Patterson BD, Payne LA, Chen YZ, Graham D (1984) An inhibitor of catalase induced by cold in chilling-sensitive plants. Plant Physiol 76:1014–1018. https://doi.org/10.1104/pp.76.4.1014
Puyaubert J, Fares A, Rézé N, Peltier JB, Baudouin E (2014) Identification of endogenously S-nitrosylated proteins in Arabidopsis plantlets: effect of cold stress on cysteine nitrosylation level. Plant Sci 215:150–156. https://doi.org/10.1016/j.plantsci.2013.10.014
Rodríguez-Ruiz M, González-Gordo S, Cañas A, Campos MJ, Paradela A, Corpas FJ, Palma JM (2019) Sweet pepper (Capsicum annuum L.) fruits contain an atypical peroxisomal catalase that is modulated by reactive oxygen and nitrogen species. Antioxidants 8:374. https://doi.org/10.3390/antiox8090374
Sami F, Faizan M, Faraz A, Siddiqui H, Yusuf M, Hayat S (2018) Nitric oxide-mediated integrative alterations in plant metabolism to confer abiotic stress tolerance, NO crosstalk with phytohormones and NO-mediated post translational modifications in modulating diverse plant stress. Nitric Oxide 73:22–38. https://doi.org/10.1016/j.niox.2017.12.005
Shi Y, Ke X, Yang X, Liu Y, Hou X (2022) Plants response to light stress. J Genet Genomics 49:735–747. https://doi.org/10.1016/j.jgg.2022.04.017
Silveira NM, Marcos FC, Frungillo L, Moura BB, Seabra AB, Salgado I, Machado EC, Hancock JT, Ribeiro RV (2017) S-nitrosoglutathione spraying improves stomatal conductance, Rubisco activity and antioxidant defense in both leaves and roots of sugarcane plants under water deficit. Physiol Plant 160:383–395. https://doi.org/10.1111/ppl.12575
Singh N, Bhatla SC (2017) Signaling through reactive oxygen and nitrogen species is differentially modulated in sunflower seedling root and cotyledon in response to various nitric oxide donors and scavengers. Plant Signal Behav 12:e1365214. https://doi.org/10.1080/15592324.2017.1365214
Somerville CR, Ogren WL (1979) A phosphoglycolate phosphatase-deficient mutant of Arabidopsis. Nature 280:833–836. https://doi.org/10.1038/280833a0
Sunil B, Saini D, Bapatla RB, Aswani V, Raghavendra AS (2019) Photorespiration is complemented by cyclic electron flow and the alternative oxidase pathway to optimize photosynthesis and protect against abiotic stress. Photosynth Res 139:67–79. https://doi.org/10.1007/s11120-018-0577-x
Timm S (2020) The impact of photorespiration on plant primary metabolism through metabolic and redox regulation. Biochem Soc Trans 48:2495–2504. https://doi.org/10.1042/BST20200055
Timm S, Bauwe H (2013) The variety of photorespiratory phenotypes–employing the current status for future research directions on photorespiration. Plant Biol 15:737–747. https://doi.org/10.1111/j.1438-8677.2012.00691.x
Timm S, Nunes-Nesi A, Pärnik T, Morgenthal K, Wienkoop S, Keerberg O, Weckwerth W, Kleczkowski LA, Fernie AR, Bauwe H (2008) A cytosolic pathway for the conversion of hydroxypyruvate to glycerate during photorespiration in Arabidopsis. Plant Cell 20:2848–2859. https://doi.org/10.1105/tpc.108.062265
Verma N, Tiwari S, Singh VP, Prasad SM (2020) Nitric oxide in plants: an ancient molecule with new tasks. Plant Growth Regul 90:1–13. https://doi.org/10.1007/s10725-019-00543-w
Vishwakarma A, Kumari A, Mur LA, Gupta KJ (2018) A discrete role for alternative oxidase under hypoxia to increase nitric oxide and drive energy production. Free Radic Biol Med 122:40–51. https://doi.org/10.1016/j.freeradbiomed.2018.03.045
Voss I, Sunil B, Scheibe R, Raghavendra AS (2013) Emerging concept for the role of photorespiration as an important part of abiotic stress response. Plant Biol 15:713–722. https://doi.org/10.1111/j.1438-8677.2012.00710.x
Wada S, Suzuki Y, Takagi D, Miyake C, Makino A (2018) Effects of genetic manipulation of the activity of photorespiration on the redox state of photosystem I and its robustness against excess light stress under CO2-limited conditions in rice. Photosynth Res 137:431–441. https://doi.org/10.1007/s11120-018-0515-y
Yamaguchi K, Nishimura M (2000) Reduction to below threshold levels of glycolate oxidase activities in transgenic tobacco enhances photoinhibition during irradiation. Plant Cell Physiol 41:1397–1406. https://doi.org/10.1093/pcp/pcd074
Zhou B, Guo Z, Xing J, Huang B (2005) Nitric oxide is involved in abscisic acid-induced antioxidant activities in Stylosanthes guianensis. J Exp Bot 56:3223–3228. https://doi.org/10.1093/jxb/eri319
Zhou X, Joshi S, Khare T, Patil S, Shang J, Kumar V (2021) Nitric oxide, crosstalk with stress regulators and plant abiotic stress tolerance. Plant Cell Rep 40:1395–1414. https://doi.org/10.1007/s00299-021-02705-5
Zhou X, Joshi S, Patil S, Khare T, Kumar V (2022) Reactive oxygen, nitrogen, carbonyl and sulfur species and their roles in plant abiotic stress responses and tolerance. J. Plant Growth Regul 41:119–142. https://doi.org/10.1007/s00344-020-10294-y
Acknowledgements
A grant supported this work to ASR (No. EMR/2017/005171). DS held a CSIR-SRF.
Author information
Authors and Affiliations
Contributions
ASR developed the concept and designed the work plan. DS performed most of the experiments. RBB, CKV, PB, and SG did some. ASR and DS drafted the first version. KJG further edited the MS. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Handling Editor: Bhumi Nath Tripathi
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Supplementary Table 1
Sequences of primers used in semi-quantitative RT-PCR (DOCX 17.8 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Saini, D., Bapatla, R.B., Vemula, C.K. et al. Moderate modulation by S-nitrosoglutathione of photorespiratory enzymes in pea (Pisum sativum) leaves, compared to the strong effects of high light. Protoplasma 261, 43–51 (2024). https://doi.org/10.1007/s00709-023-01878-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-023-01878-y