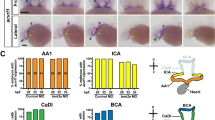
Abstract
MicroRNAs (miRNAs) play important roles in various biological processes. Our previous study showed that inhibition of MTOR with rapamycin treatment suppressed human endothelial cell tube formation, concomitant with the down-regulation of miR-107. Similarly, inhibition of Ztor by rapamycin also suppressed vascular development in zebrafish embryos. To gain a better understanding of the role of miR-107 and MTOR in vascular development, the present study sought to validate its function by over-expressing miR-107 in zebrafish embryos via microinjection with mimic miR-107 duplexes. Alkaline phosphatase (ALP) staining was used to visualise blood vessels in the zebrafish embryo, and expressions of Pten, Ztor and Rap1 were investigated by immunoblotting. Over-expression of miR-107 in zebrafish embryos inhibited the sprouting of intersegmental vessels (ISVs) with concomitant down-regulation of phosphorylated Rps6 expression, which confirmed the inhibition of Ztor signalling. As expected, pten, which is the target of miR-107, was down-regulated. Interestingly, Rap1, a small GTPase protein that is involved in intersomitic vessels sprouting during angiogenesis, was also down-regulated when miR-107 was over-expressed. Overall, our findings suggest that miR-107 and Ztor-mediated suppression of vascular development in zebrafish embryo involves Rap1.








Similar content being viewed by others
Availability of data and materials
Not applicable.
Code availability
Not applicable.
Abbreviations
- ALP:
-
Alkaline phosphatase
- ISVs:
-
Intersegmental vessels
- miRNAs:
-
MicroRNAs
- 3′ UTRs:
-
3′-Untranslated regions
- HIF-1:
-
Hypoxia inducible factor-1
- MTOR:
-
Mammalian target of rapamycin
- PI3K:
-
Phosphatidylinositol-3-OH kinase
- Rps6:
-
Ribosomal protein S6
- Pten:
-
Phosphatase and tensin homolog deleted on chromosome 10
- HUVECs:
-
Human umbilical vein endothelial cells
- Dpf:
-
Day post-fertilisation
- Dpi:
-
Day post-injection
- VEGF:
-
Vascular endothelial growth factor
References
Boettner B, Van Aelst L (2009) Control of cell adhesion dynamics by Rap1 signaling. Curr Opin Cell Biol 21:684–693. https://doi.org/10.1016/j.ceb.2009.06.004
Carmona G, Göttig S, Orlandi A, Scheele J, Bäuerle T, Jugold M, Kiessling F, Henschler R, Zeiher AM, Dimmeler S, Chavakis E (2009) Role of the small GTPase Rap1 for integrin activity regulation in endothelial cells and angiogenesis. Blood 113:488–497. https://doi.org/10.1182/blood-2008-02-138438
Chalhoub N, Baker SJ (2009) PTEN and the PI3-kinase pathway in cancer. Annu Rev Pathol 4:127–150. https://doi.org/10.1146/annurev.pathol.4.110807.092311
Chen L, Li ZY, Xu SY, Zhang XJ, Zhang Y, Luo K, Li WP (2016) Upregulation of miR-107 inhibits glioma angiogenesis and VEGF expression. Cell Mol Neurobiol 36:113–120. https://doi.org/10.1007/s10571-015-0225-3
Choorapoikayil S, Weijts B, Kers R, de Bruin A, den Hertog J (2013) Loss of Pten promotes angiogenesis and enhanced vegfaa expression in zebrafish. Dis Model Mech 6:1159–1166. https://doi.org/10.1242/dmm.012377
Chrzanowska-Wodnicka M, Kraus AE, Gale D, White GC, VanSluys J (2008) Defective angiogenesis, endothelial migration, proliferation, and MAPK signaling in Rap1b-deficient mice. Blood 111:2647–2656. https://doi.org/10.1182/blood-2007-08-109710
Chung JY, Hong SM, Choi BY, Cho H, Yu E, Hewitt SM (2009) The expression of phospho-AKT, phospho-mTOR, and PTEN in extrahepatic cholangiocarcinoma. Clin Cancer Res 15:660–667. https://doi.org/10.1158/1078-0432.CCR-08-1084
Faucherre A, Taylor GS, Overvoorde J, Dixon JE, den Hertog J (2008) Zebrafish pten genes have overlapping and non-redundant functions in tumorigenesis and embryonic development. Oncogene 27:1079–1086. https://doi.org/10.1038/sj.onc.1210730
Folkman J (1997) Angiogenesis and angiogenesis inhibition: an overview. EXS 79:1–8. https://doi.org/10.1007/978-3-0348-9006-9_1
Gerard A, Mertens AEE, van der Kammen RA, Collard JG (2007) The Par polarity complex regulates Rap1-and chemokine-induced T cell polarization. J Cell Biol 176:863–875. https://doi.org/10.1083/jcb.200608161
Hara K, Yonezawa K, Weng QP, Kozlowski MT, Belham C, Avruch J (1998) Amino acid sufficiency and mTOR regulate p70 S6 kinase and eIF-4E BP1 through a common effector mechanism. J Biol Chem 273:22160–22160. https://doi.org/10.1074/jbc.273.23.14484
Ito T, Yagi S, Yamakuchi M (2010) MicroRNA-34a regulation of endothelial senescence. Biochem Biophys Res Commun 398:735–740. https://doi.org/10.1016/j.bbrc.2010.07.012
Kelly P, Bailey CL, Fueger PT, Newgard CB, Casey PJ, Kimple ME (2010) Rap1 promotes multiple pancreatic islet cell functions and signals through mammalian target of rapamycin complex 1 to enhance proliferation. J Biol Chem 285:15777–15785. https://doi.org/10.1074/jbc.M109.069112
Khor ES, Noor SM, Wong PF (2016) Expression of zTOR-associated microRNAs in zebrafish embryo treated with rapamycin. Life Sci 150:67–75. https://doi.org/10.1016/j.lfs.2016.02.076
Khor ES, Wong PF (2018) Endothelial replicative senescence delayed by the inhibition of MTORC1 signaling involves MicroRNA-107. Int J Biochem Cell Biol 101:64–73. https://doi.org/10.1016/j.biocel.2018.05.016
Lakshmikanthan S, Sobczak M, Chun CZ, Henschel A, Dargatz J, Ramchandran R, Chrzanowska-Wodnicka M (2011) Rap1 promotes VEGFR2 activation and angiogenesis by a mechanism involving integrin alpha(v)beta(3). Blood 118:2015–2026. https://doi.org/10.1182/blood-2011-04-349282
Laplante M, Sabatini DM (2012) mTOR signaling in growth control and disease. Cell 149:274–93. https://doi.org/10.1016/j.cell.2012.03.017
Leong IU, Lan CC, Skinner JR, Shelling AN, Love DR (2012) In vivo testing of microRNA-mediated gene knockdown in zebrafish. J Biomed Biotechnol 2012:350352. https://doi.org/10.1155/2012/350352
Li Y, Mao L, Gao Y, Baral S, Zhou Y, Hu B (2015) MicroRNA-107 contributes to post-stroke angiogenesis by targeting Dicer-1. Sci Rep 5:13316. https://doi.org/10.1038/srep13316
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(T)(-Delta Delta C) method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Luo Z, Zheng Y, Zhang W (2018) Pleiotropic functions of miR107 in cancer networks. Onco Targets Ther 11:4113–4124. https://doi.org/10.2147/OTT.S151236
Makky K, Tekiela J, Mayer AN (2007) Target of rapamycin (TOR) signaling controls epithelial morphogenesis in the vertebrate intestine. Developmental Biology 303:501–513. https://doi.org/10.1016/j.ydbio.2006.11.030
Martin HC, Wani S, Steptoe AL et al (2014) Imperfect centered miRNA binding sites are common and can mediate repression of target mRNAs. Genome Biol 15:R51. https://doi.org/10.1186/gb-2014-15-3-r51
Orom UA, Lund AH (2007) Isolation of microRNA targets using biotinylated synthetic microRNAs. Methods 43:162–165. https://doi.org/10.1016/j.ymeth.2007.04.007
Risau W (1997) Mechanisms of angiogenesis. Nature 386:671–674. https://doi.org/10.1038/386671a0
Ristori E, Lopez-Ramirez MA, Narayanan A et al (2015) A Dicer-miR-107 interaction regulates biogenesis of specific miRNAs crucial for neurogenesis. Dev Cell 32:546–560. https://doi.org/10.1016/j.devcel.2014.12.013
Sabatel C, Malvaux L, Bovy N et al (2011) MicroRNA-21 exhibits antiangiogenic function by targeting RhoB expression in endothelial cells. PLoS ONE 6:e16979. https://doi.org/10.1371/journal.pone.0016979
Serbedzija GN, Flynn E, Willett CE (1999) Zebrafish angiogenesis: a new model for drug screening. Angiogenesis 3:353–359. https://doi.org/10.1023/a:1026598300052
Shi FP, Wang XH, Zhang HX, Shang MM, Liu XX, Sun HM, Song YP (2018) MiR-103 regulates the angiogenesis of ischemic stroke rats by targeting vascular endothelial growth factor (VEGF). Iran J Basic Med Sci 21:318–324. https://doi.org/10.22038/IJBMS.2018.27267.6657
Sonkoly E, Pivarcsi A (2009) Advances in microRNAs: implications for immunity and inflammatory diseases. J Cell Mol Med 13:24–38. https://doi.org/10.1111/j.1582-4934.2008.00534.x
Suarez Y, Fernandez-Hernando C, Pober JS, Sessa WC (2007) Dicer dependent microRNAs regulate gene expression and functions in human endothelial cells. Circ Res 100:1164–1173. https://doi.org/10.1161/01.RES.0000265065.26744.17
Sun H, Lesche R, Li DM et al (1999) PTEN modulates cell cycle progression and cell survival by regulating phosphatidylinositol 3,4,5,-trisphosphate and Akt/protein kinase B signaling pathway. Proc Natl Acad Sci USA 96:6199–6204. https://doi.org/10.1073/pnas.96.11.6199
Sun X, Sit A, Feinberg MW (2014) Role of miR-181 family in regulating vascular inflammation and immunity. Trends Cardiovasc Med 24:105–112. https://doi.org/10.1016/j.tcm.2013.09.002
Tian F, Wang JH, Zhang ZH, Yang J (2019) miR-107 modulates chondrocyte proliferation, apoptosis, and extracellular matrix synthesis by targeting PTEN. Int J Clin Exp Pathol 12:488–497
Tobia C, Gariano G, Guerra J, Presta M (2015) Zebrafish embryo intersegmental vessels: a tool for investigating sprouting angiogenesis. Methods Mol Biol 1214:173–184. https://doi.org/10.1007/978-1-4939-1462-3_10
Varani G (2015) Twenty years of RNA: the discovery of microRNAs. RNA 21:751–752. https://doi.org/10.1261/rna.050237.115
Vejnar CE, Blum M, Zdobnov EM (2013) miRmap web: comprehensive microRNA target prediction online. Nucleic Acids Res 41:W165-168. https://doi.org/10.1093/nar/gkt430
Wang WX, Wilfred BR, Madathil SK et al (2010) miR-107 regulates granulin/progranulin with implications for traumatic brain injury and neurodegenerative disease. Am J Pathol 177:334–345. https://doi.org/10.2353/ajpath.2010.091202
Wong PF, Jamal J, Tong KL et al (2017) Deregulation of hsa-miR-20b expression in TNF-alpha-induced premature senescence of human pulmonary microvascular endothelial cells. Microvasc Res 114:26–33. https://doi.org/10.1016/j.mvr.2017.06.002
Xiong J, Wang D, Wei A et al (2017) Deregulated expression of miR-107 inhibits metastasis of PDAC through inhibition PI3K/Akt signaling via caveolin-1 and PTEN. Exp Cell Res 361:316–323. https://doi.org/10.1016/j.yexcr.2017.10.033
Xu YP, Liang L, Wang XM (2011) The levels of Pdx1/insulin, Cacna1c and Cacna1d, and beta-cell mass in a rat model of intrauterine undernutrition. J Matern Fetal Neonatal Med 24:437–443. https://doi.org/10.3109/14767058.2010.497571
Yamakuchi M, Lotterman CD, Bao C et al (2010) P53-induced microRNA-107 inhibits HIF-1 and tumor angiogenesis. Proc Natl Acad Sci USA 107:6334–6339. https://doi.org/10.1073/pnas.0911082107
Yan JL, Li F, Ingram DA, Quilliam LA (2008) Rap1a is a key regulator of fibroblast growth factor 2-induced angiogenesis and together with Rap1b controls human endothelial cell functions. Mol Cell Biol 28:5803–5810. https://doi.org/10.1128/Mcb.00393-08
Yu QF, Liu P, Li ZY, Zhang CF, Chen SQ, Li ZH, Zhang GY, Li JC (2018) MiR-103/107 induces tumorigenicity in bladder cancer cell by suppressing PTEN. Eur Rev Med Pharmacol Sci 22:8616–8623. https://doi.org/10.26355/eurrev_201812_16625
Funding
This study was supported by the University of Malaya Research Grant (RG500-13HTM) and the Postgraduate Research Grant (PPP) (PG306-2016A).
Author information
Authors and Affiliations
Contributions
KES performed experiments, data analyses and drafted the manuscript.
SMN and WPF conceived and designed the study.
WPF revised the manuscript. SMN provided critical review of the manuscript.
All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
Animal work was performed in accordance with established European Commission regulation. European Commission Direction (2020) does not regulate earliest animal life-stages as animal studies. Embryos used in this study were all below 5 dpf.
Conflict of interest
The authors declare no competing interests.
Additional information
Handling Editor: Jörn Bullerdiek
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Khor, ES., Noor, S.M. & Wong, PF. MiR-107 inhibits the sprouting of intersegmental vessels of zebrafish embryos. Protoplasma 259, 691–702 (2022). https://doi.org/10.1007/s00709-021-01695-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-021-01695-1