Abstract
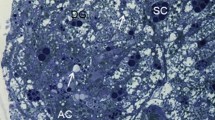
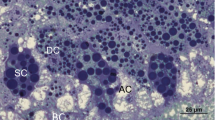
The herald moths, Scoliopteryx libatrix, overwinter in hypogean habitats. The ultrastructure of their fat body (FB) cells and Malpighian tubule (MT) epithelial cells was studied by light microscopy and transmission electron microscopy, and essential biometric and biochemical measurements were performed. The FB was composed of adipocytes and sparse urocytes. The ultrastructure of both cells did not change considerably during this natural starvation period, except for rough endoplasmic reticulum (rER) which became more abundant in March females. In the cells, the reserve material consisted of numerous lipid droplets, glycogen rosettes, and protein granula. During overwintering, the lipid droplets diminished, and protein granula became laminated. The MTs consisted of a monolayer epithelium and individual muscle cells. The epithelial cells were attached to the basal lamina by numerous hemidesmosomes. The apical plasma membrane was differentiated into numerous microvilli, many of them containing mitochondria. Nuclei were surrounded by an abundant rER. There were numerous spherites in the perinuclear part of the cells. The basal plasma membrane formed infoldings with mitochondria in between. Nuclei were located either in the basal or in the central part of the cells. During overwintering, spherites were gradually exploited, and autophagic structures appeared: autophagosomes, autolysosomes, and residual bodies. There were no statistical differences between the sexes in any measured biometric and biochemical variables in the same time frames. The energy-supplying lipids and glycogen, and spherite stores were gradually spent during overwintering. In March, the augmented rER signified the intensification of synthetic processes prior to the epigean ecophase.










Similar content being viewed by others
References
Arrese EL, Soulages JL (2010) Insect fat body: energy, metabolism, and regulation. Annu Rev Entomol 55:207–225
Arrese EL, Canavoso LE, Jouni ZE, Pennington JE, Tsuchida K, Wells MA (2001) Lipid storage and mobilization in insects: current status and future directions. Insect Biochem Mol Biol 31:7–17
Ballan-Dufrancais C (2002) Localization of metals in cells of pterygote insects. Microsc Res Techniq 56:403–420
Beyenbach KW, Skaer H, Dow JAT (2010) The developmental, molecular, and transport biology of Malpighian tubules. Annu Rev Entomol 55:351–374
Bourne JD, Cherix D (1978) Note sur l'écophase souterraine de Triphosa dubitata L.(Lep., Geometridae) et Scoliopteryx libatrix L. (Lep., Noctuiidae). Bull Soc Vaudoise Sc Nat 354(74):147–156
Bouvet Y, Turquin M-J, Bornard C, Desvignes S, Notteghem P (1974) Étude des déclencheurs visuels intervenant lors de la pénétration souterraine de Scoliopteryx libatrix L. et Triphosa dubitata L. (Lépidoptères trogloxènes). Ann Spéléol 29(2):229–236
Bradle TJ (1985) The excretory system: structure and physiology. In: Kerkut GA, Chopard L (eds) La biologie des Orthoptères. Lechevalier, Paris, p 541
Bradley TJ (2003) Excretion. In: Resh VH, Cardé RT (eds) Encyclopaedia of insects. Academic Press, Elsevier Science, San Diego, pp 380–386
Chapman RF (2008) The insects: structure and function. Cambridge University Press, New York, pp 478–508
Christian E, Moog O (1982) Zur Frage der ökologischen Klassifikazion der Kavernikolen am Beispiel der Höhlen-Schmetterlinge Österreichs. Zool Anz 5(6):382–392
Cohen E (2003) Fat body. In: Resh VH, Cardé RT (eds) Encyclopedia of insects. Academic, Amsterdam, pp 407–409
Dow JAT (2009) Insights into the Malpighian tubule from functional genomics. J Exp Biol 212:435–445
Fibiger M, Hacker HH (2005) Systematic list of Noctuoidea of Europe. Esperiana Schwanfeld Bucherei zur Entomologie 11:93–205
Furtado WCA, Azevedo DO, Martins GF, Zanuncio JC (2013) Histochemistry and ultrastructure of urocytes in the pupae of the stingless bee Melipona quadrifasciata (Hymenoptera: Meliponini). Microsc Microanal 19:1502–1510
Hahn DA, Denlinger DL (2011) Energetics of insect diapause. Annu Rev Entomol 56:103–121
Hazelton SR, Felgenhauer BE, Spring JH (2001) Ultrastructural changes in the Malpighian tubules of the house cricket, Acheta domesticus, at the onset of diuresis: a time study. J Morphol 247:80–92
Kalender Y, Kalender S, Candan S (2002) Fine structure of Malpighian tubules in the Agrotis segetum (Lepidoptera: Noctuidae) pupae. Acta Zool Bulg 54:87–96
Kates M (1991) Techniques in Lipidology. Elsevier, Amsterdam
Keeley LL (1985) Physiology and biochemistry of the fat body. Structure of the fat body. In: Kerkut, GA, Gilbert LI (Eds.), Comprehensive insect physiology and pharmacology. Integument, respiration and circulation 3. Pergamon Press, Oxford, pp. 211–248
Klionsky DJ, Abdalla FC, Zuckerbraun B (2012) Guidelines for the use and interpretation of assays for monitoring autophagy. Autophagy 8(4):445–544
Lipovšek S, Novak (2016) Autophagy in the fat body cells of the cave cricket Troglophilus neglectus Krauss, 1878 (Rhaphidophoridae, Saltatoria) during overwintering. Protoplasma 253:457–466
Lipovšek S, Letofsky-Pabst I, Novak T, Hofer F, Pabst MA (2009) Structure of the Malpighian tubule cells and annual changes in the structure and chemical composition of their spherites in the cave cricket Troglophilus neglectus Krauss, 1878 (Rhaphidophoridae, Saltatoria). Arthropod Struct Dev 38:315–327. doi:10.1016/j.asd.2009.02.001
Lipovšek S, Novak T, Janžekovič F, Pabst MA (2011) Role of the fat body in the cave crickets Troglophilus cavicola and Troglophilus neglectus (Rhaphidophoridae, Saltatoria) during overwintering. Arthropod Struct Dev 40(1):54–63
Lipovšek S, Novak T, Janžekovič F, Leitinger G (2015) Changes in the midgut diverticula in the harvestmen Amilenus aurantiacus (Phalangiidae, Opiliones) during winter diapause. Arthropod Struct Dev. doi:10.1016/j.asd.2014.12.002
Martoja R, Ballan-Dufrançais C (1984) The ultrastructure of the digestive and excretory organs. In: King RC, Akai H (eds) Insect ultrastructure. Plenum, New York, pp 199–268
Mizushima N (2007) Autophagy: process and function. Genes Dev 21:2861–2873
Mizushima N, Klionsky DJ (2007) Protein turnover via autophagy: implications for metabolism. Annu Rev Nutr 27:19–40
Motas C, Decou V, Burghele A (1967) Sur l’association pariétale des grottes d’Oltenie (Roumanie). Ann Spéléol 22(3):475–522
Novak T, Perc M, Lipovšek S, Janžekovič F (2012) Duality of terrestrial subterranean fauna. Int J Speleol 41(2):181–188
Novak T, Janžekovič F, Lipovšek S (2013) Contribution of non-troglobiotic terrestrial invertebrates to carbon input in hypogean habitats. Acta Carsologica 42:301–309
Paes de Oliveira VT, Cruz-Landim C (2003) Morphology and function of insect fat body cells: a review. Biociências, Porto Alegre 11:195–205
Pal R, Kumar K (2013) Malpighian tubules of adult flesh fly, Sarcophaga ruficornis fab. (Diptera: Sarcophagidae): an ultrastructural study. Tissue Cell 45(5):312–317
Pigino G, Migliorini M, Paccagnini E, Bernini F, Leonzio C (2005) Fine structure of the midgut and Malpighian papillae in Campodea (Monocampa) quilisi Silvestri, 1932 (Hexapoda, Diplura) with special reference to the metal composition and physiological significance of midgut intracellular electron-dense granules. Tissue Cell 37:223–232
Plummer DT (1987) An introduction to practical biochemistry. McGraw-Hill, London
Polver PD, Sacchi L, Grigolo A, Laudani U (1986) Fine structure of the fat body and its bacteroids in Blattella germanica (Blattodea). Acta Zool 67:63–71
Roeder KD, Fenton MB (1973) Acoustic responsiveness of Scoliopteryx libatrix (Lepidoptera, Noctuidae), a moth that shares hibernacula with some insectivorous bats. Canad J Zool 51(7):681–685
Scott RC, Schuldiner O, Neufeld TP (2004) Role and regulation of starvation-induced autophagy in the Drosophila fat body. Develop Cell 7:167–178
Šobotnik J, Weyda F, Hanus R, Cvačka J, Nebesáŕová J (2006) Fat body of Prorhinotermes simplex (Isoptera: Rhinotermitidae): ultrastructure, inter-caste differences and lipid composition. Micron 37:648–656
Sohal RS (1974) Fine structure of the Malpighian tubules in the housefly, Musca domestica. Tissue Cell 6:719–728
Turqiun M-J (1994) Lepidoptera. In: Encyclopaedia biospeologica I. Société de Biospéologie. Moulis (CNRS) and Bucarest (Academie Roumaine), pp. 333–339
Yang Z, Klionsky DJ (2010) Eaten alive: a history of macroautophagy. Nature Cell Biol 12(9):814–822
Yu CH (1999) Ultrastructure of the Malpighian tubule cells in the mosquito larvae, Culex pipiens pallens. Korean J Entomol 29:141–147
Yu CH (2003) Ultrastructure of the Malpighian tubule cells in the mosquito larvae, Anopheles sinesis. Korean J Entomol 33:151–159
Ziegler H (2016) Scoliopteryx libatrix (Linnaeus, 1767). In: Ziegler. H., Schmetterlinge der Paläarktischen Region. http://www.euroleps.ch/index.php Accessed 27.11.2016
Acknowledgements
We would like to thank Elisabeth Bock und Rudi Schmied (Medical University Graz) for their excellent technical assistance. Michelle Gadpaille valuably improved the English of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Handling Editor: Douglas Chandler
Rights and permissions
About this article
Cite this article
Lipovšek, S., Janžekovič, F. & Novak, T. Ultrastructure of fat body cells and Malpighian tubule cells in overwintering Scoliopteryx libatrix (Noctuoidea). Protoplasma 254, 2189–2199 (2017). https://doi.org/10.1007/s00709-017-1110-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-017-1110-3