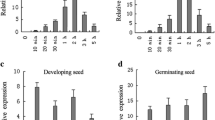
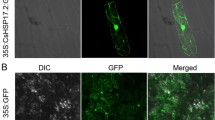
Abstract
Heat shock proteins (HSPs) exist extensively in eukaryotes and are conserved molecular chaperones with important contribution to plant’s survival under environmental stresses. Here, the cloning and characterization of one complementary DNA (cDNA) designated as BcHSP70 from young seedlings of Brassica campestris were reported in the present work. Bioinformatic analysis revealed that BcHSP70 belongs to the plant HSP gene family and had the closest relationship with HSP70-4 from Arabidopsis thaliana. Constitutive overexpression of BcHSP70 in tobacco obviously conferred tolerance to heat stress by affecting different plant physiological parameters. In our study, transgenic tobaccos exhibited higher chlorophyll content than wild-type control when exposed to heat stress. Superoxide dismutase (SOD) and peroxidase (POD) activities, which were helpful to decrease the damage to the membrane system, were significantly higher in transformants compared to wild-type lines. Meanwhile, lower comparative electrical conductivity and malondialdehyde (MDA) content and higher proline and soluble sugar accumulation were found in transgenic tobaccos than in wild-type lines. All these above results indicated that this isolated BcHSP70 cDNA owned the ability to improve the tolerance to heat stress in transgenic tobacco, which provides helpful information and good basement to culture new robust B. campestris variety resistant to high-temperature stress by molecular breeding in the future.







Similar content being viewed by others
References
Arce-Paredes P, Mora-Escobedo R, Luna-Arias J P, Mendoza-Hernández G, Rojas-Espinosa O (2011) Heat, salinity, and acidity, commonly upregulate A1aB1b proglycinin in soybean embryonic axes. Soybean—biochemistry, chemistry and physiology. InTech, Rijeka, 402–422
Asada K (1992) Ascorbate peroxidase—a hydrogen peroxide-scavenging enzyme in plants. Physiol Plant 85(2):235–241
Baniwal SK, Bharti K, Chan KY, Fauth M, Ganguli A, Kotak S, von Koskull-DÖring P (2004) Heat stress response in plants: a complex game with chaperones and more than twenty heat stress transcription factors. J Biosci 29(4):471–487
Barnes JD, Balaguer L, Manrique E, Elvira S, Davison AW (1992) A reappraisal of the use of DMSO for the extraction and determination of chlorophylls a and b in lichens and higher plants. Environ Exp Bot 32(2):85–100
Barnett T, Altschuler M, McDaniel CN, Mascarenhas JP (1979) Heat shock induced proteins in plant cells. Dev Genet 1(4):331–340
Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline for water-stress studies. Plant Soil 39(1):205–207
Boorstein WR, Ziegelhoffer T, Craig EA (1994) Molecular evolution of the HSP70 multigene family. Mol Evol 38:1–17
Boston RS, Viitanen PV, Vierling E (1996) Molecular chaperones and protein folding in plants post-transcriptional control of gene expression in plants. Plant Mol Biol 32:191–222
Bray EA, Bailey-Serres J, Weretilnyk E (2000) Responses to abiotic stresses. Biochemistry and Molecular Biology of Plants. 1158–1203
Chance B, Maehly A (1955) Assay of catalases and peroxidases. Methods Enzymol 2:764–775
Chen A, Han R, Li D, Lin LL, Luo HX, Tang SJ (2010) A comparison of two methods for electrical conductivity about plant leaves. J Guangdong Edu Inst 5:018
Cho EK, Choi YJ (2009) A nuclear-localized HSP70 confers thermoprotective activity and drought-stress tolerance on plants. Biotechnol Lett 31(4):597–606
Cho EK, Hong CB (2004) Molecular cloning and expression pattern analyses of heat shock protein 70 genes from Nicotiana tabacum. J Plant Biology 47(2):149–159
Clos J, Westwood JT, Becker PB, Wilson S, Lambert K, Wu C (1990) Molecular cloning and expression of a hexameric Drosophila heat shock factor subject to negative regulation. Cell 63(5):1085–1097
Cui L, Huang Q, Yan B, Wang Y, Qian Z, Pan J, Kai GY (2015) Molecular cloning and expression analysis of a Cu/Zn SOD gene BcCSD1 from Brassica campestris ssp. chinensis. Food Chem 186:306–311
Cvikrová M, Gemperlová L, Dobrá J, Martincová O, Prásil IT, Gubis J, Vanková R (2012) Effect of heat stress on polyamine metabolism in proline-over-producing tobacco plants. Plant Sci 182:49–58
Draper HH, Squires EJ, Mahmoodi H, Wu J, Agarwal S, Hadley M (1993) A comparative evaluation of thiobarbituric acid methods for the determination of malondialdehyde in biological materials. Free Radic Biol Med 15(4):353–363
Edwards K, Johnston C, Thompson C (1991) A simple and rapid method for the preparation of plant genomic DNA for PCR analyses. Nucl Acids Res 98:1349
Feder ME, Hofmann GE (1999) Heat-shock proteins, molecular chaperones, and the stress response: evolutionary and ecological physiology. Annu Rev Physiol 61(1):243–282
Feierabend J (1977) Capacity for chlorophyll synthesis in heat-bleached 70S ribosome-deficient rye leaves. Planta 135(1):83–88
Gosavi GU, Jadhav AS, Kale AA, Gadakh SR, Pawar BD, Chimote VP (2014) Effect of heat stress on proline, chlorophyll content, heat shock proteins and antioxidant enzyme activity in sorghum (Sorghum bicolor) at seedlings stage. Indian J Biotechnol 13(3):356–363
Gupta SC, Sharma A, Mishra M, Mishra RK, Chowdhuri DK (2010) Heat shock proteins in toxicology: how close and how far? Life Sci 86(11):377–384
Guy CL, Li Q-B (1998) The organization and evolution of the spinach stress 70 molecular chaperone gene family. Plant Cell 10(4):539–556
Han Y, Fan S, Zhang Q, Wang Y (2013) Effect of heat stress on the MDA, proline and soluble sugar content in leaf lettuce seedlings. Agric Sci 4(05):112
Hartl FU, Hayer-Hartl M (2009) Converging concepts of protein folding in vitro and in vivo. Nat Struct Mol Biol 16(6):574–581
Hodgins R, Van Huystee RB (1986) Porphyrin metabolism in chill stressed maize (Zea mays L.). J Plant Physiol 125(3):325–336
Hu W, Hu G, Han B (2009) Genome-wide survey and expression profiling of heat shock proteins and heat shock factors revealed overlapped and stress specific response under abiotic stresses in rice. Plant Sci 176(4):583–590
Irigoyen JJ, Einerich DW, Sánchez‐Díaz M (1992) Water stress induced changes in concentrations of proline and total soluble sugars in nodulated alfalfa (Medicago sativa) plants. Physiol Plant 84(1):55–60
Kai G, Ji Q, Lu Y, Qian Z, Cui L (2012) Expression of Monstera deliciosa agglutinin gene (mda) in tobacco confers resistance to peach-potato aphids. Integr Biol 4(8):937–944
Karlin S, Brocchieri L (1998) Heat shock protein 70 family: multiple sequence comparisons, function, and evolution. J Mol Evol 47(5):565–577
Key JL, Lin CY, Chen YM (1981) Heat shock proteins of higher plants. Proc Natl Acad Sci 78(6):3526–3530
Lin BL, Wang JS, Liu HC, Chen RW, Meyer Y, Barakat A, Delseny M (2001) Genomic analysis of the Hsp70 superfamily in Arabidopsis thaliana. Cell Stress Chaperones 6(3):201
Montero-Barrientos M, Hermosa R, Nicolás C, Cardoza RE, Gutiérrez S, Monte E (2008) Overexpression of a Trichoderma HSP70 gene increases fungal resistance to heat and other abiotic stresses. Fungal Genet Biol 45(11):1506–1513
Montero-Barrientos M, Hermosa R, Cardoza RE, Gutiérrez S, Nicolás C, Monte E (2010) Transgenic expression of the Trichoderma harzianum hsp70 gene increases Arabidopsis resistance to heat and other abiotic stresses. J Plant Physiol 167(8):659–665
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15(3):473–497
Pan Y, Wu LJ, Yu ZL (2006) Effect of salt and drought stress on antioxidant enzymes activities and SOD isoenzymes of liquorice (Glycyrrhiza uralensis Fisch). Plant Growth Regul 49(2–3):157–165
Park HS, Jeong WJ, Kim E, Jung Y, Lim JM, Hwang MS, Choi DW (2012) Heat shock protein gene family of the Porphyra seriata and enhancement of heat stress tolerance by PsHSP70 in Chlamydomonas. Mar Biotechnol 14(3):332–342
Park HJ, Jung WY, Lee SS, Song JH, Kwon SY, Kim H, Kim C, Ahn JC, Cho HS (2013) Use of heat stress responsive gene expression levels for early selection of heat tolerant cabbage (Brassica oleracea L.). Int J Mol Sci 14(6):11871–11894
Renner T, Waters ER (2007) Comparative genomic analysis of the Hsp70s from five diverse photosynthetic eukaryotes. Cell Stress Chaperones 12(2):172
Rousch JM, Bingham SE, Sommerfeld MR (2014) Protein expression during heat stress in thermo-intolerant and thermo-tolerant diatoms. J Exp Mar Biol Ecol 306(2):231–243
Scandalios JG (1993) Oxygen stress and superoxide dismutases. Plant Physiol 101:1–7
Scandalios JG, Foyer CH, Mullineaux PM (1994) Regulation and properties of plant catalases. Causes of photooxidative stress and amelioration of defense systems in plants., pp 275–315
Schroda M, Vallon O (2009) Chaperones and proteases. The Chlamydomonas Source Book, Secondth edn. Elsevier, Oxford, pp 671–730
Su PH, Li H (2008) Arabidopsis stromal 70-kD heat shock proteins are essential for plant development and important for thermotolerance of germinating seeds. Plant Physiol 146(3):1231–1241
Sung DY, Guy CL (2003) Physiological and molecular assessment of altered expression of Hsc70-1 in Arabidopsis. Evidence for pleiotropic consequences. Plant Physiol 132:979–987
Sung DY, Vierling E, Guy CL (2001a) Comprehensive expression profile analysis of the Arabidopsis Hsp70 gene family. Plant Physiol 126(2):789–800
Sung DY, Kaplan F, Guy CL (2001b) Plant HSP70 molecular chaperones: protein structure, gene family, expression and function. Physiol Plant 113:443–451
Tom M, Douek J, Yankelevich I, Bosch TCG, Rinkevich B (1999) Molecular characterization of the first heat shock protein 70 from a reef coral. Biochem Biophys Res Commun 262:103–108
Van Hasselt PR, Strikwerda JT (1976) Pigment degradation in discs of the thermophilic Cucumis sativus as affected by light, temperature, sugar application and inhibitors. Physiologia Plantarum (Denmark)
Wang W, Vinocur B, Altman A (2003) Plant responses to drought, salinity and extreme temperatures: towards genetic engineering for stress tolerance. Planta 218(1):1–14
Xu H, Liu G, Liu G, Yan B, Duan W, Wang L, Li S (2014) Comparison of investigation methods of heat injury in grapevine (Vitis) and assessment to heat tolerance in different cultivars and species. BMC Plant Biol 14(1):156
Zhang J, Kirkham MB (1994) Drought-stress-induced changes in activities of superoxide dismutase, catalase, and peroxidase in wheat species. Plant Cell Physiol 35(5):785–791
Zhang J, Kirkham MB (1995) Water relations of water-stressed, split-root C4 (Sorghum bicolor; Poaceae) and C3 (Helianthus annuus; Asteraceae) plants. Am J Bot 82:1220–1229
Zhou C, Qian Z, Ji Q, Xu H, Chen L, Luo X, Kai GY (2011) Expression of the zga agglutinin gene in tobacco can enhance its anti-pest ability for peach-potato aphid (Myzus persica). Acta Physiol Plant 33(5):2003–2010
Zhu Y, Zhu G, Guo Q, Zhu Z, Wang C, Liu Z (2013) A comparative proteomic analysis of Pinellia ternata leaves exposed to heat stress. Int J Mol Sci 14(10):20614–20634
Acknowledgments
This work was supported by the Shanghai Education Committee Fund (13ZZ104, J50401). Authors also wanted to acknowledge the important contribution from anonymous reviewers’ comments and revision for the paper.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Bhumi Nath Tripathi
Xiaorong Wang and Bin Yan are co-first authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Fig. S1
Schematic representation of transformation plasmid (pCAMBIA2300 + -BcHSP70) (DOCX 659 kb)
Fig. S2
Representative PCR analysis for the presence of the BcHSP70 in the transgenic tobacco plants. M-size marker (2 kb, 1 kb, 750 bp, 500 bp, 250 bp, 100 bp) N-untransformed plant (negative control) and P- p2300 + -BcHSP70 (positive control). (DOC 61 kb)
Fig. S3
Growth state comparison of the transgenic tobacco plants with the wild type after exposure at 42 °C for 0 h and 3 h. (DOC 81 kb)
Rights and permissions
About this article
Cite this article
Wang, X., Yan, B., Shi, M. et al. Overexpression of a Brassica campestris HSP70 in tobacco confers enhanced tolerance to heat stress. Protoplasma 253, 637–645 (2016). https://doi.org/10.1007/s00709-015-0867-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-015-0867-5