Abstract
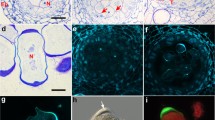
The identification of nucleolar proteins and immunocytochemical localization of small nuclear ribonucleoprotein (snRNP) elements revealed the presence of three types of nuclear bodies in Douglas fir microspore nuclei. One type consists of structures resembling Cajal bodies (CBs) and contains nucleolar proteins as well as snRNPs and U2 snRNA. The second type is bizonal bodies, which are nuclear bodies also linked with the splicing system. The bizonal body comprises two parts: the first contains Sm proteins and stains strongly with silver stain, and the second resembles CBs in terms of the degree of silver staining and molecular composition. Douglas fir is the second species after larch where the presence of bizonal bodies has been demonstrated. Pseudotsuga menziesii Mirb and Larix decidua Mill are species with one of the longest microsporogenesis processes known in plants. The presence of bizonal bodies in both species may be linked to the intensification of the splicing processes in microspores with an exceptionally long cell cycle. The third type of structure is dense bodies, whose morphology and degree of silver staining strongly indicate their functional and spatial relationship to the dense part of bizonal bodies.







Similar content being viewed by others
References
Barlow PW (1981) Argyrophilic intranuclear bodies in plant cells. Experienta 37:1017–1018
Beven AF, Simpson GG, Brown JWS, Shaw PJ (1995) The organization of spliceosomal components in the nuclei of higher plants. J Cell Sci 108:509–518
Biliński SM, Kloc M (2002) Accessory nuclei revisited: the translocation of snRNPs from the germinal vesicle to the periphery of the future embryo. Chromosoma 111:62–68
Boundonck K, Dolan L, Shaw PJ (1999) The movement of coiled bodies visualized in living plant cells by the green fluorescent protein. Mol Biol Cell 10:2297–2307
Carmo-Fonseca M (2002) New clues to the function of the Cajal body. EMBO Rep 3(8):726–727
Chen M, Schwab R, Chory J (2003) Characterization of the requirements for localization of phytochrome B to nuclear bodies. Proc Natl Acad Sci USA 100(24):14493–14498
Chwirot WB, Górska-Brylass A (1981) Variation of total protein content and protein synthesis during microsporogenesis in Larix europea DC. Acta Soc Bot Pol 50:33–38
Cmarko D, Verschure PJ, Martin TE, Dahmus ME, Krause S, Fu XD, van Driel R, Fakan S (1999) Ultrastructural analysis of transcription and splicing in the cell nucleus after bromo-UTP microinjection. Mol Biol Cell 10:211–223
Collier S, Pendle A, Boudonck K, van Rij T, Dolan L, Shaw P (2006) A distant coilin homologue is required for the formation of cajal bodies in Arabidopsis. Mol Biol Cell 7(7):2942–2951
Darzacq X, Jady BE, Verheggen C, Kiss AM, Bertrand E, Kiss T (2002) Cajal body-specific small nuclear RNAs: a novel class of 2’-Omethylation and pseudouridylation guide RNAs. EMBO J 3:2746–2756
Docquier S, Tillemans V, Deltour R, Motte P (2004) Nuclear bodies and compartmentalization of pre-mRNA splicing factors in higher plants. Chromosoma 112(5):255–266
Fakan S (2004) The functional architecture of the nucleus as analysed by ultrastructural cytochemistry. Histochem Cell Biol 122(2):83–93
Fakan S, van Driel R (2007) The perichromatin region: a functional compartment in the nucleus that determines large-scale chromatin folding. Semin Cell Dev Biol 18(5):676–681
Fang Y, Spector DL (2007) Identification of nuclear dicing bodies containing proteins for microRNA biogenesis in living Arabidopsis plants. Curr Biol 17(9):818–823
Frey MR, Matera AG (2001) RNA-mediated interaction of Cajal bodies and U2 snRNA genes. J Cell Biol 154:499–509
Gall JG (1991) Spliceosomes and snurposomes. Science 252:1499–1500
Gall JG (2000) Cajal bodies: the first 100 years. Annu Rev Cell Dev Biol 16:273–300
Gall JG, Bellini M, Wu Z, Murphy C (1999) Assembly of the nuclear transcription and processing machinery: Cajal bodies (coiled bodies) and transcriptosomes. Mol Biol Cell 10:4385–4402
Górska-Brylass A, Wróbel B (1978) An electron microscope study of extranucleolar ribonucleoprotein bodies in themicrospore of Larix europea L. Soc Bot Fr Actualites Bot 1–2:23–25
Jennane A, Thiry M, Goessens G (1999) Identification of coiled body-like structures in meristematic cells of Pisum sativum cotyledonary buds. Chromosoma 108:132–142
Lafontaine JG (1965) A light and electron microscopic study of small spherical nuclear bodies in meristematic cells of Allium cepa, Vicia faba and Raphanus sativus. J Cell Biol 26:1–24
Martin M, Moreno Diaz de la Espina S, Jiménez-Garcia LF, Fernández-Gómez ME, Medina FJ (1992) Further investigations on the functional role of two nuclear bodies in onion cells. Protoplasma 167:175–182
Mascarenhas JP (1989) The male gametophyte of flowering plants. Plant Cell 1:657–664
Niedojadło J, Górska-Brylass A (2003) New type of snRNP containing nuclear bodies in plant cells. Biol Cell 95(5):303–310
Onodera Y, Haag JR, Ream T, Nunes PC, Pontes O, Pikaard CS (2005) Plant nuclear RNA polymerase IV mediates siRNA and DNA methylation-dependent heterochromatin formation. Cell 120:613–622
Ramon y Cajal S (1903) Un sencillo metodo de coloracion seletiva del reticulo protoplasmaticoy sus efectos en los diversos organos nerviosos de vertebrados e invertebrados. Trab Lab Invest Biol (Madrid) 2:129–221
Risueño MC, Medina FJ (1986) The nucleolar structure in plant cells. Servicio Editional Universidad del Pais Vasco, Bilbao
Seguí-Simarro JM, Bárány I, Suárez R, Fadón B, Testillano PS, Risueño MC (2006) Nuclear bodies domain changes with microspore reprogramming to embryogenesis. Eur J Histochem 50(1):35–44
Shaw PJ, Brown JW (2004) Plant nuclear bodies. Curr Opin Plant Biol 7(6):614–620
Smoliński DJ, Górska-Brylass A (1996) Plant “coiled body” after in situ hybrydisation and immunocytochemical investigations. Folia Histol Cytol 34[Suppl2]:90
Smoliński DJ, Niedojadło J, Noble A, Górska-Brylass A (2007) Additional nucleoli and NOR activity during meiotic prophase I in larch (Larix decidua Mill.). Protoplasma 232(1–2):109–120
Straatman KR, Schel JH (2001) Distribution of splicing proteins and putative coiled bodies during pollen development and androgenesis in Brassica napus L. Protoplasma 216:191–200
Verheggen C, Lafontaine DL, Samarsky D, Mouaikel J, Blanchard JM, Bordonné R, Bertrand E (2002) Mammalian and yeast U3 snoRNPs are matured in specific and related nuclear compartments. EMBO J 21(11):2736–2745
Williams LM, Jordan EG, Barlow PW (1983) The ultrastructure of nuclear bodies in interphase plant cell nuclei. Protoplasma 118:95–103
Wróbel B, Stoynova E (1992) The effect of kinetin on the nucleolar cycle in the meristematic root cells of Lupinus luteus L. Cell Biol Int Rep 16:985–992
Wu Z, Gall JG (1997) “Micronucleoli” in the Xenopus germinal vesicle. Chromosoma 105:438–443
Zienkiewicz K, Niedojadło J (2004) Cajal bodies-present opinions about nature and function. Postępy Biologii Komórki 31(2):313–331
Acknowledgements
We wish to thank Prof. A. Górska-Brylass for inspiration to study Douglas microsporogenesis. We thank Dr. A. Pombo (University of Oxford) for the AF-ANA serum and Dr. C. Sharp for his revision of the English. This work has been supported by the Chancellor of Nicolaus Copernicus University grant 505-B.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Niedojadło, J., Dominowska, K. & Bednarska, E. Nuclear bodies in Douglas fir (Pseudotsuga menziesii Mirb.) microspores. Protoplasma 234, 77–85 (2008). https://doi.org/10.1007/s00709-008-0018-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-008-0018-3