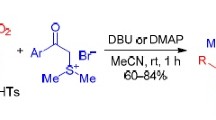
Summary.
Carbanions of chloromethyl 4-tolylsulfone, bromo- and chloromethanesulfomorpholide, and neopentyl chloromethanesulfonate react with benzonaphthyridines and their N-oxides via two pathways: vicarious nucleophilic substitution of hydrogen and annelation. The results are rationalized in terms of the negative charge delocalization in the intermediate σ-adducts.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received October 15, 2001. Accepted (revised) November 26, 2001
Rights and permissions
About this article
Cite this article
Bachowska, B. The Products of Vicarious Nucleophilic Substitution and Annelation in the Reactions of α-Haloalkyl Carbanions with Benzonaphthyridines and their N-Oxides. Monatshefte fuer Chemie 133, 1071–1076 (2002). https://doi.org/10.1007/s007060200076
Issue Date:
DOI: https://doi.org/10.1007/s007060200076