Summary.
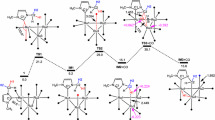
Activation of molecular hydrogen by triruthenium carbonyl clusters bearing acenaphthylene or aceanthrylene as the face-capping ligand results in partial hydrogenation of these polyaromatic ligands, giving the corresponding hydride carbonyl clusters bound to hydrogenated acenaphthylene or aceanthrylene. The first isolation of 4,5-dihydroacenaphthylene or 4,5-dihydroaceanthrylene was achieved by treatment of the resulting hydride carbonyl clusters with CO. Similarly, triruthenium carbonyl species bearing substituted azulenes as the face-capping moiety gives the corresponding hydride carbonyl cluster bearing partially hydrogenated azulenes at room temperature; the corresponding partial hydrogenation of azulenes does not occur with di- or tetraruthenium homologues. Hydrogenation of di-, tri-, and tetraruthenium carbonyl clusters bearing substituted azulenes at elevated temperatures furnishes hydrogenative cluster fragmentation to mononuclear ruthenium hydrides; the stereochemistry of these complexes suggests that the reaction takes place through the activation of H2 by the multinuclear species. The roles of the acenaphthylene or azulene ligands as hemilabile μ3-ligands for triruthenium carbonyl clusters are discussed.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received April 28, 2000. Accepted May 9, 2000
Rights and permissions
About this article
Cite this article
Nagashima, H. Facile Hydrogenation of Acenapthylenes and Azulenes on the Face of a Triruthenium Carbonyl Moiety: Discovery of Specific Reactions on the Cluster Framework Providing Unique Insight for Cluster Catalysis. Monatshefte fuer Chemie 131, 1225–1239 (2000). https://doi.org/10.1007/s007060070003
Issue Date:
DOI: https://doi.org/10.1007/s007060070003