Abstract
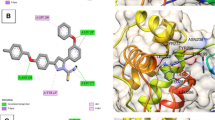
2-Cyanopyrimidine (2-CN-Pym), pyrimidine-2-carboximidamide (Pym-2-cia) and 2,4,6-tris(2-pyrimidyl)-1,3,5-triazine (TPymT), which are related to each other through chemical transformations from 2-CN-Pym through Pym-2-cia to TPymT, were computationally studied. The structures of all the reported compounds were optimized by the DFT calculations to reveal their fine features (electronic and optical). ADMET properties of 2-CN-Pym, Pym-2-cia and TPymT were also predicted using a set of online tools (SwissADME, BOILED-Egg and ProTox-II). Potential inhibition activity of 2-CN-Pym, Pym-2-cia and TPymT toward a series of the SARS-CoV-2 proteins was studied using a molecular docking approach, which revealed that for the applied proteins, both 2-CN-Pym and Pym-2-cia are the best inhibitors of RdRp-RNA, while TPymT exhibits the best activity toward nonstructural protein 14 (N7-MTase).
Graphical abstract










Similar content being viewed by others
Data availability
Data available on request from the authors.
References
Taylor RD, MacCoss M, Lawson AD (2014) J Med Chem 57:5845
Kumar S, Narasimhan B (2018) Chem Cent J 12:38
Liu H, Long S, Rakesh KP, Zha GF (2020) Eur J Med Chem 185:111804
Morandini A, Spadati E, Leonetti B, Sole R, Gatto V, Rizzolio F, Beghetto V (2021) RSC Adv 11:28092
Sun X, Cao Z, Sun Y (2009) Ind Eng Chem Res 48:607
Pfaffenroth C, Winkel A, Dempwolf W, Gamble LJ, Castner DG, Stiesch M, Menzel H (2011) Macromol Biosci 11:1515
Hassan G, Forsman N, Wan X, Keurulainen L, Bimbo LM, Johansson LS, Stehl S, Van Charante F, Chrubasik M, Prakash AS, Johansson LS, Mullen DC, Zimmermann R, Werner C, Yli-Kauhaluoma J, Coenye T, Saris PEJ, Österberg M, Moreira VM (2020) ACS Appl Biomater 3:4095
Duan J, Jiang G (2022) Polymers (Basel) 14:213
Keeley A, Ábrányi-Balogh P, Hrast M, Imre T, Ilaš J, Gobec S, Keserű GM (2018) Arch Pharm 351:1800184
Keeley A, Ábrányi-Balogh P, Keserű GM (2019) MedChemComm 10:263
Available online: https://covid19.who.int/. Accessed 31 Aug 2023
Wahl A, Gralinski LE, Johnson CE, Yao W, Kovarova M, Dinnon KH, Liu H, Madden VJ, Krzystek HM, De C, White KK, Gully K, Schäfer A, Zaman T, Leist SR, Grant PO, Bluemling GR, Kolykhalov AA, Natchus MG, Askin FB, Painter G, Browne EP, Jones CD, Pickles RJ, Baric RS, Garcia JV (2021) Nature 591:451
Cox RM, Wolf JD, Plemper RK (2021) Nat Microbiol 6:11
Rosenke K, Hansen F, Schwarz B, Feldmann F, Haddock E, Rosenke R, Barbian K, Meade-White K, Okumura A, Leventhal S, Hawman DW, Ricotta E, Bosio CM, Martens C, Saturday G, Feldmann H, Jarvis MA (2021) Nat Commun 12:2295
Kabinger F, Stiller C, Schmitzová J, Dienemann C, Kokic G, Hillen HS, Höbartner C, Cramer P (2021) Nat Struct Mol Biol 28:740
Sharov AV, Burkhanova TM, Taskın Tok T, Babashkina MG, Safin DA (2022) Int J Mol Sci 23:1508
Rai GS, Maru J (2020) Chem Heterocycl Comp 56:1517
Kalil AC, Mehta AK, Patterson TF, Erdmann N, Gomez CA, Jain MK, Wolfe CR, Ruiz-Palacios GM, Kline S, Regalado Pineda J, Luetkemeyer AF, Harkins MS, Jackson PEH, Iovine NM, Tapson VF, Oh MD, Whitaker JA, Mularski RA, Paules CI, Ince D, Takasaki J, Sweeney DA, Sandkovsky U, Wyles DL, Hohmann E, Grimes KA, Grossberg R, Laguio-Vila M, Lambert AA, Lopez de Castilla D, Kim E, Larson L, Wan CR, Traenkner JJ, Ponce PO, Patterson JE, Goepfert PA, Sofarelli TA, Mocherla S, Ko ER, Ponce de Leon A, Doernberg SB, Atmar RL, Maves RC, Dangond F, Ferreira J, Green M, Makowski M, Bonnett T, Beresnev T, Ghazaryan V, Dempsey W, Nayak SU, Dodd L, Tomashek KM, Beigel JH (2021) Lancet Resp Med 9:1365
Zhang L, Qu Z, Wu J, Yao S, Zhang Q, Zhang T, Mo L, Yao Q, Xu Y, Chen R (2021) Eur J Med Chem 214:113188
Triazine Market Growth By Forthcoming Developments 2022. Business Insights with Covid-19 Impact, Top Key Players and Detailed Analysis of Market with Production Capacity Estimates till 2026. https://rivercountry.newschannelnebraska.com/story/46138694/triazine-market-growth-by-forthcoming-developments-2022-business-insights-with-covid-19-impact-top-key-players-and-detailed-analysis-of-market-with. Accessed 12 July 2022
Douangamath A, Fearon D, Gehrtz P, Krojer T, Lukacik P, Owen CD, Resnick E, Strain-Damerell C, Aimon A, Ábrányi-Balogh P, Brandão-Neto J, Carbery A, Davison G, Dias A, Downes TD, Dunnett L, Fairhead M, Firth JD, Jones SP, Keeley A, Keserü GM, Klein HF, Martin MP, Noble MEM, O’Brien P, Powell A, Reddi RN, Skyner R, Snee M, Waring MJ, Wild C, London N, von Delft F, Walsh MA (2020) Nat Commun 11:5047
Cho CC, Li SG, Lalonde TJ, Yang KS, Yu G, Qiao Y, Xu S, Ray LW (2022) ChemMedChem 17:202100455
Safin DA, Tumanov NA, Leitch AA, Brusso JL, Filinchuk Y, Murugesu M (2015) CrystEngComm 17:2190
Safin DA, Xu Y, Korobkov I, Bryce DL, Murugesu M (2013) CrystEngComm 15:10419
Safin DA, Burgess K, Korobkov I, Bryce DL, Murugesu M (2014) CrystEngComm 16:3466
Safin DA, Holmberg RJ, Burgess KMN, Robeyns K, Bryce DL, Murugesu M (2015) Eur J Inorg Chem 2015:441
Safin DA, Pialat A, Korobkov I, Murugesu M (2015) Chem Eur J 16:6144
Safin DA, Pialat A, Leitch A, Tumanov NA, Korobkov I, Filinchuk Y, Brusso JL, Murugesu M (2015) Chem Commun 51:9547
Safin DA, Szell PMJ, Keller A, Korobkov I, Bryce DL, Murugesu M (2015) New J Chem 39:7147
Safin DA, Frost JM, Murugesu M (2015) Dalton Trans 44:20287
Ay B, Şahin O, Saygıdeğer Demir B, Saygideger Y, López-de-Luzuriaga JM, Mahmoudi G, Safin DA (2020) New J Chem 44:9064
Shiryaev AA, Goncharenko AN, Burkhanova TM, Alkhimova LA, Babashkina MG, Chandrasekaran R, Safin DA (2021) J Iran Chem Soc 18:2897
Babashkina MG, Frontera A, Kertman AV, Saygideger Y, Murugavel S, Safin DA (2022) J Iran Chem Soc 19:85
Burkhanova TM, Babashkina MG, Taskin-Tok T, Sharov AV, Safin DA (2022) J Iran Chem Soc 19:1979
Geerlings P, De Proft F, Langenaeker W (2003) Chem Rev 103:1793
Pérez P, Domingo LR, Aizman A, Contreras R (2007) Theor Comput Chem 19:139
Abraham JP, Sajan D, Joe IH, Jayakumar VS (2008) Spectrochim Acta A 71:355
Karamanis P, Pouchan C, Maroulis G (2008) Phys Rev A 77:013201
Eme A, Sağdınç SM (2017) J Mol Struct 1147:322
Banerjee P, Eckert AO, Schrey AK (2018) Nucleic Acids Res 46:w257
Daina A, Michielin O, Zoete V (2017) Sci Rep 7:42717
Diana A, Zoete V (2016) ChemMedChem 11:1117
Meng X-Y, Zhang H-X, Mezei M, Cui M (2011) Curr Comp Aid Drug 7:146
Ding X-C, He J, Zhang X, Jiang C, Sun Y, Zhang Y, Chen Q, He H, Li W, Xie J, Liu Z, Gao Y (2021) Front Immunol 12:693775
Reynolds CH, Tounge BA, Bembenek SD (2008) J Med Chem 51:2432
Schultes S, de Graaf C, Haaksma EE, de Esch IJP, Leurs R, Krämer O (2010) Drug Discovery Today: Technol 7:e157
Hughes JP, Rees S, Kalindjian SB, Philpott KL (2011) Br J Pharmacol 162:1239
Hopkins AL, Keserü GM, Leeson PD, Rees DC, Reynolds CH (2014) Nat Rev Drug Discov 13:105
Abdul-Hammed M, Adedotun IO, Falade VA, Adepoju AJ, Olasupo SB, Akinboade MW (2021) VirusDis 32:642
Dennington R, Keith TA, Millam JM (2016) GaussView, Version 6.0, Semichem Inc., Shawnee Mission, KS
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada Y, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery Jr JA, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Keith T, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas O, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2016) Gaussian 09, Revision D.01
Krishnan R, Binkley JS, Seeger R, Pople JA (1980) J Chem Phys 72:650
Becke AD (1993) J Chem Phys 98:5648
Frisch MJ, Pople JA, Binkley JS (1984) J Chem Phys 8:3265
Trott O, Olson AJ (2010) J Comput Chem 31:455
Eberhardt J, Santos-Martins D, Tillack AF, Forli S (2021) J Chem Inf Modeling 61:3891
Rose Y, Duarte JM, Lowe R, Segura J, Bi C, Bhikadiya C, Chen L, Rose AS, Bittrich S, Burley SK, Westbrook JD (2021) J Mol Biol 433:166704
Accelrys Software Inc. Discovery Studio Modeling Environment; Release 3.5; Accelrys Software Inc., San Diego, CA, USA, 2013
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Taskin-Tok, T., Safin, D.A. Computational studies of closely related 2-cyanopyrimidine, pyrimidine-2-carboximidamide and 2,4,6-tris(2-pyrimidyl)-1,3,5-triazine with a potency against SARS-CoV-2. Monatsh Chem 155, 57–71 (2024). https://doi.org/10.1007/s00706-023-03133-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-023-03133-9