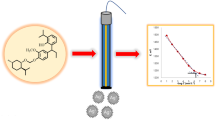
Abstract
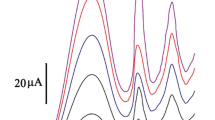
An electrochemical sensor based on (E)-2-[[(4-hydroxyphenyl)imino]methyl]phenol (anil) was developed for the detection of iodide anions. The complexation ability of the anil with I−, Br−, Cl−, CN−, SCN−,\({\mathrm{NO}}_{3}^{-}\), CH3COO−,\({\mathrm{SO}}_{4}^{2-}\), F−, and S2− was explored by UV–Vis spectroscopy. The results showed that the proposed probe has high sensibility for the detection of I−, with the absorption intensity changing due to the formation of anil–I− complex. The anil and iodide anion bind by a 1:1 complex stoichiometry and the association constant is determined to be 8.5 × 103 M−1. In addition, the sensing film was elaborated by drop coating on a platinum electrode. The electrochemical sensors for the detection of I− were analyzed by DPV. It showed a linear response in the range of 1 × 10–7 to 1 × 10–4 M, with a low detection limit of 4 × 10–8 M. The sensor shows good analytical performance. The developed sensor is suitable for application in water quality monitoring.
Graphical abstract









Similar content being viewed by others
References
Chen J, Liu X, Hou X, Hou X, Chen Y, Xing F, Feng L (2020) Anal Bioanal Chem 412:2893
Boali AA, Mansha M, Waheed A, Ullah N (2018) J Chin Inst Chem Eng 91:420
Wang H, Lu Q, Liu Y, Li H, Zhang Y, Yao S (2017) Sens Actuators B Chem 250:429
Indumathy R, Parameswaran PS, Aiswarya CV, Nair BU (2014) Polyhedron 75:22
Chen J, Lin Q, Li Q, Li WT, Zhang YM, Wei T-B (2016) RSC Adv 6:86627
Wu Y, Chen W, Shen J, Tan L, L’Abbe MR, Pearce EN, Wang W, Tian X, Wang W, Zhang W (2018) Nutr Res Rev 55:72
Tuccilli C, Baldini E, Truppa E, D’Auria B, De Quattro D, Cacciola G, Aceti T, Cirillo G, Faiola A, Indigeno P, D’Aliesio L, Gazzellone F, Bononi M, D’Armiento E, Giovanni Carbotta G, Pironi D, Catania A, Sorrenti S, Ulisse S (2018) J Nutr 50:60
Zhang Y, Zhao W, Zhao Y, Mao Y, Su T, Zhong Y, Wang S, Zhai R, Cheng J, Fang X, Zhu J, Yang H (2020) J Proteome Res 19:2539
Niaz A, Bibi A, Huma, Zaman MI, Khan M, Rahim A (2018) J Mol Liq 249:1047
Kaykhaii M, Sargazi M (2014) Spectrochim Acta A Mol Biomol Spectrosc 121:173
Rong L, Lim LW, Takeuchi T (2013) Microchem J 108:113
Chen Z, Sun R, Feng S, Wang D, Liu H (2020) ACS Appl Mater Interfaces 12:11104
Sharma PR, Pandey S, Soni VK, Choudhary G, Sharma RK (2019) Supramol Chem 31:634
Sharma PR, Pandey S, Malik A, Choudhary G, Soni VK, Sharma RK (2021) RSC Adv 11:26644
Ghosh K, Saha I (2010) Supramol Chem 22:311
Panja S, Kumar A, Misra N, Ghosh S, Raza R, Ghosh K (2021) ChemistrySelect 6:6353
Li G-B, Huang X-M, Pan R-K, Liu S-G (2020) Spectrochim Acta A Mol Biomol Spectrosc 229:117941
Kaur R, Dhaka G, Singh P, Rana S, Kaur N (2020) Arab J Chem 13:6931
Abdel-Haleem FM, Shehab OR (2016) Electroanalysis 28:800
Berhanu AL, Gaurav, Mohiuddin I, Malik AK, Aulakh JS, Kumar V, Kim KH (2019) TrAC Trends Anal Chem 116:74
Alreja P, Kaur N (2018) Inorg Chim Acta 480:127
Singh AK, Gupta VK, Gupta B (2007) Anal Chim Acta 585:171
Cho E, Perebikovsky A, Benice O, Holmberg S, Madou M, Ghazinejad M (2018) Sensors 18:1486
Alizadeh T, Zargr F (2020) Mater Chem Phys 240:122118
Khunseeraksa V, Kongkaew S, Thavarungkul P, Kanatharana P, Limbut W (2020) Microchim Acta 187:591
Bao ZL, Zhong H, Li XR, Zhang AR, Liu YX, Chen P, Cheng ZP, Qian HY (2021) Sens Actuators B Chem 345:130319
Tang X, Yu H, Bui B, Wang L, Xing C, Wang S, Chen M, Hu Z, Chen W (2021) Bioact Mater 6:1541
Ali M, Maurya RR, Singh J, Negi PS, Rajor HK, Bahadur I (2022) Colloids Surf, A 639:128369
Chaabene M, Agren S, Allouche AR, Chaâbane RB, Lahcinie M, Baouab MHV (2019) Appl Organomet Chem 33:e5218
Acknowledgements
Financial support of this research from the Ministry of Higher Education and Scientific Research of Tunisia.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Echabaane, M., Moulahi, N., Baouab, M.H.V. et al. Optical, electrochemical, and sensing properties of (E)‐2‐[[(4-hydroxyphenyl)imino]methyl]phenol for the detection of iodide ions. Monatsh Chem 153, 561–567 (2022). https://doi.org/10.1007/s00706-022-02937-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-022-02937-5