Abstract
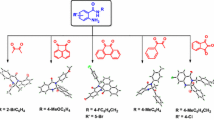
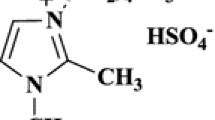
In this paper, an effective procedure is reported for the synthesis of 3-substituted indoles via the one-pot three-component reaction of an aldehyde, malononitrile or ethyl cyanoacetate and indole in the presence of [bmim]HSO4 in ethanol under reflux condition. The advantages of this protocol are the synthesis of some novel 3-substituted indoles containing furyl, 4-hydroxyphenyl, and styryl nuclei that are very important in pharmaceutical and drug discovery research in comparison to previously reported results, and the use of non-halogenated ionic liquid.
Graphic abstract

Similar content being viewed by others
References
Nunes PSG, Vidal HAD, Corrêa AG (2020) Org Biomol Chem 18:7751. https://doi.org/10.1039/d0ob01631d
Lamberth C (2020) Bioorg Med Chem 28:115471. https://doi.org/10.1016/j.bmc.2020.115471
(a) Domling A (2006) Chem Rev 106:17. https://doi.org/10.1021/cr0505728; (b) Toure BB, Hall DG (2009) Chem Rev 109:4439. https://doi.org/10.1021/cr800296p
Schreiber SL (2000) Science 287:1964. https://doi.org/10.1126/science.287.5460.1964
Carballares S, Espinosa JF (2005) Org Lett 7:2329. https://doi.org/10.1021/ol050553k
Domling A, Wang W, Wang K (2012) Chem Rev 112:3083. https://doi.org/10.1021/cr100233r
Shiri M (2012) Chem Rev 112:3508. https://doi.org/10.1021/cr2003954
Rajesh UC, Wang J, Prescott S, Tsuzuki T, Rawat DS (2015) ACS Sustain Chem Eng 3:9. https://doi.org/10.1021/sc500594w
Kumar A, Gupta MK, Kumar M (2012) Green Chem 14:290. https://doi.org/10.1039/c1gc16297g
Qu Y, Ke F, Zhou L, Li Z, Xiang H, Wu D, Zhou X (2011) Chem Commun 47:3912. https://doi.org/10.1039/c0cc05695b
Wang L, Huang M, Zhu X, Wan Y (2013) Appl Catal A Gen 454:160. https://doi.org/10.1016/j.apcata.2012.12.008
He Y-H, Cao J-F, Li R, Xiang Y, Yang D-C, Guan Z (2015) Tetrahedron 71:9299. https://doi.org/10.1016/j.tet.2015.10.027
Anselmo D, Escudero-Ad EC, Martínez Belmonte M, Kleij AW (2012) Eur J Inorg Chem 2012:4694. https://doi.org/10.1002/ejic.201200150
Vekariya RL (2017) J Mol Liq 227:44. https://doi.org/10.1016/j.molliq.2016.11.123
Shekarchi M, Behbahani FK (2020) Russ J Org Chem 56:894. https://doi.org/10.1134/s1070428020050243
Mohammadi B, Behbahani FK, Marandi GB, Mirza B (2021) Phosphorus. Sulfur Silicon Relat Elem 196:54. https://doi.org/10.1080/10426507.2020.1800702
Bahekar SS, Kotharkar SA, Shinde DB (2004) Mendeleev Commun 14:210. https://doi.org/10.1070/mc2004v014n05abeh001895
Totten GE, Shah R, Forester D (2019) Fuels and lubricants handbook: technology, properties, performance, and testing, 2nd edn. Portland State University Library. https://doi.org/10.1520/mnl37-2nd-eb
Ghohe N, Tayebee M, Amini R, Mostafa M, Osatiashtiani A, Isaacs MA, Lee AF (2017) Tetrahedron 73:5862. https://doi.org/10.1016/j.tet.2017.08.030
Omidi M, Amrollahi MA (2017) C R Chimie 20:549. https://doi.org/10.1016/j.crci.2016.09.004
Pradhan S, Saha JM (2017) New J Chem 41:6616. https://doi.org/10.1039/c7nj00249a
Jiang H, Wang L, Xie J (2016) J Chem Res 40:338. https://doi.org/10.3184/174751916x14622783380474
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shekarchi, M., Behbahani, F.K. & Shekarchi, M. Green route for the synthesis of 3-substituted indoles using [bmim]HSO4 as non-halogenated ionic liquid. Monatsh Chem 152, 659–664 (2021). https://doi.org/10.1007/s00706-021-02782-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-021-02782-y