Abstract
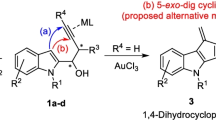
Gold-catalyzed or NaH-supported formation mechanisms of pyrazine and diazepines were investigated computationally. The structural properties of these heterocyclic compounds were studied in the gas phase and various solvents. Density functional theory, including B3LYP, WB97X-D, M06-2X, and M06 hybrid functional methods, was used to locate and discuss the energetics of the intermediates and the transition states. The nature of the substituents attached to the terminal alkyne played an essential role in these mechanisms. Electron-donating or electron-withdrawing substituents indicated in which direction the cyclization would evolve. Natural bond orbital analysis was performed to determine the chemoselectivity of the products.
Graphic abstract


















Similar content being viewed by others
References
Eicher T, Hauptmann S, Speicher A (2003) The chemistry of heterocycles, 2nd edn. Wiley-VCH Verlag GmbH & Co
Bartoli G, Dalpozzo R, Nardi M (2014) Chem Soc Rev 43:4728
Likhosherstov AM, Filippova OV, Peresada VP, Kryzhanovskii SA, Vititnova MB, Kaverina NV, Reznikov KM (2003) Pharm Chem J 37:6
Seredenin SB, Voronina TA, Beshimov A, Peresada VP, Likhosherstov AM (1997) Tetrahydropyrrolopyrazine with antiamnesic and antihypoxic activity. Patent RU 2099055, Dec 20, 1997; (1998) Chem Abstr 128:290245
Park S, Jung Y, Kim I (2014) Tetrahedron 70:7534
Vlahov IR, Qi L, Krishna R, Santhapuram H, Felten A, Parham GL, Zou N, Wang K, You F, Vaughn JF, Hahn SJ, Klein HF, Kleindl PJ, Reddy J, Reno D, Nicoson J, Leamon CP (2020) Bioorg Med Chem Lett 30:126987
Dimitriou E, Jones RH, Pritchard RG, Miller GJ, O’Brien M (2018) Tetrahedron 74:6795
Smith SG, Sanchez R, Zhou MM (2014) J Chem Biol 21:573
Basceken S, Balci M (2015) J Org Chem 80:3806
Basceken S, Kaya S, Balci M (2015) J Org Chem 80:12552
Çetinkaya Y, Balci M (2014) Tetrahedron Lett 55:6698
Ferrer C, Echavarren AM (2006) Angew Chem Int Ed 45:1105
Basceken S (2020) Struct Chem 31:1765
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Petersson GA, Nakatsuji H, Li X, Caricato M, Marenich A, Bloino J, Janesko BG, Gomperts R, Mennucci B, Hratchian HP, Ortiz JV, Izmaylov AF, Sonnenberg JL, Williams-Young D, Ding F, Lipparini F, Egidi F, Goings J, Peng B, Petrone A, Henderson T, Ranasinghe D, Zakrzewski VG, Gao J, Rega N, Zheng G, Liang W, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Throssell K, Montgomery JA, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Keith T, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Millam JM, Klene M, Adamo C, Cammi R, Ochterski JW, Martin RL, Morokuma K, Farkas O, Foresman JB, Fox DJ (2009) Gaussian 09, Revision E.01. Gaussian, Inc, Wallingford, CT
Kohn W, Sham L (1965) Phys Rev 140:A1133
Parr RG, Yang W (1995) Ann Rev Phys Chem 46:701
Kohn W, Becke AD, Parr RG (1996) J Phys Chem 100:12974
Chai JD, Gordon MH (2009) J Chem Phys 131:174105
Escrich CR, Davis RL, Jiang H, Stiller J, Johansen TK, Jørgensen KA (2013) Chem Eur J 19:2932
Silla JM, Duarte CJ, Rittner R, Freitas MP (2013) RSC Adv 3:25765
Zhao Y, Truhlar DG (2008) Theor Chem Acc 120:215
Hohenstein EG, Samuel ST, Sherrill CD (2008) J Chem Theory Comput 4:1996
Wadt WR, Hay PJ (1985) J Chem Phys 82:299
Dennington RD, Keith TA, Millam JM (2000) Semichem Inc
Fukui K (1981) Acc Chem Res 14:363
CYLview v1.0beta (2009) University of Sherbrooke, Canada
Miertuš S, Scrocco E, Tomasi J (1981) Chem Phys 55:117
Foster JP, Weinhold F (1980) J Am Chem Soc 102:7211
Reiher M, Salomon O, Hess BA (2001) Theor Chem Acc 107:48
Schultz N, Zhao Y, Truhlar DG (2005) J Phys Chem A 109:4388
Schultz N, Zhao Y, Truhlar DG (2005) J Phys Chem A 109:11127
Harvey JN (2006) Annu Rep Prog Chem Sect C Phys Chem 102:203
Dinesh VV, Marie EK, Igor VA (2016) J Am Chem Soc 138:2769
Rajaei I, Mirsattari SN (2018) J Mol Struct 1163:236
Najafi M, Morsali A, Bozorgmehr MR (2019) Struct Chem 30:715
Morgan KM, Gronert S (2000) J Org Chem 65:1461
Raspoet G, Vanquickenborne LG, Nguyen MT (1997) J Am Chem Soc 119:2552
Ozen SA, Erdem SS, Aviyente V (1998) Struct Chem 9:15
Cramer CJ, Truhlar DG (1991) J Am Chem Soc 113:8305
Wiest O, Houk KN (1994) J Org Chem 59:7582
Wong MW, Frisch MJ, Wiberg KB (1991) J Am Chem Soc 113:4776
Acknowledgements
Financial support from the Scientific Research Department (BAP, Project No. FEF.19001.19.003) of Hitit University is gratefully acknowledged. Thanks are also due to the Scientific and Technological Research Council of Turkey (TUBITAK) to use its ULAKBIM/TRUBA high performance and grid computing centre.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Basceken, S. Theoretical modeling of homogenous gold-catalyzed or NaH-supported alkyne cyclization. Monatsh Chem 152, 607–624 (2021). https://doi.org/10.1007/s00706-021-02775-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-021-02775-x