Abstract
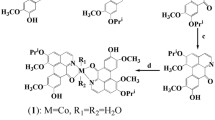
Naphthalimide has emerged as an interesting DNA intercalator and possessed attracting antitumor properties. In this context, naphthalimide group was linked to platinum(IV) core to construct a series of new mono naphthalimide platinum(IV) derivatives. The title compounds exert effective antitumor activities to the tested tumor cells lines in vitro, especially the one with propionyl chain displays comparable or even better bioactivities than platinum(II) reference drugs cisplatin and oxaliplatin. Moreover, the mono naphthalimide platinum(IV) derivative displays comparable tumor growth inhibitory competence against CT26 xenograft tumors in BALB/c mice in vivo without severe toxic effects in contrast to oxaliplatin. A dual DNA damage mechanism was proven for the title complex. Both naphthalimide ligand and the liberated platinum(II) moiety could generate DNA lesions to tumor cells synergistically and active the apoptotic pathway by up-regulating the expression of caspase 9 and caspase 3. Meanwhile, the conversion of platinum(II) drug into tetravalent form by incorporating naphthalimide moiety increases the uptake of platinum in whole cells and DNA remarkably. All these facts might be the factors for the title platinum(IV) complexes to overcome platinum(II) drug resistance. Additionally, the mono naphthalimide platinum(IV) complex could interact with human serum albumin by hydrogen bond and van der Waals force which would further influence their storage, transport and bioactivities.
Graphic abstract












Similar content being viewed by others
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A (2018) CA Cancer J Clin 68:394
Allemani C, Matsuda T, Carlo DV, Harewood R, Matz M, Nikšić M, Bonaventure A, Valkov M, Johnson CJ, Estève J, Ogunbiyi OJ, Silva GA, Chen WQ, Eser S, Engholm G, Stiller CA, Monnereau A, Woods RR, Visser O, Lim GH, Aitken J, Weir HK, Coleman MP (2018) Lancet 391:1023
Wong E, Christen MG (1999) Chem Rev 99:2451
Allardyce CS, Dyson PJ (2016) Dalton Trans 45:3201
Wheate NJ, Walker S, Craig GE, Oun R (2010) Dalton Trans 39:8113
Oun R, Mouss YE, Wheate NJ (2018) Dalton Trans 47:6645
Johnstone TC, Suntharalingam K, Lippard SJ (2016) Chem Rev 116:3436
Gibson D (2016) Dalton Trans 45:12983
Schreiber-Brynzak E, Pichler V, Heffeter P, Hanson B, Theiner S, Lichtscheidl-Schultz I, Kornauth C, Bamonti L, Dhery V, Groza D, Berry D, Berger W, Galanski M, Jakupec MA, Keppler BK (2016) Metallomics 8:422
Tan XX, Li GS, Wang QP, Wang BQ, Li DC, Wang PG (2018) Prog Chem 30:831
Wexselblatt E, Gibson D (2012) J Inorg Biochem 117:220
Basu U, Banik B, Wen R, Pathak RK, Dhar S (2016) Dalton Trans 45:12992
Gabano E, Ravera M, Osella D (2014) Dalton Trans 43:9813
Liu H, Ma J, Li Y, Yue K, Li L, Xi Z, Zhang X, Liu J, Feng K, Ma Q, Liu S, Guo S, Wang PG, Wang C, Xie S (2019) J Med Chem 62:11324
Ma J, Liu H, Xi Z, Hou J, Li Y, Niu J, Liu T, Bi S, Wang X, Wang C, Wang J, Xie S, Wang PG (2018) Front Chem 6:386
Chen Z, Liang X, Zhang HY, Xie H, Liu JW, Xu YF, Zhu WP, Wang Y, Wang X, Tan SY, Kuang D, Qian XH (2010) J Med Chem 53:2589
Seliga R, Pilatova M, Sarissky M, Viglasky V, Walko M, Mojzis J (2013) Mol Biol Rep 40:4129
El-Azab AS, Alanazi AM, Abdel-Aziz NI, Al-Suwaidan IA, El-Sayed MAA, El-Sherbeny MA, Abdel-Aziz AAM (2013) Med Chem Res 22:2360
Chua EY, Davey GE, Chin CF, Dröge P, Ang WH, Davey CA (2015) Nucleic Acids Res 43:5285
Pérez JM, López-Solera I, Montero EI, Braña MF, Alonso C, Robinson SP, Navarro-Ranninger C (1999) J Med Chem 42:5482
Herrera JM, Mendes F, Gama S, Santos I, Navarro-Ranninger C, Cabrera S, Quiroga AG (2014) Inorg Chem 53:12627
Navas F, Mendes F, Santos I, Navarro RC, Cabrera S, Quiroga AG (2017) Inorg Chem 56:6175
Wang QP, Li GS, Liu ZF, Tan XX, Ding Z, Ma J, Li LJ, Li DC, Han J, Wang BQ (2018) Eur J Inorg Chem 40:4442
Wang QP, Tan XX, Liu ZF, Li GS, Zhang RY, Wei JJ, Wang SB, Li DC, Wang BQ, Han J (2018) Eur J Pharm Sci 124:127
Li GS, Zhang JF, Liu ZF, Wang QP, Chen Y, Liu M, Li DC, Han J, Wang BQ (2019) J Inorg Biochem 194:34
Wang QP, Chen Y, Li GS, Liu ZF, Ma J, Liu M, Li DC, Han J, Wang BQ (2019) Bioorg Med Chem 27:2112
Wang Q, Huang Z, Ma J, Lu X, Zhang L, Wang X, Wang PG (2016) Dalton Trans 45:10366
Ma J, Wang Q, Huang Z, Yang X, Nie Q, Hao W, Wang PG, Wang X (2017) J Med Chem 60:5736
Wang Q, Chen Y, Li G, Zhao Y, Liu Z, Zhang R, Liu M, Li D, Han J (2019) Bioorg Med Chem Lett 29:126670
Quaquebeke EV, Mahieu T, Dumont P, Dewelle J, Ribaucour F, Simon G, Sauvage S, Gaussin JF, Tuti J, Yazidi ME, Vynckt FV, Mijatovic T, Lefranc F, Darro F, Kiss R (2007) J Med Chem 50:4122
Göschl S, Schreiber-Brynzak E, Pichler V, Cseh K, Heffeter P, Jungwirth U, Jakupec MA, Bergerbcd W, Keppler BK (2017) Metallomics 9:309
Jungwirth U, Xanthos DN, Gojo J, Bytzek AK, Körner W, Heffeter P, Abramkin SA, Jakupec MA, Hartinger CG, Windberger U, Galanski M, Keppler BK, Berger W (2012) Mol Pharmacol 81:719
Wei Q, Dong JF, Zhao PR, Li MM, Cheng FL, Kong JM, Li LZ (2016) J Photochem Photobiol B 161:355
Zhao PR, Wei Q, Dong JF, Ding FF, Li JH, Li LZ (2016) J Coord Chem 69:2437
Li XL, Hu YJ, Wang H, Yu BQ, Yue HL (2012) Biomacromolecules 13:873
Yuan LX, Liu M, Liu GQ, Li DC, Wang ZP, Wang BQ, Han J, Zhang M (2017) Spectrochim Acta A 173:584
Yuan LX, Liu M, Sun B, Liu J, Wei XL, Wang ZP, Wang BQ, Han J (2017) J Mol Liq 248:330
Mayr J, Heffeter P, Groza D, Galvez L, Koellensperger G, Roller A, Alte B, Haider M, Berger W, Kowol CR, Keppler BK (2017) Chem Sci 8:2241
Yuan LX, Liu M, Shi YB, Yan H, Han J, Liu LY (2018) J Biomol Struct Dyn 37:2776
Acknowledgements
The work was supported by National Natural Science Foundation of China (No. 21807056), Natural Science Foundation of Shandong (No. ZR2017BH092), Doctoral Foundation of Liaocheng University (No. 318051635), Open Project of Shandong Collaborative Innovation Center for Antibody Drugs (No. CIC-AD1836, CIC-AD1835), National Science and Technology Major Project of China (No. 2017ZX09201-003) and Taishan Scholar Research Foundation. This work was also technically supported by Shandong Collaborative Innovation Center for Antibody Drugs and Engineering Research Center for Nanomedicine and Drug Delivery Systems.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, Z., Chen, Y., Liu, Z. et al. Synthesis and biological evaluation of new mono naphthalimide platinum(IV) derivatives as antitumor agents with dual DNA damage mechanism. Monatsh Chem 151, 353–367 (2020). https://doi.org/10.1007/s00706-020-02561-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-020-02561-1