Abstract
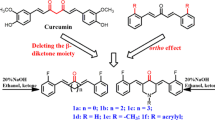
Curcumin, bearing two electrophilic α,β-unsaturated ketones, is a promising anticancer agent by an electrophilicity-based prooxidant strategy (ROS-generating) due to the Michael acceptors. Considering that ROS generation depends on Michael acceptor unit, we have designed and synthesized two series coumarin–chalcone hybrids containing one or two Michael acceptor units through Claisen–Schmidt condensation, and evaluated the cytotoxicity against A549 cells as well as ROS accumulation. (E)-3-[3-(2-Hydroxyphenyl)acryloyl]-2H-chromen-2-one was identified as the strongest ROS inducer and cytotoxic agent. The structure–activity relationships (SAR) indicated that the Michael acceptor unit was more important than the position of hydroxyl to the cytotoxicity mediated by increasing the cellular lever of ROS. In addition, (E)-3-[3-(2-hydroxyphenyl)acryloyl]-2H-chromen-2-one caused S-phase cell cycle arrest in A549 cells. Therefore, this work provides an example of coumarin–chalcone scaffold as cytotoxic agent by a prooxidant (ROS-generating agent) strategy.
Graphical abstract





Similar content being viewed by others
References
Zhao HP, Donnelly AC, Kusuma BR, Brandt GEL, Brown D, Rajewski RA, Vielhauer G, Holzbeierlein J, Cohen MS, Blagg BSJ (2011) J Med Chem 54:3839
Wu L, Wang X, Xu W, Farzaneh F, Xu R (2009) Curr Med Chem 16:4236
Dayam R, Gunla R, Al-Mawsawi LQ, Neamati N (2008) Med Res Rev 28:118
Basanagouda M, Shivashankar K, Kulkarni MV (2010) Eur J Med Chem 45:1151
Ishihara M, Yokote Y, Sakagami H (2006) Anticancer Res 26:2883
Wu X-Q, Huang C, Jia Y-M, Song B-A, Li J, Liu X-X (2014) Eur J Med Chem 74:717
Vianna DR, Hamerski L, Figueiró F, Bernardia A, Visentind LC, Piresc ENS, Teixeiraa HF (2012) Eur J Med Chem 57:268
Nasr T, Bondock S, Youns M (2014) Eur J Med Chem 76:539
Chen Y, Liu H-R, Liu H-S, Xia P, Qian K-D, Wu P-C (2012) Eur J Med Chem 49:74
Riveiro ME, Moglioni A, Vazquez R, Gomez N, Facorro G, Piehl L, Rubin de Celis E, Shayo C, Davio C (2008) Bioorg Med Chem 16:2665
Riveiro ME, Vazquez R, Moglioni A, Gomez N, Baldi A, Davio C, Shayo C (2008) Biochem Pharmacol 75:725
Vázquez R, Riveiro ME, Vermeulen M (2012) Bioorg Med Chem 20:5537
Batovska DI, Todorova IT (2010) Curr Clin Pharmacol 5:1
Singh P, Anand A, Kumar V (2014) Eur J Med Chem 85:758
Iwamura C, Shinoda K, Yoshimura M, Watanabe Y, Obata A, Nakayama T (2010) Allergol Int 59:67
Sahu NK, Balbhadra SS, Choudhary J, Kohli DV (2012) Curr Med Chem 19:209
Karthikeyana C, Moorthy NSHN, Ramasamyc S, Vanamc U, Manivannand E, Karunagaranc D, Trivedi P (2014) Recent Pat Anticancer 10:97
Gan FF, Kaminska KK, Yang H, Liew CY, Leow PC, So CL, Tu LN, Roy A, Yap CW, Kang TS (2013) Antioxid Redox Signal 19:1149
Emami S, Dadashpour S (2015) Eur J Med Chem 102:611
López-Lázaro M (2008) Mol Nutr Food Res 52:S103
Dai F, Liu G-Y, Li Y, Yan W-J, Wang Q, Yang J, Lu D-L (2015) Free Radic Biol Med 85:127
Chew EH, Nagle AA, Zhang Y, Scarmagnani S, Palaniappan P, Bradshaw TD, Holmgren A, Westwell AD (2010) Free Radic Biol Med 48:98
Zheng S, Santosh Laxmi Y, David E, Dinkova-Kostova AT, Shiavoni KH, Ren Y, Zheng Y, Trevino I, Bumeister R, Ojima I (2012) J Med Chem 55:4837
Wanare G, Aher R, Kawathekar N, Ranjan R, Kaushik NK, Sahal D (2010) Bioorg Med Chem Lett 20:4675
Moodley T, Momin M, Mocktar C, Kannigadu C, Koorbanally NA (2016) Magn Reson Chem 54:610
Aleem MAE, El-Remaily AA (2015) Chin J Catal 36:1124
Hamdi N, Fischmeister C, Carmen Puerta M, Valerga P (2011) Med Chem Res 20:522
Manaev AV, Chibisova TA, Traven VF (2006) Russ Chem Bull Int Ed 55:2226
Bian J-L, Li X, Xu L-L, Wang N, Qian X, You Q-D, Zhang X-J (2017) Eur J Med Chem 127:828
Cathcart R, Schwiers E, Ames BN (1983) Anal Biochem 134:111
Tang J-J, Fan GJ, Dai F, Ding D-J, Wang Q, Lu DL, Li R-R, Li X-Z (2011) Free Radic Biol Med 50:1447
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shang, Yj., Wei, Q. & Sun, Zb. Studying the cytotoxicity of coumarin–chalcone hybrids by a prooxidant strategy in A549 cells. Monatsh Chem 149, 2287–2292 (2018). https://doi.org/10.1007/s00706-018-2273-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-018-2273-0