Abstract
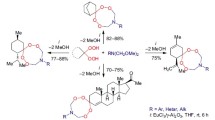
Obtaining cuparene-type sesquiterpenes is a significant synthetic challenge mainly because of the construction of the sterically hindered quaternary centres. We report herein the successful construction of such quaternary moiety by a highly regioselective opening of (+/−)-2,5-dimethoxy-4-methyl-α-methylstyrene oxide by the acetone silyl enol ether catalysed by Lewis acid. Additionally, an efficient epoxidation of the highly activated 2,5-dimethoxy-4-methyl-α-styrene was accomplished by modifications on the dioxirane-promoted epoxidation protocol. Intensive optimisations for both key steps allowed the synthesis of the desired branched homo-aldol adduct, proposed as a key intermediate for a short synthetic alternative towards enokipodins.
Graphical abstract


Similar content being viewed by others
References
Wasser SP, Weis AL (1999) Crit Rev Immunol 19:65
Wang H, Ng TB, Ooi VE (1998) Mycol Res 102:897
Yaoita Y, Matsuki K, Iijima T, Nakano S, Kakuda R, Machida K, Kikushi M (1998) Chem Pharm Bull 46:944
Hirai Y, Ikeda M, Murayama T, Ohata T (1998) Biosci Biotechnol Biochem 62:1364
Ishikawa NK, Fukushi Y, Yamaji K, Tahara S, Takahashi K (2001) J Nat Prod 64:932
Ishikawa NK, Yamaji K, Tahara S, Fukushi Y, Takahashi K (2000) Phytochemistry 54:777
Ishikawa NK, Yamaji K, Ishimoto H, Miura K, Fukushi Y, Takahashi K (2005) Mycoscience 46:39
Melo MR, Paccola-Meirelles LD, Faria TJ, Ishikawa NK (2009) Mycoscience 50:78
Nascimento CB, Macedo FC Jr (2014) Quim Nova 8:1377
Srikrishna A, Srinivasa Rao M (2004) Synlett 2004:374
Srikrishna A, Lakshini BV, Ravikumar PC (2006) Tetrahedron Lett 47:1277
Secci F, Frongia A, Olliver J, Piras PP (2007) Synthesis 2007:999
Srikrishna A, Srinivasa Rao M (2010) Indian J Chem Sect B 49:1363
Kuwahara S, Saito M (2004) Tetrahedron Lett 45:5047
Kuwahara S, Saito M (2005) Biosci Biotechnol Biochem 69:374
Yoshida M, Shoji Y, Shishido K (2009) Org Lett 11:1441
Luján-Montelongo JA, Ávila-Zárraga JG (2010) Tetrahedron Lett 51:2232
Leboeuf D, Wright CM, Frontier AJ (2013) Chem Eur J 19:4835
Hodgson DM, Chung YK, Nuzzo I, Freixas G, Kulikiewicz KK, Cleator E, Paris JM (2007) J Am Chem Soc 129:4456
Lalić G, Petrovski Z, Galonić D, Matović R, Saičić RN (2000) Tetrahedron Lett 41:763
Lalić G, Petrovski Z, Galonić D, Matović R, Saičić RN (2001) Tetrahedron 57:583
Lalić G, Petrovski Z, Galonić D, Matović R, Saičić RN (2002) J Serbian Chem Soc 67:221
Curci R, Fiorentino M, Serio MR (1984) J Chem Soc Chem Commun 1984:155
Wang Z, Tu Y, Frohn M, Shi Y (1997) J Org Chem 62:2328
Fuganti C, Serra SJ (2000) J Chem Soc Perkin Trans 1:3758
Macedo FC Jr, Andrei CC, Campiom D, Ishikawa NK (2011) Tetrahedron Lett 52:1612
Berens U, Scharf HD (1995) J Org Chem 60:5127
Serra S (2011) Tetrahedron Asymmetry 22:619
Sudalai A, Krishna Rao GS (1989) Indian J Chem 28B:720
Delgado A, Granados R, Mauleon D, Soucheiron I, Feliz M (1985) Can J Chem 63:3186
Lecornué F, Paugam R, Oliver J (2005) Eur J Org Chem 2005:2589
Meinwald J, Labana SS, Chadha MS (1963) J Am Chem Soc 85:582
Mukaiyama T, Banno K, Narasaka K (1974) J Am Chem Soc 96:7503
Acknowledgements
C. B. N. are recipients of scholarships from CAPES. We thank ‘Laboratório Multiusuário de Espectroscopia’ (SPEC)-UEL for acquisition of the NMR spectra.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Nascimento, C.B., Avelar, L.A., Arantes, D.C. et al. Synthetic approach towards cuparene-type sesquiterpenes via highly regioselective epoxide opening under acid catalysis. Monatsh Chem 149, 1899–1904 (2018). https://doi.org/10.1007/s00706-018-2199-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-018-2199-6