Abstract
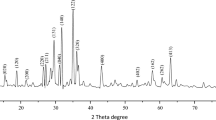
ZnO was successfully synthesized using a simple precipitation method with zinc acetate dihydrate as the starting material and ammonium hydroxide solution as the oxidizer. Morphological analysis showed ZnO particle formation with a Zn/O atomic ratio of 0.75–0.92. The synthesized ZnO product was examined using X-ray diffraction measurement (XRD) with the corresponding major peaks implying a hexagonal wurtzite structure. The average crystal size was 18.26–20.49 nm and the specific surface area was 18.17–21.67 m2/g. The optical band gap was estimated in the range of 2.95–3.21 eV. For photocatalytic activity, the synthesized ZnO was dispersed in two kinds of organic dyes (eosin-Y and methyl orange) with an initial concentration of 10 mg/cm3. The activity was carried out under ultraviolet illumination and the absorbance was measured. The best degradation efficiency was observed with a maximum degradation rate constant of 6.75 × 10−3/min and 1.73 × 10−3/min for eosin-Y and methyl orange, respectively. The degradation efficiency exhibited a direct correlation to the SSA, with the high SSA provided by the nanostructure size exhibiting better degradation efficiency. Therefore, ZnO nanostructures synthesized from zinc acetate dihydrated using an ammonium hydroxide oxidizer was shown to be a potential material for photocatalytic activity.
Graphical abstract










Similar content being viewed by others
References
Danwittayakul S, Jaisai M, Dutta J (2015) Appl Catal B-Environ 163:1
Santhosh C, Velmurugan V, Jacob G, Jeong SK, Grace AN, Bhatnagar A (2016) Chem Eng J 306:1116
Zheng L, Zheng Y, Chen C, Zhan Y, Lin X, Zheng Q, Wei K, Zhu J (2009) Inorg Chem 48:1819
Moreira FC, Boaventura RAR, Brillas E, Vilar VJP (2017) Appl Catal B-Environ 202:217
Aslam M, Charfi A, Lesage G, Heran M, Kim J (2017) Chem Eng J 307:897
Jo WK, Tayade RJ (2014) Chin J Catal 35:1781
Thepnurat M, Chairuangsri T, Hongsith N, Ruankham P, Choopun S (2015) ACS Appl Mater Interfaces 7:24177
Hong LY, Ke HW, Tsai CE, Lin HN (2016) Mater Lett 185:243
Kim YK, Hwang SH, Kim S, Park H, Lim SK (2017) Sensor Actuat B-Chem 240:1106
Ponhan W, Thepnurat M, Phadungdhitidhada S, Wongratanaphisan D, Choopun S (2016) Surf Coat Tech 306:41
Ruankham P, Choopun S, Wongratanaphisan D, Sagawa T (2016) Integr Ferroelectr 175:113
Chava RK, Kang M (2017) J Alloys Compd 692:67
Qin J, Zhang X, Yang C, Cao M, Ma M, Liu R (2017) Appl Surf Sci 392:196
Li W, Gao S, Li L, Jiao S, Yu Q, Li H, Wang J, Yu Q, Zhang Y, Wang D (2016) Mater Lett 185:161
Wang J, Pei C, Cheng L, Wan W, Zhao Q, Yang H, Liu S (2016) Sensor Actuat B-Chem 223:650
Lang J, Wang J, Zhang Q, Li X, Han Q, Wei M, Sui Y, Wang D, Yang J (2016) Ceram Int 42:14175
Ng SS, Ooi PK, Yaakob S, Abdullah MJ, Hassan HA, Hassan Z (2014) Sains Malays 43:1077
Jia T, Fu F, Long F, Min Z, Zhao J, Chen J, Li J (2016) Ceram Int 42:13893
Mamaghani AH, Haghighat F, Lee CS (2017) Appl Catal B-Environ 203:247
Khuili M, Fazouan N, El Makarim HA, El Halani G, Atmani EH (2016) J Alloys Compd 688:368
Zhang P, Li X, Wu X, Zhao T, Wen L (2016) J Alloys Compd 673:405
Rajamanickam D, Dhatshanamurthi P, Shanthi M (2015) Spectrochim Acta A 138:489
Nouri A, Fakhri A (2015) Spectrochim Acta A 138:563
Zhang L, Dai CH, Zhang XX, Liu YN, Yan JH (2016) Trans Nonferrous Met Soc China 26:2380
Akhil K, Jayakumar J, Gayathri G, Khan SS (2016) J Photoch Photobio B 160:32
Wang Y, Zhu L, Wang M, Zhang M, Yang Y, Zhu Q, Lei Y (2016) Int J Hydrogen Energy 41:15703
Mageshwari K, Mali SS, Sathyamoorthy R, Patil PS (2013) Powder Technol 249:456
Shen Y, Wang W, Xiao K (2016) J Environ Chem Eng 4:1259
Rezaei M, Salem S (2016) Spectrochim Acta A 167:41
Acknowledgements
This work was supported by the Kasetsart University Research and Development Institute (KURDI), Kasetsart University, Bangkok, Thailand.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Moungsrijun, S., Sujinnapram, S. & Sutthana, S. Synthesis and characterization of zinc oxide prepared with ammonium hydroxide and photocatalytic application of organic dye under ultraviolet illumination. Monatsh Chem 148, 1177–1183 (2017). https://doi.org/10.1007/s00706-017-1959-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-017-1959-z