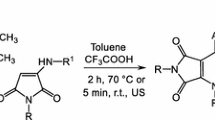
Abstract
An efficient, straightforward, and general method for the synthesis of pyrido[1,2-a]pyrazine and pyrazino[1,2-a]quinoline derivatives via a one-pot three-component reaction between cyclic enamineones, aromatic aldehydes, and 1,3-dicarbonyl compound is reported. On our knowledge, this is the first reported one-pot multicomponent reaction for synthesis of pyrido[1,2-a]pyrazine and pyrazino[1,2-a]quinoline derivatives. A proposed reaction pathway for the synthesis has been proved experimentally, consisting in addition of 3-(alkoxycarbonylmethylidene)piperazine-2-ones to the products of condensation of aromatic aldehydes and 1,3-dicarbonyl compounds, followed by further cyclocondensation Michael addition. The structure of the thirteen new compounds was proven by elemental and spectral analysis (1H and 13C NMR, HPLC/MS, and FT-IR).
Graphical abstract

Similar content being viewed by others
References
Nielsen TE, Schreiber SL (2008) Angew Chem Int Ed 47:48
Mangalagiu II (2011) Curr Org Chem 15:730
Dawidowski M, Herold F, Chodkowski A, Kleps J (2012) Eur J Med Chem 48:347
Pettersson M, Johnson DS, Humphrey JM, Wood KM, Verhoest PR (2015) Bioorg Med Chem Lett 25:908
Mathew B, Srivastava S, Ross LJ, Lenaerts AJ, Reynolds RC (2011) Bioorg Med Chem 19:7120
Rao VA, Jain PC, Anand N, Srimal RC, Dua PR (1970) J Med Chem 13:516
Singh GB, Nityanand S, Srimal RC, Rao VA, Jain FC, Dhawan BN (1973) Experientia 29:1529
Singh GB, Srimal RC, Dhawan BN (1978) Arzneim Forsch 28:1403
Saxena AK, Murthy VA, Jain PC, Srimal RC, Anand N (1980) Indian J Chem, Sect B 19B:879
Baxter CA, Richards HC (1972) J Med Chem 15:351
Pellegrino J, Pereira LH, Mello RT, Katz N (1974) J Parasitol 60:723
Seymour PA (1992) Synergistic combinations in the treatment of anxiety. US Patent 5,124,346 Jun 23, 1992. Chem Abstr 117:157647
Boyfield I, Codwell MC, Hadley MS, Healy MA, Johns A, Nash DJ, Riley GJ, Scott EE, Smith SA, Stemp G, Wilson K (1996) Bioorg Med Chem Lett 6:1227
Harrington MA, Sleight AJ, Pitha J, Peroutka SJ (1991) Eur J Pharmacol 194:83
Sleight AJ, Peroutka SJ (1991) Naunyn Schmiedeberg’s Arch Pharmacol 343:109
Mokrosz JL, Boksa J, Bojarski AJ, Charakchieva-Minol S (1993) Med Chem Res 3:240
Kobayashi M, Yoshinaga T, Seki T, Wakasa-Morimoto C, Brown KW, Ferris R, Foster SA, Hazen RJ, Miki S, Suyama-Kagitani A, Kawauchi-Miki S, Taishi T, Kawasuji T, Johns BA, Underwood MR, Garvey E, Sato A, Fujiwara T (2011) Antimicrob Agents Chemother 2:813
Yoshinaga T, Kobayashi M, Seki T, Miki S, Wakasa-Morimoto C, Suyama-Kagitani A, Kawauchi-Miki S, Taishi T, Kawasuji T, Johns BA, Underwood MR, Garvey EP, Sato A, Fujiwara T (2015) Antimicrob Agents Chemother 1:397
Kothandaraman S, Guiadeen D, Butora G, Doss G, Mills SG, MacCoss M, Yan L (2009) Tetrahedron Lett 50:4050
Cai G, Zhu W, Ma D (2006) Tetrahedron 62:5697
Garkusha GA (1965) Zh Org Khim 1:2222
Takahashi K, Tachiki A, Ogura K, Iida H (1986) Heterocycles 24:2835
Jezo I, Luzak I (1964) Chem Zvesti 18:186
Nair V, Devipriya S, Suresh E (2008) Tetrahedron 64:3567
Weber L (2002) Drug Discov Today 7:143
Zhu HJ, Bienayme H (2005) Multicomponent Reactions. Wiley, Weinheim, p 484
Balme G, Bossharth E, Monteiro N (2003) J Org Chem 21:4101
Yan J, Xu Y, Zhuang F, Tian J, Zhang G (2016) Mol Divers 20:567
Vovk MV, Kushnir OV, Melnichenko NV, Tsymbal IF (2011) Chem Heterocycl Comp 8:989
Kawahara N, Shimamori T, Itoh T, Takayanagi H, Ogura H (1987) Chem Pharm Bull 2:457
Moradi L, Piltan M, Rostami H, Abasi G (2013) Chin Chem Lett 24:740
Piltan M, Moradi L, Abasi G, Zarei AS (2013) Beilstein J Org Chem 9:510
Iwanami VY (1971) Bull Chem Soc Jpn 44:1311
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Eften’eva, R.I., Kushnir, O.V., Lyavinets, O.S. et al. Synthesis of polyfunctionalized pyrido[1,2-a]pyrazines and pyrazino[1,2-a]quinolines via one-pot multicomponent reactions. Monatsh Chem 147, 2127–2133 (2016). https://doi.org/10.1007/s00706-016-1836-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-016-1836-1