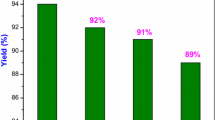
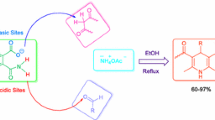
Abstract
The reaction of aromatic aldehydes, dimedone, and malononitrile in the presence of ammonium acetate has afforded tetrahydrobenzo[b]pyrans instead of polyhydroquinolines under both grinding and reflux conditions; thus ammonium acetate acts as a catalyst for this transformation instead of a reactant. Polyhydroquinoline derivatives were also synthesized via a one-pot condensation of aromatic aldehydes, malononitrile and an enaminone, which was prepared by the reaction of dimedone and ammonium acetate, in refluxing ethanol in high yields.
Graphical abstract


Similar content being viewed by others
References
Lu P, Wang YG (2010) Synlett 165
Ganem B (2009) Acc Chem Res 42:463
Dömling A (2006) Chem Rev 106:17
Ramon DJ, Yus M (2005) Angew Chem Int Ed 44:1602
Orru RVA, De Greef M (2003) Synthesis 1471
Ugi I, Heck S (2001) Comb Chem High Throughput Screen 4:1
Weber L, Illgen K, Almstetter M (1999) Synlett 366
Gao Sh, Tsai ChH, Tseng Ch, Ch-Fa Yao (2008) Tetrahedron 64:9143
Bonsignore L, Loy G, Secci D, Calignano A (1993) Eur J Med Chem 28:517
Saini A, Kumar S, Sandhu JS (2006) Synlett 1928
Singh K, Singh J, Singh H (1996) Tetrahedron 52:14273
Witte EC, Neubert P, Roesch A (1986) 7-(Piperazinylpropoxy)-2H-1-benzopyran-2-ones. Ger Offen DE 3427985, Jan 30, 1986, Chem Abstr 104:224915f
Wang XS, Shi DQ, Tu ST, Yao CS (2003) Synth Commun 33:119
Hatakeyama S, Ochi N, Numata H, Takano S (1988) J Chem Soc Chem Commun 17:1202
Fokialakis N, Magiatis P, Chinou L, Mitaku S, Tillequin F (2002) Chem Pharm Bull 50:413
Morgan LR, Jursic BS, Hooper CL, Neumann DM, Thangaraj K, Leblanc B (2002) Bioorg Med Chem Lett 12:3407
Beagley P, Blackie MAL, Chibale K, Clarkson C, Meijboom R, Moss JR, Smith P, Su H (2003) Dalton Trans 3046
Ryckebusch A, Derprez-Poulain R, Maes L, Debreu-Fontaine MA, Mouray E, Grellier P, Sergheraert C (2003) J Med Chem 46:542
Sabitha G, Reddy GSKK, Reddy CS, Yadav JS (2003) Tetrahedron Lett 44:4129
Ji SJ, Jiang ZQ, Lu J, Loa TP (2004) Synlett 831
Sridhar R, Perumal PT (2005) Tetrahedron 61:2465
Breitenbucher JG, Figliozzi G (2000) Tetrahedron Lett 41:4311
Dondoni A, Massi A, Minghini E, Bertolasi V (2004) Tetrahedron 60:2311
Wang LM, Sheng J, Zhang L, Han JW, Fan ZY, Tian H, Qian CT (2005) Tetrahedron 61:1539
Evans CG, Jinwal UK, Makley LN, Dickey CA, Gestwicki JE (2011) Chem Commun 47:529
Tu SJ, Zhou JF, Deng X, Cai PJ, Wang H, Feng JC (2001) Chin J Org Chem 21:313
Reddy CS, Raghu M (2008) Chin Chem Lett 19:775
Mekheimer RA, Hameed AA, Sadek KU (2008) Green Chem 10:592
Nagarapu L, Apuri S, Gaddam S, Bantu R, Mahankhali VC, Kantevari S (2008) Lett Org Chem 5:60
Kumar A, Maurya RA (2007) Tetrahedron Lett 48:3887
Kumar A, Maurya RA (2007) Tetrahedron 63:1946
Karade NN, Budhewar VH, Shinde SV, Jadhave WN (2007) Lett Org Chem 4:16
Heravi MM, Bakhtiari K, Javadi NM, Bamoharram FF, Saeedi M, Oskooie HA (2007) J Mol Cat A Chem 264:50
Song G, Wang B (2005) Synth Commun 35:2875
Foroughifar N, Mobinikhaledi A, Bodaghifard MA, Moghanian H, Ebrahimi S (2009) Synth React Inorg Met-Org, Nano-Met Chem 39:161
Surasani R, Kalita D, Rao AVD, Yarbagi K, Chandrasekhar KB (2012) J Fluorine Chem 135:91
Bandgar BP, More PE, Kamble VT, Totre JV (2008) Arkivoc xv:1
Suarez M, Verdecia Y, Ochoa E, Martin N, Martinez R, Quinteiro M, Seoane C, Soto JL, Novoa H, Blaton N, Peeters OM, De Ranter C (2000) J Heterocycl Chem 37:35
Tu Sh, Zhang J, Zhu X, Zhang Y, Wang Q, Xu J, Jiang B, Jia R, Zhang J, Shi F (2006) J Heterocycl Chem 43:985
Kumar S, Sharma P, Kapoor KK, Hundal MS (2008) Tetrahedron 64:536
Undale KA, Park Y, Park K, Dagade DH, Pore DM (2011) Synlett 6:791
Patil D, Chandam D, Mulik A, Jagdale S, Patil P, Deshmukh M (2014) J Saudi Chem Soc. doi:10.1016/j.jscs.2014.04.001
Moshtaghi ZA, Eskandari I, Khavasi HR (2012) Tetrahedron Lett 53:5519
Moshtaghi ZA, Eskandari I, Moghani D (2012) Chem Sci Trans 1:91
Moshtaghi ZA, Moghani D, Okhravi S (2014) Curr Chem Lett 3:71
Moshtaghi ZA, Raeisolsadati OM (2013) J Chin Chem Soc 60:275
Banerjee S, Horn A, Khatri H, Sereda G (2011) Tetrahedron Lett 52:1878
Jin TS, Wang AQ, Shi F, Han LS, Lio LB, Li TS (2006) Arkivoc xiv:78
Montazeri N, Noghani T, Ghorchibeigy M, Zoghi RE (2014) J Chem Article ID 596171
Cadogan JIG, Ley SV, Pattenden G, Raphael RA, Rees CW (1992) Dictionary of organic compounds, vol 1, 6th edn. Chapman & Hall, London, p 213
Acknowledgments
The authors sincerely acknowledge the Research Office of Azarbaijan Shahid Madani University for financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Moshtaghi Zonouz, A., Okhravi, S. & Moghani, D. Ammonium acetate as a catalyst and/or reactant in the reaction of dimedone, aromatic aldehyde, and malononitrile: synthesis of tetrahydrobenzo[b]pyrans and hexahydroquinolines. Monatsh Chem 147, 1819–1824 (2016). https://doi.org/10.1007/s00706-016-1683-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-016-1683-0