Abstract
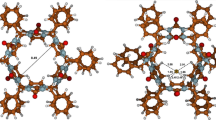
Using quantum mechanical calculations, the most probable structures of the anionic complex species dodecabenzylbambus[6]uril–Cl−, dodecabenzylbambus[6]uril–Br−, and dodecabenzylbambus[6]uril–I− were derived. In these three complexes, each of the considered univalent halide anions, included in the center of the macrocyclic cavity, is bound by 12 weak C–H···X− (X− = Cl−, Br−, I−) hydrogen bonds between methine hydrogen atoms on the convex face of glycoluril units and the respective anion. The lengths of these C–H···X− hydrogen bonds increase in the series of Cl− < Br− < I−.
Graphical abstract





Similar content being viewed by others
References
Lagona J, Mukhopadhyay P, Chakrabarti S, Isaacs L (2005) Angew Chem Int Ed 44:4844
Lee JW, Samal S, Selvapalam N, Kim HJ, Kim K (2003) Acc Chem Res 36:621
Liu S, Ruspic C, Mukhopadhyay P, Chakrabarti S, Zavalij PY, Isaacs L (2005) J Am Chem Soc 127:15959
Freeman WA, Mock WL, Shih NY (1981) J Am Chem Soc 103:7367
Mock WL, Shih NY (1983) J Org Chem 48:3618
Mock WL, Shih NY (1986) J Org Chem 51:4440
Mock WL, Shih NY (1988) J Am Chem Soc 110:4706
Mock WL, Shih NY (1989) J Am Chem Soc 111:2697
Isobe H, Tomita N, Lee JW, Kim HJ, Kim K, Nakamura E (2000) Angew Chem Int Ed 39:4257
Isobe H, Sota S, Lee JW, Kim HJ, Kim K, Nakamura E (2005) Chem Commun 1549–1551
Tan Y, Choi S, Lee JW, Ko YH, Kim K (2002) Macromolecules 35:7161
Márquez C, Hudgins RR, Nau WM (2004) J Am Chem Soc 126:5806
Buschmann HJ, Mutihac L, Mutihac RC, Schollmeyer E (2005) Thermochim Acta 430:79
Buschmann HJ, Schollmeyer E, Mutihac L (2003) Thermochim Acta 399:203
Svec J, Necas M, Sindelar V (2010) Angew Chem Int Ed 49:2378
Havel V, Svec J, Wimmerova M, Dusek M, Pojarova M, Sindelar V (2011) Org Lett 13:4000
Kříž J, Dybal J, Makrlík E, Budka J, Vaňura P (2009) J Phys Chem A 113:5896
Kříž J, Toman P, Makrlík E, Budka J, Shukla R, Rathore R (2010) J Phys Chem A 114:5327
Kříž J, Dybal J, Makrlík E, Vaňura P, Moyer BA (2011) J Phys Chem B 115:7578
Makrlík E, Toman P, Vaňura P (2013) Monatsh Chem 144:919
Makrlík E, Toman P, Vaňura P (2013) Monatsh Chem 144:1461
Makrlík E, Toman P, Vaňura P (2014) Monatsh Chem 145:551
Turi L, Dannenberg JJ (1993) J Phys Chem 97:2488
Rode JE, Dobrowolski JC (2002) Chem Phys Lett 360:123
Lee C, Yang W, Parr RG (1988) Phys Rev B 37:785
Becke AD (1993) J Chem Phys 98:5648
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery Jr JA, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas Ö, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) Gaussian 09, Revision C.01. Gaussian Inc, Wallingford, CT
Acknowledgments
This work was supported by the Grant Agency of Faculty of Environmental Sciences, Czech University of Life Sciences, Prague, Project No.: 42900/1312/3114 entitled “Environmental Aspects of Sustainable Development of Society,” and by the Czech Ministry of Education, Youth, and Sports (Project MSMT No.: 20/2015).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Böhm, S., Makrlík, E. & Vaňura, P. Theoretical study on the complexation of the chloride, bromide, and iodide anions with dodecabenzylbambus[6]uril. Monatsh Chem 147, 697–703 (2016). https://doi.org/10.1007/s00706-015-1627-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-015-1627-0