Abstract
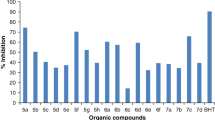
This work describes a facile synthesis and characterization of a new series of pyrazole containing pyrimidine, 1,4-dihydropyridine, and imidazole derivatives using substituted 4-formylpyrazole as a key intermediate via multicomponent reaction sequence. The structures of these unknown compounds were elucidated by IR, 1H NMR, 13C NMR, LC–MS, and elemental analysis. The synthesized products were screened for their in vitro antimicrobial and antioxidant properties. Among the tested 3-(3,4-dihalophenyl)-1-phenyl-1H-pyrazol-4-yl, incorporated acetyl dihydropyrimidine compounds exhibited promising antimicrobial activity and DPPH radical scavenging activity with levels of inhibition at 89.4 and 83.3 %, respectively.
Graphical abstract

Similar content being viewed by others
References
Hitchock CA (1993) Biochem Soc Trans 21:1039
Amir M, Javed SA, Hassan MZ (2012) Med Chem Res 21:1261
Walsh C (2000) Nature 406:775
Laguerre M, Lecomte J, Villeneuve P (2007) Prog Lipid Res 46:244
Aruoma OI (1998) J Am Oil Chem Soc 75:199
Winkler C, Frick B, Schroecksnadel K, Schennach H, Fuchs D (2006) Food Chem Toxicol 44:2003
Malladi S, Isloor AM, Peethambar SK, Fun HK (2013) Med Chem Res 22:2654
Thumar NJ, Patel MP (2011) Med Chem Res 21:1751
Zelenin KN, Bezhan IP, Pastushenkov LV, Gromova EG, Lesiovskaja EE, Chakchir BA, Melnikova LF (1999) Arzneimittelforsch 49:843
Hall A, Billinton A, Brown SH, Clayton NM, Chowdhury A, Gerald MP, Goldsmith GP, Hayhow TG, Hurst DN, Kilford IR, Naylor A, Passingham B (2008) Bioorg Med Chem Lett 18:3392
Rashad AE, Hegab MI, Abdel-Megeid RE, Micky JA, Abdel-Megeid FME (2008) Bioorg Med Chem 16:7102
Kumar KA, Jayaroopa P (2013) Int J PharmTech Res 5:1473
Chauhan A, Sharma PK, Kaushik N (2011) Int J ChemTech Res 3:11
Jamwal A, Javed A, Bhardwaj V (2013) J Pharm BioSci 3:114
Kumar P, Chandak N, Kaushik P, Sharma C, Kaushik D, Aneja KR, Sharma PK (2012) Med Chem Res 21:3396
El-Sabbagh OI, Baraka MM, Ibrahim SM, Pannecouque C, Andrei G, Snoeck R, Balzarin J, Rashad AA (2009) Eur J Med Chem 44:3746
Sadanadam YS, Shetty MM, Diwan PV (1992) Eur J Med Chem 27:87
Baranda AB, Alonso RM, Jimenez RM, Weinmann W (2006) Forensic Sci Int 156:23
Klegeris A, Liutkevicius E, Mikalauskiene G, Duburs G, McGeer PL, Klusa V (2002) Eur J Pharmacol 441:203
Swarnalatha G, Prasanthi G, Sirisha N, Chetty CM (2011) Int J ChemTech Res 3:75
Zhou K, Wang X, Zhao Y, Cao Y, Fu Q, Zhang S (2011) Med Chem Res 20:1325
Achar KCS, Hosamani KM, Seetharamareddy HR (2010) Eur J Med Chem 45:2048
Vijesh AM, Isloor AM, Telkar S, Peethambar SK, Rai S, Isloor N (2011) Eur J Med Chem 46:3531
Sharma V, Chitranshi N, Agarwal AK (2014) Int J Med Chem 2014:202784
Tirzitis A, Tirzite D, Hyvonen Z (2001) Czech J Food Sci 19:81
Kalidhar U, Kaur A (2011) Res J Pharm Biol Chem Sci 2:1116
Zarghi A, Arfaei S, Ghodsi R (2012) Med Chem Res 21:1803
Viveka S, Dinesha, Laxmeshwar SS, Nagaraja GK (2012) Molbank 3:M776
Viveka S, Prabhuswamy M, Dinesha, Lokanath NK, Nagaraja GK (2014) Mol Cryst Liq Cryst 588:83
Dinesha, Viveka S, Naik P, Nagaraja GK (2014) Med Chem Res 23:4189
Oktay M, Gulcin I, Kufrevioglu OI (2003) LWT Food Sci Technol 36:263
Samshuddin S, Narayana B, Sarojini BK, Madhu LN (2013) Med Chem Res 22:3002
Nandakumar A, Thirumurugan P, Perumal PT, Vembu P, Ponnuswamy MN, Ramesh P (2010) Bioorg Med Chem Lett 20:4252
James D, Lowry M, Jaqua MJ, Selepak ST (1970) Appl Microbiol 20:46
Fenlon CH, Cynamon MH (1986) Antimicrob Agents Chemother 29:386
Arthington-Skaggs BA, Motley M, Warnock DW, Morrison CJ (2000) J Clin Microbiol 38:2254
Blois MS (1958) Nature 181:1199
Oyaizu M (1986) Japan J Nutr 44:307
Acknowledgments
The authors gratefully acknowledge the University Grants Commission (UGC) and Promotion of University Research and Scientific Excellence (PURSE) for the financial assistance. They are grateful to IISC Bangalore and USIC Mangalore University for providing the spectral analysis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Viveka, S., Dinesha, Madhu, L.N. et al. Synthesis of new pyrazole derivatives via multicomponent reaction and evaluation of their antimicrobial and antioxidant activities. Monatsh Chem 146, 1547–1555 (2015). https://doi.org/10.1007/s00706-015-1428-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-015-1428-5