Abstract
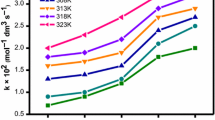
The kinetics of reduction of the surfactant complex ions cis-[Co(phen)2(C12H25NH2)2]3+ and cis-[Co(bpy)2(C12H25NH2)2]3+ (phen = 1,10-phenanthroline, bpy = 2,2′-bipyridine, C12H25NH2 = dodecylamine) in solutions of unilamellar vesicles of dipalmitoylphosphatidylcholine (DPPC) were studied at different temperatures by a spectrophotometric method under pseudo-first-order conditions using an excess of the reductant. Experimentally the reaction was found to be second order and the electron transfer postulated as outer sphere. Below the phase temperature of DPPC the second-order rate constant for this electron-transfer reaction of both the complexes was found to decrease with increasing concentration of the liposome. Above the phase transition temperature the reaction rate was found to increase with increasing concentration of DPPC. Below the phase transfer temperature the results have been explained on the basis of electrostatic double layer repulsion which prevents the vesicles from accumulation of surfactant–cobalt(III) complexes in the interior of the vesicle membrane. Above the phase transfer temperature the release of the surfactant–cobalt(III) complex molecules from the interior core to the exterior surface of the vesicle through the strong hydrophobic effect increases the reaction rate. The main driving force for this phenomenon is considered to be the intervesicular hydrophobic interaction between the surfactant complexes and vesicles surface. We could observe isokinetic relationships for both the complexes of the present study at the phase transfer temperature of DPPC.
Graphical abstract











Similar content being viewed by others
References
Almog S, Litman BJ, Wimley W, Cohen J, Wachtel EJ, Barenholz Y, Ben-Shaul A, Lichtenberg D (1990) Biochemistry 29:4582
Zheng Y, Lin Z, Zakin JL, Talmon Y, Davis HT, Scriven CE (2000) J Phys Chem B 104:5263
Parteaarroyo A, Alonso A, Gone FM, Tryout M, Parades S (1996) J Colloid Interface Sci 178:156
Wenk MR, Seelig J (1997) J Phys Chem B 101:5224
Lichtenberg D, Robinson RJ, Dennis EA (1983) Biophys Acta 737:285
Gulati M, Grover M, Singh S, Singh M (1998) Int J Pham 165:129
Paternostre M, Meyer O, Grabielle-Madelmont C, Leisure S, Shaman S, Olivine M (1995) Biophys J 69:2476
Jortner J, Bixon M (eds) (1999) Electron transfer—from isolated molecules to biomolecules. Advances in chemical physics, vol 106–107. Wiley, New York
Babich OA, Gould ES (2002) Inorg Chim Acta 336:80
Hopfield JJ, Onuchic JN, Beratan DN (1989) J Phys Chem 93:6350
Prado-Gotor R, Jimmenez R, Lopez B, Perez B, Herera CG, Sanchez F (1998) Langmuir 14:1539
Pal SK, Mandal D, Sukul D, Bhattacharyya K (1999) Chem Phys 249:63
Schwögler A, Carell LTB (2013) Angew Chem Int Ed 39:3918
Borsarelli CD, Cosa JJ, Previtali CM (1998) Photochem Photobiol 68:438
Hackett JWI, Turro C (1998) J Phys Chem A 102:5728
Lerebours B, Chevalier Y, Pileni MP (1985) Chem Phys Lett 117:89
Gaswick D, Haim A (1971) J Am Chem Soc 93:7347
Miralles AJ, Szecsy AP, Haim A (1982) Inorg Chem 21:697
Sasikala K, Arunachalam S (2009) Colloids Surf A 335:98
Sasikala K, Arunachalam S (2010) J Dispersion Sci Technol 31:1618
Kumaraguru N, Santhakumar K, Arunachalam S, Arumugam MN (2006) Polyhedron 25:3253
Santhakumar K, Kumaraguru N, Arunachalam S, Arumugam MN (2006) Trans Met Chem 31:475
Kumaraguru N, Santhakumar K (2006) Int J Chem Kinet 38:629
Nagaraj K, Arunachalam S (2012) Trans Met Chem 37:423
Sasikala K, Arunachalam S (2010) Colloid J 72:530
Sasikala K, Arunachalam S (2010) Monatsh Chem 141:309
Jones GR, Cosins AR (1989) In: New RRC (ed) Liposomes: a practical approach. IRL, Oxford
Subuddhi U, Mishra AK (2006) Photochem Photobiol 5:283
Eeman M, Deleu M, Eeman M (2010) Biotechnol Agron Soc Environ 14:719
Tumuli MS, Fondler JH (1981) J Am Chem Soc 103:2507
Arulsamy N, Bohle DS, Goodson PA, Jaeger DA, Reddy VB (2001) Inorg Chem 40:836
McBane GC (1998) J Chem Educ 75:919
Sasikala K, Senthil Kumar R, Arunachalam S (2010) Z Phys Chem 224:1411
Cannon RD, Gardiner JJ (1972) J Chem Soc Dalton Trans 89:887
Kitson RE (1950) Anal Chem 22:664
Link RG (1970) Inorg Chem 9:2529
Acknowledgments
We are grateful to the UGC-SAP & COSIST and DST-FIST programmes of the Department of Chemistry, Bharathidasan University, and UGC-SAP RFSMS Scholarship sanctioned to one of the authors, K. Nagaraj, by University Grants Commission (UGC), New Delhi. Financial assistance from the CSIR (Grant No. 01(2461)/11/EMR-II), DST (Grant No. SR/S1/IC-13/2009), and UGC (Grant No. 41-223/2012(SR)) sanctioned to S. Arunachalam is also gratefully acknowledged. We thank the referee for enlightening us on the possibility of false correlation in isokinetic relationships.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nagaraj, K., Arunachalam, S. Kinetics of reduction of cis-bis(dodecylamine)bis(1,10-phenanthroline)cobalt(III) perchlorate and cis-bis(dodecylamine)bis(2,2′-bipyridine)cobalt(III) perchlorate by Fe(II) in dipalmitoylphosphatidylcholine vesicles. Monatsh Chem 145, 427–433 (2014). https://doi.org/10.1007/s00706-013-1080-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-013-1080-x