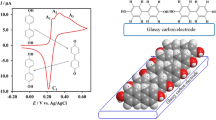
Abstract
The electrochemical oxidation of some dihydroxybenzoic acids (DHBAs), 2,5-, 2,3-, and 3,4-dihydroxybenzoic acid, has been studied using cyclic voltammetry and chronocoulometry. It was found that the position of the carboxylic acid group on the phenyl ring is an effective factor in electrochemical oxidation of these molecules. The electrochemical oxidation of 2,5-DHBA and 2,3-DHBA shows the features of an ECE mechanism. Based on the voltammetric results, hydroxylation is considered to be the subsequent chemical reaction of electrochemical oxidation of 2,5-DHBA and 2,3-DHBA. In addition, the pH values were found to have great impacts on the hydroxylation rate of oxidized forms of 2,5-DHBA and 2,3-DHBA, which decreases with increasing pH. In contrast with 2,5-DHBA and 2,3-DHBA, our results show that the electrochemical oxidation of 3,4-DHBA is a simple electron transfer (E r). In addition, our results show that both diffusion and adsorption processes control the electrochemical oxidation pathway. Using chronocoulometry, the amount of surface excess for each species was obtained, and their orientations on the surface of a glassy electrode were estimated.
Graphical abstract












Similar content being viewed by others
References
Costentin C (2008) Chem Rev 108:2145
Savéant JM (2012) Energy Environ Sci 5:7718
Houmam A (2008) Chem Rev 108:2180
Savéant JM (2008) Chem Rev 108:2111
Lund H, Hammerich O (2001) Organic electrochemistry. Marcel Dekker, Inc., New York
Zare HR, Namazian M, Coote ML (2009) Electrochim Acta 54:5353
Chaudhari VR, Bhat MA, Ingole PP, Haram SK (2009) Electrochem Commun 11:994
Costentin C, Louault C, Robert M, Savéant JM (2008) J Am Chem Soc 130:15817
Laviron E (1995) J Electroanal Chem 391:187
Quan M, Sanchez D, Wasylkiw MF, Smith DK (2007) J Am Chem Soc 129:12847
Hapiot P, Neudeck A, Pinson J, Fulcrand H, Neta P, Rolando C (1996) J Electroanal Chem 405:169
Sokolová R, Ramešová Š, Degano I, Hromadová M, Gál M, Žabka J (2012) Chem Commun 28:3433
Timbola AK, de Souza CD, Giacomelli C, Spinelli A (2006) J Braz Chem Soc 17:139
Nematollahi D, Shayani-Jam H, Alimoradi M, Niroomand S (2009) Electrochim Acta 54:7407
Bard AJ, Faulker LR (2001) Electrochemical methods, 2nd edn. Wiley, New York, p 339
Vecchio S, Brunetti B (2011) Thermochim Acta 515:84
Babich H, Sedletcaia A, Kenigsberg B (2002) Pharmacol Toxicol 91:245
Lima Leite RH, Cognet P, Wilhelm AM, Delmas H (2003) J Appl Electrochem 33:693
Borah JM, Sarma J, Mahiuddin S (2011) Colloid Surface A 387:50
Tyson CA, Levalley SE, Chan R, Hobbs PD, Dawson MI (1984) J Pharmacol Exp Ther 228:676
Kotsaki-Kovatsi VP, Vafiadou AJ, Koehler-Samuilidou G, Kovatsis A (1997) Vet Hum Toxicol 39:211
Kable EP, Parsons PG (1988) Biochem Pharmacol 37:1711
Aletras V, Karaliota A, Kamariotaki M, Hatzipanayioti D, Hadjiliadis N (2001) Inorgan Chim Acta 312:151
Trabelsi SK, Tahar NB, Abdelhedi R (2004) Electrochim Acta 49:1647
Ren W, Luo HQ, Li NB (2006) Sensors 6:80
Moghaddam AB, Kobarfard F, Fakhari A, Nematollahi D, Davarani SH (2005) Electrochim Acta 51:739
Hatzipanayioti D, Karaliota A, Kamariotaki M, Veneri A (1998) Transition Met Chem 23:407
Andreozzi R, Caprio V, Marotta R (2002) Water Res 36:2761
Wang J, Li M, Shi Z, Li N, Gu Z (2001) Electrochim Acta 47:651
Costentin C, Robert M, Savéant JM (2006) J Am Chem Soc 128:8726
Pariente F, Tobalina F, Darder M, Lorenzo E, Abruña HD (1996) Anal Chem 68:3135
Bodini M, Osorio C, del Valle MA, Arancibia V, Muñoz G (1995) Polyhedron 14:2933
Sazou D, Papadopoulos N (1986) Can J Chem 64:11
Papouchado L, Petri G, Sharp GH, Adams RN (1968) J Am Chem Soc 90:5620
Papouchado L, Petri G, Adams RN (1972) J Electroanal Chem 38:389
Anson FC (1966) Anal Chem 38:54
Wu Y, Bull FS (2008) Korean Chem Soc 29:38
Nematollahi D, Dehdashtian S, Niazi A (2008) J Electroanal Chem 616:79
Bailey SI (1985) Ritchie IM 30:3
Nematollahi D, Esmaili R (2011) Electrochim Acta 56:3899
Nematollahi D, Amani A, Tammari E (2007) J Org Chem 72:3646
Nematollahi D, Esmaili R (2010) J Iran Chem Soc 7:260
Nematollahi D, Khoshsafar H (2009) Tetrahedron 65:4742
Yannai S (ed) (2010) Dictionary of food compounds, 2nd edn. CRC Press, Boca Raton
Kiss T, Atkari K, Bojczuk MJ, Decock P (1993) J Coord Chem 29:81
Nematollahi D, Rafiee M (2005) Green Chem 7:638
Golabi SM, Nematollahi D (1997) J Electroanal Chem 430:141
Simić AZ, Verbić TZ, Sentić MN, Vojić MP, Juranić IO, Manojlović DD (2013) Monatsh Chem 144:121
Adams RN (1969) Electrochemistry at solid electrodes. Marcel Dekker, New York (chapters 3 and 8)
Bowling R, McCreery RL (1988) Anal Chem 60:605
Soriaga MP, Hubbard AT (1982) J Am Chem Soc 104:3937
Soriga MP, Wilson PH, Hubbard AT (1982) J Electroanal Chem 142:317
Pauling LC (1960) The nature of the chemical bond, 3rd edn. Cornell University Press, New York, p 221
Beiginejad H, Nematollahi D, Varmaghani F (2013) J Electrochem Soc 160:H41
Acknowledgments
We acknowledge the Bu-Ali Sina University Research Council and Center of Excellence in Development of Environmentally Friendly Methods for Chemical Synthesis (CEDEFMCS) for their support of this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Beginejad, H., Nematollahi, D., Varmaghani, F. et al. Investigation of the electrochemical behavior of some dihydroxybenzoic acids in aqueous solution. Monatsh Chem 144, 1481–1488 (2013). https://doi.org/10.1007/s00706-013-1031-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-013-1031-6